-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 689-691
doi:10.5923/j.ajmms.20261602.62
Received: Dec. 22, 2025; Accepted: Jan. 19, 2026; Published: Feb. 25, 2026

Clinical and Microbiological Characteristics of Post-Chemotherapy Oral Mucositis and Evidence-Based Prevention Strategies
Elova Hanifa Rakhmatovna, Jabborova Feruza Uzakovna
Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
Correspondence to: Elova Hanifa Rakhmatovna, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Oral mucositis (OM) is a common and debilitating complication of chemotherapy, affecting up to 80% of patients and significantly impairing oral function and quality of life. Chemotherapy disrupts epithelial turnover, salivary defenses, and microbial balance, resulting in ulceration and inflammation. This review analyzes clinical features, microbiological associations, and prevention strategies using current evidence. Methods include synthesis of published studies, comparative clinical analysis, and microbiological data evaluation. Results confirm strong associations between OM severity, microbial dysbiosis, Candida overgrowth, and salivary dysfunction. Three evidence-based prevention models were analyzed. Comprehensive oral hygiene measures, probiotics, antifungal prophylaxis, and photobiomodulation reduce OM incidence by 40–63%. Conclusions emphasize the importance of early detection, microbial screening, and multimodal prevention strategies to decrease OM severity and improve patient outcomes.
Keywords: Oral mucositis, Chemotherapy, Microbiome imbalance, Candida, Epithelial injury, Prevention
Cite this paper: Elova Hanifa Rakhmatovna, Jabborova Feruza Uzakovna, Clinical and Microbiological Characteristics of Post-Chemotherapy Oral Mucositis and Evidence-Based Prevention Strategies, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 689-691. doi: 10.5923/j.ajmms.20261602.62.
Article Outline
1. Introduction
- Oral mucositis (OM) is one of the most clinically significant toxicities of chemotherapy. It develops in 40–80% of patients depending on treatment regimen, immune status, and oral conditions [1]. Pathophysiologically, OM results from chemotherapy-induced epithelial injury, immune suppression, reactive oxygen species activation, and microbial dysbiosis [2]. Initially, erythema and soreness appear, followed by ulcerations, pseudomembranes, and severe pain. These lesions limit food intake, speech, and hygiene practices and may predispose to systemic infection. Severe OM often forces clinicians to modify or interrupt chemotherapy schedules [3]. Recent research highlights the crucial role of oral microbiota in exacerbating mucosal damage. Pathogenic bacteria and fungal species proliferate when mucosal barriers are weakened, further intensifying inflammation [4].The aim of this review is to present an expanded clinical and microbiological analysis of OM and provide evidence-based preventive strategies.
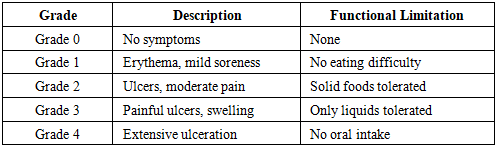
2. Clinical Characteristics of Oral Mucositis
- OM progresses through well-defined stages—from erythema to ulceration and healing—usually 5–14 days after chemotherapy initiation [1].
|
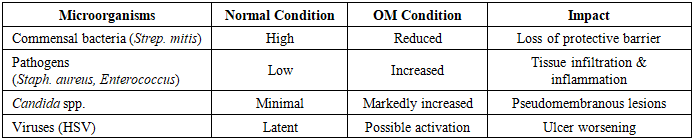
3. Microbiological Factors and Dysbiosis
- The oral cavity hosts >700 microbial species. Healthy balance is maintained by saliva, epithelial turnover, and immune mechanisms. Chemotherapy disrupts this equilibrium, resulting in dysbiosis [5].
|
4. Pathophysiology
- OM develops through a 5-step biological cascade:1. Initiation: Chemotherapy induces DNA damage, ROS formation.2. Up-regulation: Inflammatory transcription factors activate NF-κB [6].3. Amplification: Cytokines (IL-1β, TNF-α) increase tissue injury.4. Ulceration: Epithelial breakdown, microbial invasion.5. Healing: Re-epithelialization and microbial normalization occur.Salivary dysfunction—reduced flow and antimicrobial proteins—contributes significantly, allowing pathogens to proliferate [7].
5. Evidence-Based Prevention Strategies
- OM prevention is most effective when applied BEFORE and DURING chemotherapy. Recommended modalities include:
|
5.1. Probiotics
- Probiotics maintain microbial balance and inhibit pathogenic overgrowth. Clinical trials demonstrate reduced OM severity in patients receiving Lactobacillus rhamnosus–based therapy [6].
5.2. Photobiomodulation Therapy
- PBM (low-level laser therapy) accelerates epithelial healing and reduces inflammation. It is recommended by MASCC/ISOO guidelines as a standard intervention [7].
5.3. Antimicrobial Regimen
- Targeted antifungal and antibacterial therapy decreases pathogen load and supports mucosal regeneration [10].
6. Discussion
- This review confirms that OM is not merely epithelial damage but a complex disorder involving microbiological, immunological, and salivary dysfunction components. Microbial dysbiosis significantly worsens clinical severity, especially when Candida proliferation and pathogenic bacterial colonization occur [8,9].Evidence strongly supports the use of combined preventive strategies, including oral hygiene, probiotics, antimicrobials, and PBM therapy [5,6,7]. A multimodal prevention plan results in improved healing time, reduced symptom severity, and fewer chemotherapy interruptions.
7. Conclusions
- 1. OM severity is strongly associated with microbial imbalance.2. Early microbial screening and oral hygiene optimization are essential.3. Preventive interventions (PBM, probiotics, antifungals) significantly reduce OM severity.4. Multimodal prevention strategies enhance patient comfort and help maintain chemotherapy schedules.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML