-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 695-701
doi:10.5923/j.ajmms.20261602.64
Received: Dec. 28, 2025; Accepted: Jan. 22, 2026; Published: Feb. 25, 2026

Immunohistochemical Markers P53, Ki-67, and VEGF as Predictors of Response to Neoadjuvant Chemoradiotherapy in Locally Advanced Rectal Cancer
Nishanov D. A., Chen E. E., Mansurova G. B., Saidova K. A., Izrailbekova K. Sh.
Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

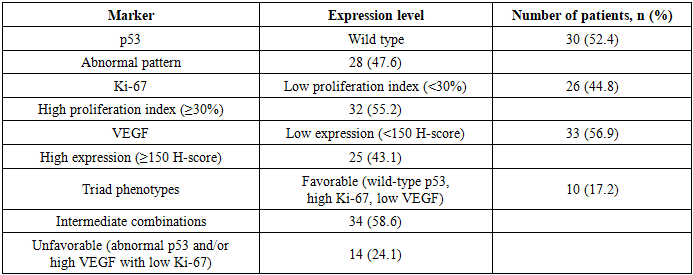
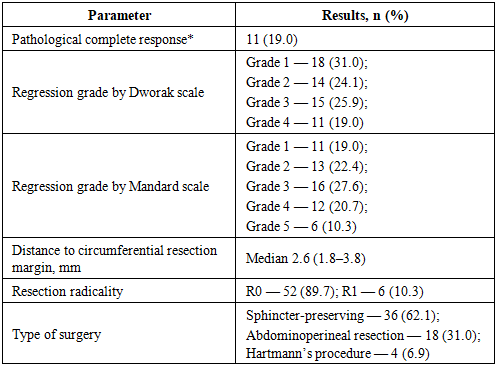
Relevance. Rectal cancer is often diagnosed at locally advanced stages, where pretreatment risk stratification is crucial. Objective. To evaluate the prognostic significance of p53, Ki-67, and VEGF expression in patients with locally advanced rectal cancer who received neoadjuvant combined therapy. Materials and Methods. A total of 58 patients were analyzed (2019–2024, Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology). Immunohistochemistry of biopsy samples was performed for p53, Ki-67, and VEGF; findings were compared with MRI staging and clinico-morphological parameters. Results. Complete response was observed in 19% of cases. High Ki-67 expression was associated with better tumor regression and survival, whereas abnormal p53 and high VEGF levels correlated with poor prognosis. Conclusion. The combined p53/Ki-67/VEGF panel together with MRI stagingincreases prognostic accuracy and can be applied for therapy personalization.
Keywords: Rectal cancer, Neoadjuvant therapy, p53, Ki-67, VEGF
Cite this paper: Nishanov D. A., Chen E. E., Mansurova G. B., Saidova K. A., Izrailbekova K. Sh., Immunohistochemical Markers P53, Ki-67, and VEGF as Predictors of Response to Neoadjuvant Chemoradiotherapy in Locally Advanced Rectal Cancer, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 695-701. doi: 10.5923/j.ajmms.20261602.64.
1. Relevance
- Rectal cancer is one of the most pressing issues in modern oncology due to its high incidence, significant mortality, and frequent diagnosis at locally advanced stages. According to Sung et al. (2021), colorectal cancer ranks third in global cancer incidence and second in cancer-related mortality, accounting for approximately 10% of all cancer cases and deaths [7, CA Cancer J Clin, 2021].In Asian countries, including the Central Asian region, there is a persistent upward trend in incidence. Wiley (2022) notes that more than 50% of new colorectal cancer cases and nearly half of cancer-related deaths are registered in Asia, with the disease increasingly being diagnosed among people of working age [8].Analysis of the presented map indicates significant geographical differences in the incidence of rectal cancer (Fig. 1). The highest rates (9.5–17.2 per 100,000 population) are observed in Europe, North America, Australia, and Russia. Intermediate levels (5.4–9.5 per 100,000) are typical for several countries of South America and East Asia. The lowest rates (<2.1 per 100,000) are predominantly registered in African countries and some regions of South Asia. In Central Asia, including Uzbekistan, incidence rates fall within the moderate range (2.1–3.6 per 100,000), which underlines the growing medical and social significance of this pathology in the region.
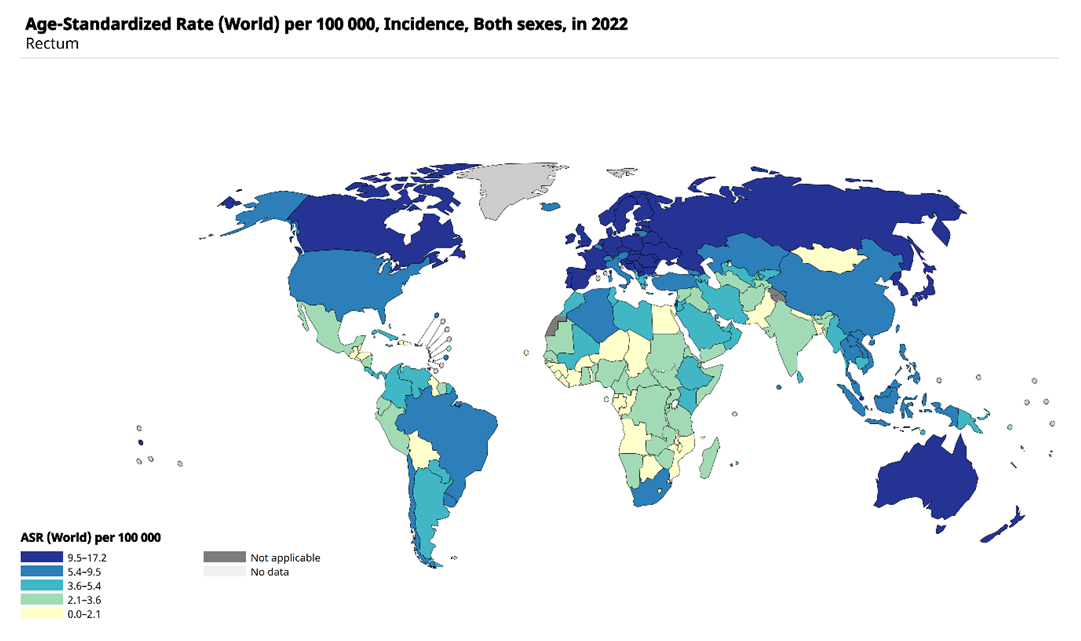 | Figure 1. Global incidence of rectal cancer (age-standardized rates per 100,000 population, both sexes, 2022, according to GLOBOCAN/IARC) |
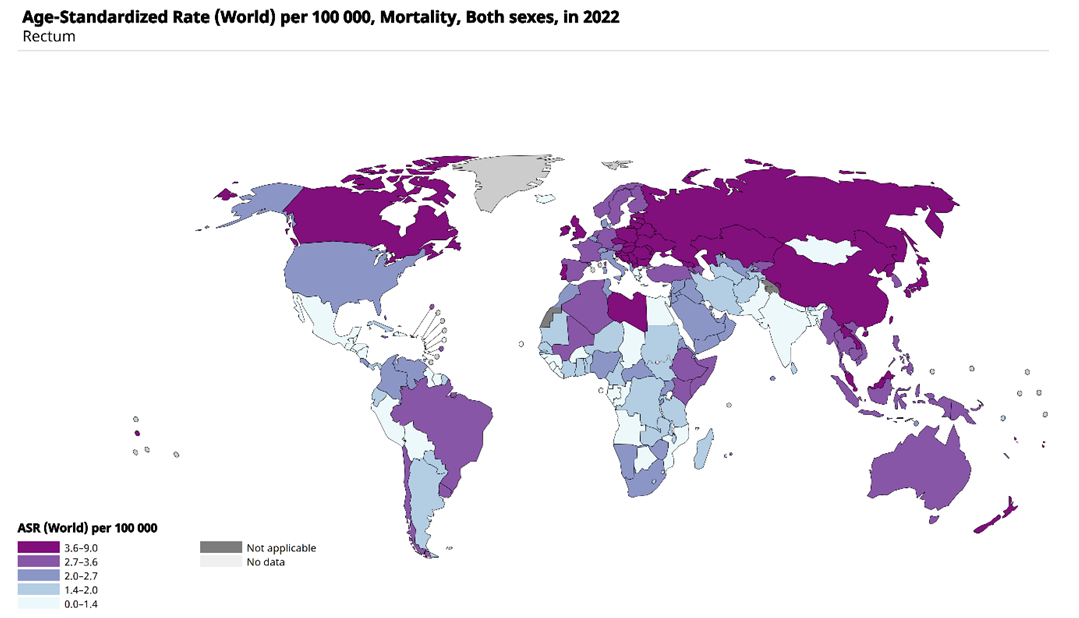 | Figure 2. Global mortality from rectal cancer (age-standardized rates per 100,000 population, both sexes, 2022, according to GLOBOCAN/IARC) |
2. Materials and Methods
- This retrospective study was based on the analysis of clinical, morphological, radiological, and therapeutic data of 58 patients with histologically verified adenocarcinoma of locally advanced rectal cancer, who underwent treatment at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR) between January 2019 and June 2024.The inclusion criteria for the study were: patient age 18 years and older at the time of diagnosis; presence of histologically confirmed rectal adenocarcinoma located no higher than 12 cm from the anal verge; disease stage T3–T4 and/or N+ according to pretreatment pelvic magnetic resonance imaging of the pelvis; somatic status of 0–2 points on the ECOG scale; completion of a course of neoadjuvant chemoradiotherapy followed by radical surgery in the form of total mesorectal excision. The exclusion criteria included: presence of distant metastases (M1) at the time of diagnosis; previous radiotherapy to the pelvic region; synchronous or previously treated malignant tumors; absence of complete medical documentation or loss of paraffin blocks for immunohistochemical examination; as well as refusal of treatment or deviation from standard protocols. Within the framework of the study, demographic characteristics (age, sex), clinical parameters (somatic status, tumor location), pretreatment pelvic magnetic resonance imaging data with detailed staging according to T, N, presence of vascular invasion, and distance to the mesorectal fascia were analyzed. Morphological analysis included the histological variant of the tumor, degree of differentiation, and pathomorphological response to therapy. Immunohistochemical analysis was performed on pretreatment biopsies to assess the expression of p53, Ki-67, and VEGF. Therapeutic data included chemoradiotherapy regimens (total dose 50.4 Gy/28 fractions, IMRT/VMAT technique with concurrent capecitabine 825–900 mg/m²), interval to surgical intervention, and surgical volume. Treatment outcomes were assessed by the rate of achieving pathological complete response (absence of invasive carcinoma and lymph node metastases), degree of tumor regression, frequency of local recurrences, and recurrence-free survival indicators. For systematization of information, a unified coding table was used, which enabled the creation of cross-tabulation tables and multivariate analysis. Statistical processing of the data was carried out using the R software package. Descriptive statistical methods with calculation of absolute and relative indicators were applied. To analyze the relationship of categorical variables, Pearson’s χ² test and Fisher’s exact test were used. To evaluate factors associated with the probability of achieving pathological complete response, logistic regression was applied with calculation of odds ratios and 95% confidence intervals. Statistical significance was established at p < 0.05.
3. Results
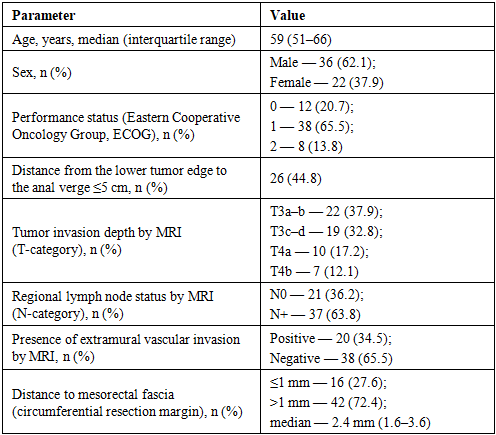
- A total of 58 patients with locally advanced rectal cancer were included in the study. Analysis of baseline clinical and magnetic resonance characteristics showed that the median age of the patients was 59 years (interquartile range 51–66 years), with a predominance of males (62.1%). In almost half of the patients, the tumor was located in the lower ampullary section of the rectum (≤5 cm from the anal verge — 44.8%). According to pre-treatment pelvic MRI, stage T3 was detected in 70.7% of patients, and stage T4 in 29.3%. Regional lymph node involvement was noted in 63.8% of cases. Signs of extra-organ vascular invasion were observed in 34.5% of patients. A threatened circumferential resection margin (≤1 mm) was recorded in 27.6% of patients, with the median distance to the mesorectal fascia being 2.4 mm (interquartile range 1.6–3.6 mm).
|
|
|
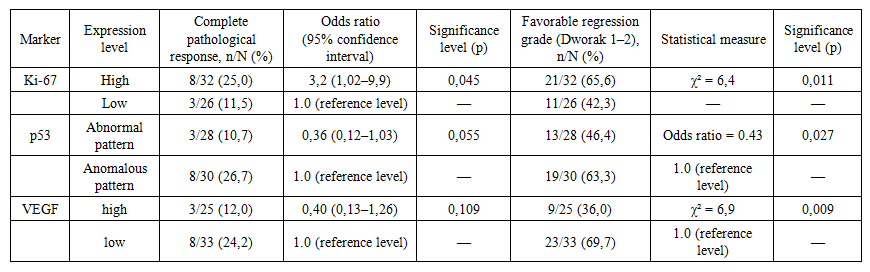 | Table 4. Associations of p53, Ki-67, and VEGF expression with pathological complete response and degree of tumor regression (n=58) |
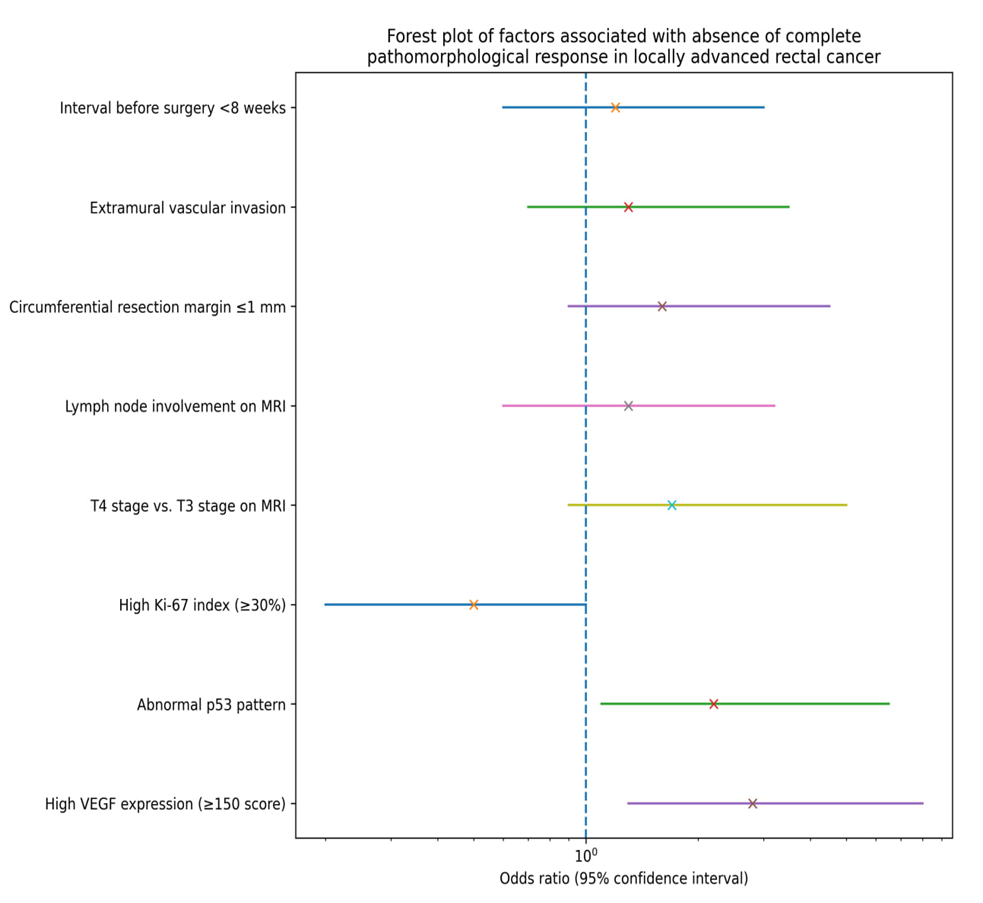 | Figure 3. Factors associated with the absence of a complete pathological response in patients with locally advanced rectal cancer |
4. Discussion
- Our study demonstrated that the immunohistochemical markers p53, Ki-67, and VEGF possess prognostic value in locally advanced rectal cancer. We found that a high Ki-67 proliferation index was associated with a higher likelihood of achieving a complete pathological response and with improved disease-free survival rates. In contrast, an abnormal p53 pattern and high VEGF expression emerged as independent unfavorable factors, linked to lower tumor regression rates and an increased risk of recurrence.Similar findings have been reported in the international literature. Jakob et al. (2008) showed that elevated Ki-67 correlates with greater tumor sensitivity to neoadjuvant chemoradiotherapy. Chen et al. (2011) noted that abnormal p53 predicts poorer treatment response and reduced survival [1,5]. The role of VEGF as a factor of radioresistance and as a prognostic biomarker has been confirmed by Zlobec et al. (2008) and Yu et al. (2019). More recent studies emphasize the value of combined panels incorporating multiple markers, which significantly improve prognostic accuracy [2,10,11].Our observations are also consistent with epidemiological data from GLOBOCAN (Sung et al., 2021), which report colorectal cancer as the third most common and the second most deadly malignancy worldwide. In Asian countries, including Central Asia, a steady increase in incidence rates has been observed (Wiley, 2022), underlining the importance of risk stratification for treatment optimization [4,7,8].It should be noted that MRI-based staging (T, N), as recommended by the ESMO (2024) and NCCN (2025) guidelines, in our cohort showed only a trend toward unfavorable impact without reaching statistical significance [3,6]. Nevertheless, in clinical practice, these parameters remain crucial for surgical planning.Our findings support that the use of an IHC panel (p53, Ki-67, VEGF) in combination with radiological characteristics may improve prognostic assessment and facilitate the individualization of treatment strategies in patients with locally advanced rectal cancer.
5. Conclusions
- In a single-center cohort of 58 patients with locally advanced rectal cancer, a high pre-treatment Ki-67 proliferation index was associated with a higher frequency of complete pathological response and improved disease-free survival. In contrast, an abnormal p53 pattern and high VEGF expression predicted incomplete regression, reduced oncological radicality, and an increased risk of unfavorable outcomes. In multivariate models, VEGF and p53 retained their independent adverse prognostic roles, whereas Ki-67 demonstrated a protective effect. The addition of pre-treatment magnetic resonance imaging characteristics (depth of invasion, regional lymph nodes, extramural vascular invasion, and distance to the mesorectal fascia) enhanced the discriminatory power of the combined model. Joint pre-treatment assessment of p53, Ki-67, and VEGF together with standardized MRI staging may be recommended for personalized risk stratification and for guiding the intensity of neoadjuvant and surgical treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML