-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 702-706
doi:10.5923/j.ajmms.20261602.65
Received: Jan. 3, 2026; Accepted: Jan. 28, 2026; Published: Feb. 25, 2026

Preoperative Radiation Therapy in Combined Treatment of Locally Advanced Rectal Cancer
Nishanov D. A., Chen E. E., Mansurova G. B., Saidova K. A., Izrailbekova K. Sh.
Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Relevance: Rectal cancer is often diagnosed at locally advanced stages, where choosing the optimal preoperative strategy is crucial. Objective: To evaluate the effectiveness of preoperative radiation therapy as part of combined treatment. Materials and Methods: Analysis of 62 patients (RSNPMC and Radiation Oncology Center, 2019–2024): Group A – FOLFOX ± chemoradiotherapy (CRT), Group B – standard CRT 50–50.4 Gy. Results: Radical resections were achieved in over 96% of patients in both groups; pathological response was 20.0% (A) and 21.9% (B). Toxicity grade ≥III: 10.0% (A) and 18.8% (B). Three-year overall survival: 78.3% vs 75.0%; OS: 90.0% vs 87.5%. Conclusion: Selective strategy using FOLFOX ± CRT is comparable to standard CRT in oncological outcomes with lower toxicity and radiation burden.
Keywords: Rectal cancer, Neoadjuvant therapy, Chemoradiotherapy
Cite this paper: Nishanov D. A., Chen E. E., Mansurova G. B., Saidova K. A., Izrailbekova K. Sh., Preoperative Radiation Therapy in Combined Treatment of Locally Advanced Rectal Cancer, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 702-706. doi: 10.5923/j.ajmms.20261602.65.
1. Relevance
- Rectal cancer, which is part of the group of colorectal malignancies, remains one of the leading causes of cancer incidence and mortality worldwide. According to GLOBOCAN 2020 estimates, rectal tumors account for a significant proportion of colorectal cancer cases and related deaths, highlighting the clinical importance of optimizing locoregional therapy in patients with locally advanced disease [6].Figure 1 shows the age-standardized incidence and mortality rates for the top 10 cancer sites worldwide (2022). For the combined category “colorectum,” the incidence rate was 18.4 per 100,000 and the mortality rate was 8.1 per 100,000 — a mortality-to-incidence ratio of approximately 0.44, reflecting moderate lethality compared to, for example, lung cancer, which has high mortality despite lower incidence.
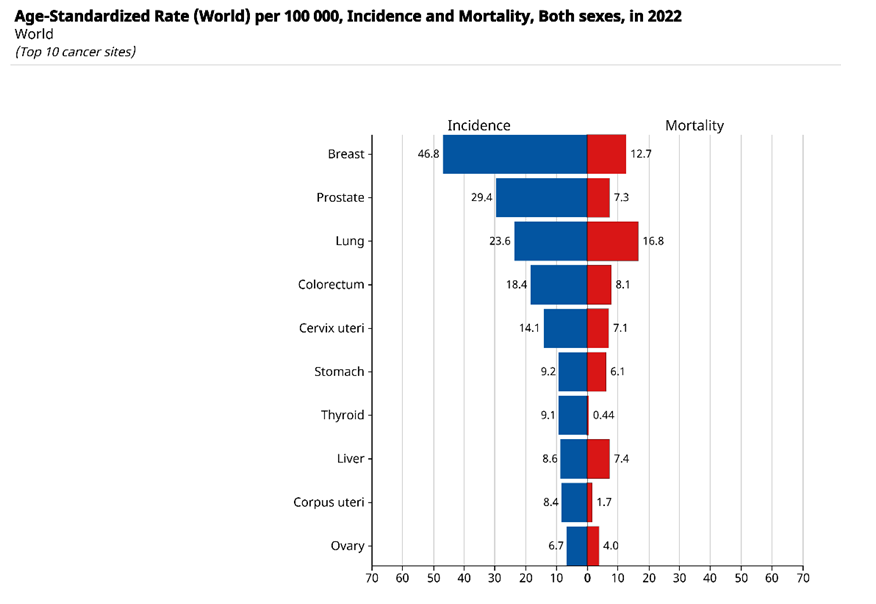 | Figure 1. Age-standardized incidence and mortality rates (per 100,000 population, both sexes), 2022 – Top 10 cancer sites (GLOBOCAN 2022) |
 | Figure 2. Age-standardized incidence rate of rectal cancer (ASR, per 100,000 population, both sexes), map of prevalence by countries in the region (GLOBOCAN 2022) |
2. Materials and Methods
- This study was conducted as a single-center retrospective cohort analysis at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSNPMC OR) in the departments of radiotherapy, coloproctology, and chemotherapy. The analysis included 62 consecutive patients treated between 2019 and 2024.Patients with morphologically confirmed adenocarcinoma of the rectum, stages cT2N1 or cT3N0–1 based on pelvic MRI, without signs of tumor fixation to the external sphincter or levator muscles, were included. All patients were candidates for sphincter-preserving surgery, had satisfactory performance status (ECOG 0–2), and underwent a standard examination protocol including colonoscopy with biopsy, computed tomography of the chest and abdomen, and pelvic magnetic resonance imaging.Patients were divided into two groups depending on the preoperative treatment approach.Group A (FOLFOX ± CRT): Patients received six cycles of neoadjuvant chemotherapy according to the FOLFOX regimen. If an objective tumor regression of ≥20% was observed on MRI and clinical evaluation, the chosen strategy was continued without radiation; in cases of insufficient response (<20%) or chemotherapy intolerance, preoperative chemoradiotherapy was performed at a dose of 50–50.4 Gy to the pelvic area in 25–28 fractions with concurrent fluoropyrimidine as a radiosensitizer.Group B (Standard CRT): Patients received a course of preoperative chemoradiotherapy at a dose of 50–50.4 Gy in 25–28 fractions to the pelvic area combined with fluoropyrimidine. A dose boost using modern techniques (IMRT/VMAT) was applied if indicated by the radiation oncologist. In some cases, a short-course radiation (5×5 Gy) followed by surgery was allowed.After completion of neoadjuvant treatment, all patients underwent total mesorectal excision. The type of surgical access was determined by the surgeon based on the clinical situation.Assessment of Response and Postoperative TherapyTreatment efficacy was evaluated based on clinical and radiological examinations (pelvic MRI), as well as morphological analysis of the surgical specimen. The primary indicators included pathological complete response, resection radicality (R0/R1), tumor downstaging, and the frequency of sphincter-preserving surgeries. In cases of residual tumor or metastatic lymph node involvement, the decision to prescribe adjuvant chemotherapy was made at multidisciplinary tumor board meetings.Descriptive statistics were presented as medians with interquartile ranges and relative values (n, %). Group comparisons were performed using Pearson’s χ² test or Fisher’s exact test, as well as the Mann–Whitney U test or t-test depending on data distribution. Survival was calculated using the Kaplan–Meier method with the log-rank test for intergroup comparisons. Multivariate analysis using Cox regression was performed to identify independent prognostic factors. Differences were considered statistically significant at p<0.05, and regression results were presented as hazard ratios with 95% confidence intervals.
3. Results
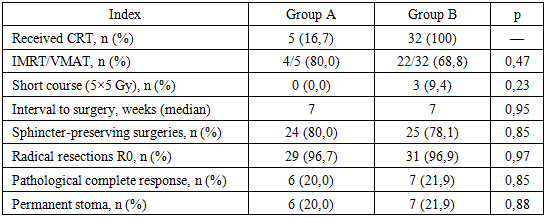
- The study included 62 patients with locally advanced rectal cancer, divided into two groups: Group A consisted of 30 patients who received neoadjuvant chemotherapy according to the FOLFOX regimen with selective chemoradiotherapy in cases of insufficient response; Group B included 32 patients who underwent standard preoperative chemoradiotherapy. The median age was 58 years (57 years in Group A and 59 years in Group B), with no significant differences between cohorts (p=0.41) (Table 1). Males comprised the majority (61.3%), with comparable distribution in both groups (63.3% vs. 59.4%; p=0.77). Clinically, tumors of category cT3N0 (41.9%) and cT3N1 (38.7%) predominated; patients with stage cT2N1 accounted for 19.4%, with no statistically significant differences between groups (p>0.7). Functional status according to the ECOG scale was 0–1 in 88.7% of patients (90.0% in Group A and 87.5% in Group B; p=0.76), indicating high baseline therapy tolerance. The mean distance from the lower tumor pole to the dentate line was 6.2±1.8 cm (6.1±1.9 cm and 6.3±1.7 cm, respectively; p=0.64), reflecting predominantly mid- and low-ampullary tumor localization. Thus, the study groups were homogeneous in terms of age and sex characteristics, clinical stage, functional status, and anatomical tumor features, ensuring the validity of the subsequent comparative analysis of radiotherapy and surgical treatment outcomes.
|
|
4. Discussion
- The results of this single-center study, which included 62 patients with locally advanced rectal cancer, indicate comparable efficacy between the selective strategy of radiation therapy following FOLFOX chemotherapy and the standard preoperative chemoradiotherapy approach. These findings align with international experience, particularly the PROSPECT trial [5], which demonstrated that neoadjuvant FOLFOX chemotherapy with subsequent radiation in cases of insufficient response is not inferior to standard preoperative chemoradiotherapy regarding relapse-free survival and overall survival.Selective use of preoperative radiation reduced the frequency of chemoradiotherapy application in Group A to 16.7%, while maintaining a high rate of radical resections (96.7%) and pathological complete response (20.0%), comparable to the standard chemoradiotherapy group (96.9% and 21.9%, respectively). This confirms the feasibility of a personalized approach in which radiation is administered only to patients with an inadequate response to systemic therapy.From a practical standpoint, it is important to note that the standard chemoradiotherapy group exhibited a higher frequency of severe acute toxic reactions (≥grade III) (18.8% vs. 10.0% in Group A) and postoperative complications (≥grade II) (21.9% vs. 16.7%). Although these differences were not statistically significant, the data are consistent with meta-analyses showing that pelvic radiation is associated with an increased risk of late radiation complications and impaired quality of life [1,8].A significant finding was the high rate of sphincter-preserving surgeries (79–80%) with no differences between groups, confirming the oncological safety of both strategies. This is particularly important for patients with tumors located in the mid- and low-rectal ampulla, where preservation of anal sphincter function is a key aspect of quality of life [3].Kaplan–Meier curves for DFS and OS demonstrated similar outcomes between groups: three-year DFS was 78.3% in the FOLFOX±CRT group and 75.0% in the standard CRT group (p=0.87), and three-year OS was 90.0% and 87.5%, respectively (p=0.74). These data confirm the absence of compromise in oncological radicality with reduced radiation exposure, consistent with current principles of treatment de-escalation.
5. Conclusions
- This study showed that a selective preoperative treatment strategy for locally advanced rectal cancer, using FOLFOX chemotherapy with chemoradiotherapy reserved for patients with insufficient response, achieves comparable results to traditional preoperative chemoradiotherapy in terms of radical resection rates, pathological complete response rates, relapse-free survival, and overall survival. At the same time, it reduces the frequency of severe toxic reactions and radiation burden on patients, aligning with contemporary trends of treatment de-escalation and therapy individualization. The findings support the safe limitation of radiation therapy volume in certain patient categories without compromising oncological efficacy.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML