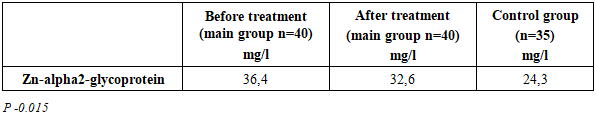-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 716-718
doi:10.5923/j.ajmms.20261602.68
Received: Dec. 18, 2025; Accepted: Jan. 12, 2026; Published: Feb. 26, 2026

The Role of Some Biochemical Markers in the Women with External Genital Endometriosis
Kurbanov B. B., Kochovskaya M. D.
Tashkent State Medical University, Uzbekistan
Correspondence to: Kurbanov B. B., Tashkent State Medical University, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

External genital endometriosis is one of the most dangerous diseases in reproductive age, which also have social aspects. The aim of the study was to determine the role of the specific marker Zn-alpha2-glycoprotein in the diagnosis of the disease. Materials and methods: We studied 40 women diagnosed with External genital endometriosis grade I-II (main group). The control group consisted of 45 conditionally healthy women. Results: All examined women in the main group complained of pelvic pain (100%) and menstrual dysfunction (in the form of polymenorrhea). When assessing pelvic pain, women noted pain mainly during menstruation and during sexual intercourse. Less often, pain was noted during stress, as well as physical exercise. According to the classification of pelvic pain (SRI 2022), all women in the main group noted stage 3 or 4 pain (with a maximum of 5-6). Conclusions: Thus, it can be stated about the importance of determining the Zn-alpha2-glycoprotein marker in the early diagnosis of external genital endometriosis in women.
Keywords: Endometriosis, Ovary, Zn-alpha2-glycoprotein
Cite this paper: Kurbanov B. B., Kochovskaya M. D., The Role of Some Biochemical Markers in the Women with External Genital Endometriosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 716-718. doi: 10.5923/j.ajmms.20261602.68.
1. Background
- Endometriosis is a chronic, inflammatory, estrogen-dependent gynecological disease characterized by the growth of endometrial cells outside the uterine cavity [1]. Endometrial cells migrate from their original site, the uterus, to other organs and form endometrial-like tissues in various anatomical locations outside the uterine cavity, particularly in the ovaries and peritoneum [1,2]. Although the symptoms of endometriosis are not unique and many of them are similar to those of other gynecological diseases, it can cause pelvic discomfort and infertility [3]. According to the literature, the disease is diagnosed with a significant delay of 3 to 11 years, which leads to disruption of the reproductive cycle in women of reproductive age. The true prevalence of endometriosis cannot be determined due to the lack of effective non-invasive diagnostic methods; however, epidemiological data indicate that approximately 10% of women of childbearing age suffer from endometriosis. Furthermore, its incidence increases from 20% to 50% in women experiencing pelvic discomfort or infertility [3,4,5]. Endometriosis is a disease with a complex etiopathogenesis, involving the participation of genetic and immunological pathways (including inflammation), as well as environmental influences such as dietary habits and nutrients or antibacterial therapy, which, according to recent studies, may be one of the factors altering the quantity and diversity of microflora. Studies show that the development of endometriosis involves changes in many biological pathways, mainly in metabolic regulation underlying estrogen-dependent disorders [6,7].The exact etiology of endometriosis remains unclear; however, the most acceptable hypothesis is retrograde menstruation of viable endometrial tissue infiltrating the abdominal cavity, which subsequently leads to the invasion of pathological tissue, thereby forming diffuse pelvic endometriosis. [3,6,7]. This process subsequently triggers the development of chronic inflammatory reactions, generating proinflammatory activity. However, pathological reflux of menstrual blood may not explain all cases of endometriosis. Differences in the biochemical and pathological properties of endometrial tissue found in the ovaries, rectovaginal septum, or peritoneum indicate that endometriosis may result from various diseases [12].Recently, inflammation has been recognized as an important factor in the pathogenesis of endometriosis. Ectopic endometriotic tissue secretes cytokines and chemokines that promote inflammation and attract macrophages into the peritoneal cavity, further stimulating the inflammatory response [13]. Increased numbers of activated macrophages are detected in the peritoneal cavity of patients with endometriosis [10]. It has also been found that expression levels of monocyte chemotactic protein 1 (MCP-1), interleukin (IL)-8, IL-6, and tumor necrosis factor (TNF)-α are elevated in the peritoneal cavity of patients with endometriosis [13].Today, gynecological scientists from around the world are searching for modern, reliable markers that could solve the key problem of disease prevention.The aim of this study was to determine the role of the biochemical substrate Zn-alpha2-glycoprotein as a marker for the development of external genital endometriosis in women.
2. Study Materials and Methods
- We studied 40 women admitted to the outpatient department of the Tashkent City Interdistrict Perinatal Center. They constituted the study group with a preliminary clinical diagnosis of external genital endometriosis of grades I-II. The control group consisted of 45 apparently healthy women of reproductive age. The women studied were of similar age, ranging from 22 to 36 years. The preliminary diagnosis (external genital endometriosis) was based on clinical history, laboratory, and instrumental examination. The study was conducted at the Department of Obstetrics and Gynecology with a course in pediatric gynecology at Tashkent State Medical University.A biochemical profile of the patients was analyzed after their written consent. Zinc-alpha2-glycoprotein was determined in all women studied using the ELISA method. A reference value of 30 mg/L was adopted. Zinc-alpha2-glycoprotein (AZGP1) is a specific secreted active protein encoded by the AZGP1 gene, which is synthesized directly by epithelial cells of the body, as well as by adipocytes, and is involved in lipid metabolism, the cell cycle, and tumor progression, including those of carcinogenic origin [14].
3. Results
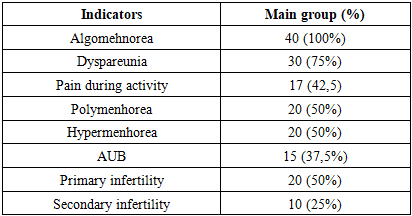
- All women in the study group complained of pelvic pain (100%) and menstrual irregularities (polymenorrhea). When assessing pelvic pain, women noted pain primarily during menstruation and intercourse. Less frequently, pain was observed during stress and physical exercise (Table 1). According to the pelvic pain classification (SRI 2022), all women in the study group experienced pain stage 3 or 4 (with a maximum of 5-6).
|
|
4. Conclusions
- Our study demonstrates the importance of Zn-alpha2-glycoprotein testing in the early diagnosis of external genital endometriosis in women. The factor of complex treatment, both surgical and conservative, leads to a significant decrease in the Zn-alpha2-glycoprotein level.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML