Babaeva A. I., Alieva D. A.
Republican Specialized Scientific and Practical Medical Center of Maternal and Child Health, Tashkent, Uzbekistan
Correspondence to: Babaeva A. I., Republican Specialized Scientific and Practical Medical Center of Maternal and Child Health, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
To evaluate the effect of restoring vaginal microflora during the preparation period for destruction in patients with mild cervical displasia and disturbed vaginal microbiocenosis using complex therapy with the drug Bioterol. The effectiveness of complex therapy with Bioterol was assessed in 60 patients with cervical displasia and confirmed HPV infection. The obtained results allow recommending this diagnostic and therapeutic approach during the preparation for destruction in patients with cervical displasia.
Keywords:
Cervical displasia, HPV, Vaginal microbiocenosis, Bioterol
Cite this paper: Babaeva A. I., Alieva D. A., Correction of Vaginal Microbiocenosis in Women with Cervical Dysplasia, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 684-688. doi: 10.5923/j.ajmms.20261602.61.
1. Introduction
Cervical dysplasia is one of the most common pathologies encountered in ambulatory gynecology [1,2]. Despite advancements in the management of HPV-associated precancerous cervical lesions, an increase in oncological morbidity in this field continues to be observed [1,3].At the same time, in 26–50% of patients who undergo destructive treatment of pathologically altered epithelium, precancerous processes recur and symptoms worsen [1,3,4].Treatment difficulties are associated with immune dysfunction induced by the E7 oncoprotein, which is expressed when high-risk HPV types transition to the integrative phase. This leads to dysbiotic processes in the genital tract and creates a favorable environment for the manifestation of human papillomavirus infection [3,5].Since conditionally pathogenic bacteria can reach high concentrations in the vagina and cervix, potentially initiating and supporting dysplastic processes [2,6,7], it is necessary to develop comprehensive measures during the pre-destruction preparation stage. These measures should include etiological and pathogenetically based therapy aimed at restoring vaginal acidity.Currently used systemic antibacterial therapy methods may induce an immunodeficiency-like syndrome, manifested by impaired humoral and cellular immunity, decreased synthesis of complement components, reduced activity of cytotoxic lymphocytes and macrophages [4]. This destabilizes the vaginal ecosystem, sharply reduces the quantity and quality of normal synergistic microflora, increases vaginal pH, and creates favorable conditions for the growth of conditionally pathogenic flora [8,9].Local application of antibacterial agents also often fails to adequately restore the microbiocenosis, or may not restore it at all [10]. Against this background, destructive procedures lead to prolonged healing time, recurrence of the process, and increased risk of reinfection with other pathogens.Insufficient correction of vaginal microbiocenosis, the occurrence of infectious complications in 21–37% of cases after destruction [3,4], as well as recurrence of dysplastic processes [5], determine the need to develop complex methods of pre-destruction microbiocenosis correction. These methods should include the combined use of antiseptic preparations and restoration of vaginal acidity.For this study, we proposed the use of the probiotic-effect preparation Bioterol capsules to normalize vaginal microflora and reduce infectious and inflammatory factors during the treatment of cervical dysplasia.
2. Study Objective
To evaluate the effectiveness of restoring vaginal microflora in patients with cervical dysplasia after treatment with Bioterol compared to standard therapy.
3. Materials and Methods
To achieve the stated objective, the study was conducted at the Republican Specialized Scientific-Practical Medical Center for Mother and Child Health and the “Family and Marriage” Scientific-Consultative Polyclinic during 2024–2025.The main prospective group consisted of 120 patients who presented with mild dysplasia of the multilayer squamous epithelium of the cervix. These patients were divided into two groups:• Group 1: 60 (50.0%) CIN I patients with HPV PCR-positive results• Group 2: 60 (50.0%) CIN I patients with HPV PCR-negative resultsA control group included 96 practically healthy patients with normal colposcopic findings and no evidence of cervical dysplasia.• All women underwent the following clinical and laboratory examinations:• collection of complaints gynecological examination• Femoflor testing• triple-site vaginal smears• bacteriological culture from the vaginaVaginal biocenosis was assessed using PCR-RT (“real-time” detection), Femoflor testing, triple-site smear, and bacteriological culture prior to treatment and one month after treatment.
4. Results and Discussion
Considering that the study targeted women aged 21 to 49 years according to regulatory documents of the Republic of Uzbekistan, all examined patients were conditionally divided into three age groups (21–29, 30–39, and 40–49 years). The mean age of women included in the analysis was 28.3 ± 6.35 years (Figure 1).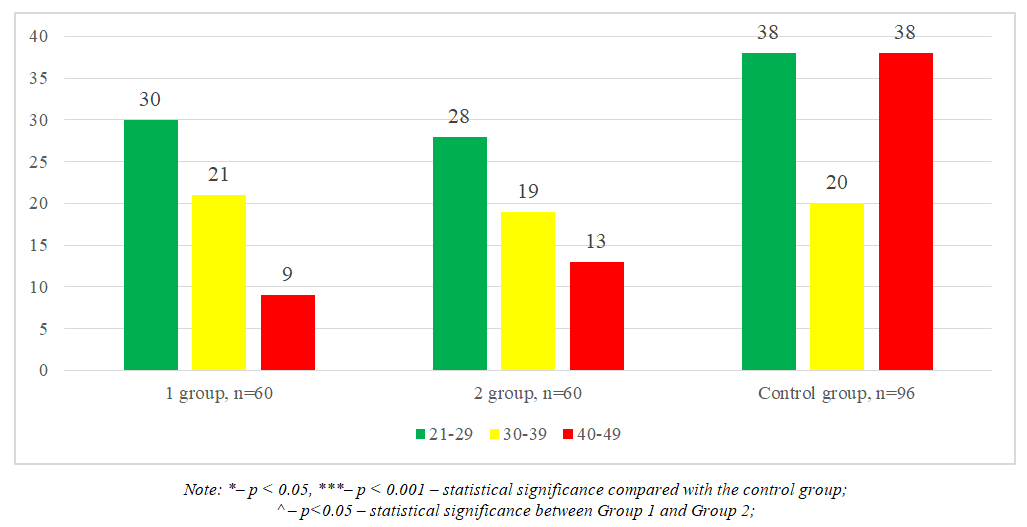 | Figure 1. Age characteristics of the examined women |
The distribution of age categories among the examined Group 1 (CIN I + HPV(+)), Group 2 (CIN I + HPV(-)), and the control group showed the following:• The proportion of women aged 21–29 years was similar between Groups 1 and 2: 30 (50.0 ± 6.5%) in Group 1 and 28 (46.7 ± 6.5%) in Group 2. Both groups were significantly higher compared to the control group, which included 38 (39.6 ± 5.0%) women, p < 0.05.• In the 30–39 years age category, there was no significant difference between the groups (p > 0.05). The distribution was 21 (35.0 ± 6.2%) in Group 1, 19 (31.7 ± 6.1%) in Group 2, and 20 (20.8 ± 4.2%) in the control group.• In the 40–49 years age category, a significant difference was observed. Group 1 included 9 (15.0 ± 4.6%) women, Group 2 had 13 (21.7 ± 5.4%) women, and the control group had 38 (39.6 ± 5.0%) women, which was more than twice higher compared to Group 2, p < 0.001.Analysis of Vaginal MicrobiocenosisTo normalize vaginal microflora and reduce infectious and inflammatory factors during the treatment of dysplasia, the probiotic preparation Bioterol capsules was proposed.Accordingly, the main group of 60 patients was divided into two subgroups based on the efficacy of Bioterol after administration:• Group 1: 52 (86.7%) patients with high efficacy• Group 2: 8 (13.3%) patients with low efficacyFollow-up examinations of the main group and the two subgroups were conducted one month after treatment. These included:• Femoflor analysis of urogenital tract biocenosis• Triple-site vaginal smear• Bacteriological culture from the vaginaThe results of the post-treatment urogenital tract biocenosis analysis by Femoflor are presented in Table 1.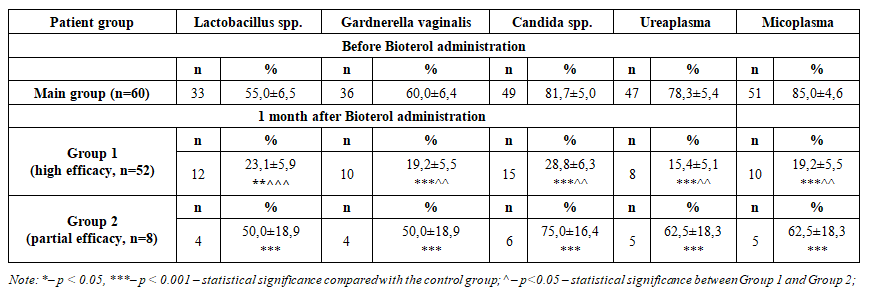 | Table 1. Comparative description of urogenital tract biocenosis before and after Bioterol administration using Femoflor analysis (M±m), % |
In the main group (n=60) before Bioterol administration, a high frequency of pathogenic flora characteristic of dysbiosis was detected in the vaginal microbiocenosis: Gardnerella vaginalis – 36 (60.0 ± 6.4%), Candida spp. – 49 (81.7 ± 5.0%), Ureaplasma – 47 (78.3 ± 5.4%), Mycoplasma – 51 (85.0 ± 4.6%). At the same time, the level of Lactobacillus spp., representing normal and protective flora, was low – 33 (55.0 ± 6.5%).One month after administration of Bioterol, in Group 1 (high efficacy, n=52), the pathogenic flora decreased significantly: Gardnerella vaginalis – 10 (19.2 ± 5.5%), Candida spp. – 15 (28.8 ± 6.3%), Ureaplasma – 8 (15.4 ± 5.1%), Mycoplasma – 10 (19.2 ± 5.5%). These values were 3–5 times lower compared to baseline, p < 0.001.Although Lactobacillus spp. decreased from 33 (55.0%) to 12 (23.1 ± 5.9%), this was associated with reduced inflammation and elimination of pathogenic flora. Recovery of normal microbiota was expected in the following months, p < 0.05.In Group 2 (partial efficacy, n=8), the reduction of pathogenic flora was insufficient: Gardnerella vaginalis and Lactobacillus spp. – 4 (50.0 ± 18.9%), Candida spp. – 6 (75.0 ± 16.4%), Ureaplasma and Mycoplasma – 5 (62.5 ± 18.3%). These findings indicate the need for an additional course or adjustment of treatment strategy to achieve full efficacy of Bioterol, p < 0.05.Additionally, analysis of triple-site smears showed that before Bioterol administration, the main group (n=60) had high levels of inflammation and pathogenic flora:• Leukocytes: vagina – 56 (93.3 ± 3.2%), cervix – 56 (93.3 ± 3.2%), urethra – 56 (93.3 ± 3.2%)• Epithelial cells: vagina – 48 (80.0 ± 5.2%), cervix – 44 (73.3 ± 5.8%), urethra – 50 (83.3 ± 4.9%)• Mucus: vagina – 42 (70.0 ± 5.9%), cervix – 48 (80.0 ± 5.2%), urethra – 54 (90.0 ± 3.9%)• Fungi: vagina – 55 (91.7 ± 3.6%), cervix – 48 (80.0 ± 5.2%), urethra – 46 (76.7 ± 5.5%)• Trichomonads: vagina – 16 (26.7 ± 5.8%), cervix – 18 (30.0 ± 5.9%), urethra – 3 (5.0 ± 2.8%)One month after administration, in Group 1, inflammation decreased sharply:• Leukocytes: vagina – 11 (21.1 ± 5.7%), cervix – 5 (9.6 ± 4.1%), urethra – 13 (25.0 ± 6.1%), p < 0.001• Epithelial cells and mucus: decreased 3–4 times, vagina – 14 (26.9 ± 5.2%), mucus – 9 (17.3 ± 5.3%)• Fungi: vagina – 15 (28.8 ± 6.3%), cervix – 16 (30.8 ± 6.5%), urethra – 11 (21.1 ± 5.7%)• Trichomonads: decreased nearly 3 times, vagina – 6 (11.5 ± 4.5%), cervix – 4 (7.7 ± 3.7%), urethra – 2 (3.8 ± 7.2%), p < 0.001In Group 2 (n=8), some positive dynamics were also observed:• Leukocytes: vagina – 4 (50.0 ± 18.9%), cervix – 3 (37.5 ± 18.3%), urethra – 5 (62.5 ± 18.3%)• Epithelial cells and mucus: decreased by 25–62%• Fungi: remained high – vagina 7 (87.5 ± 12.5%), cervix 6 (75.0 ± 16.4%), urethra 5 (62.5 ± 18.3%)• Trichomonads: did not exceed 1–2 casesThus, Bioterol reduced signs of inflammation by 3–5 times and pathogenic flora by 3–7 times, and the differences between groups were statistically significant, p = 0.001–0.05.Comparative bacteriological culture analysis of vaginal flora before and after Bioterol administration showed that, prior to treatment, the main group exhibited significant dysbiosis:• Streptococci: 38 (63.3 ± 6.3%)• Enterococci: 42 (70.0 ± 5.9%)• Staphylococci: 32 (53.3 ± 6.5%)• Enterobacteriaceae: 35 (58.3 ± 6.4%)• Candida: 55 (91.7 ± 3.6%). (Table 2).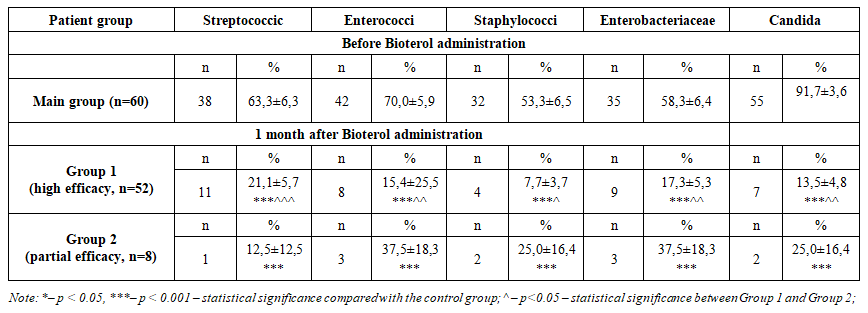 | Table 2. Comparative description of vaginal bacteriological culture analysis before and after Bioterol administration (M±m), % |
One month after administration of the preparation, a marked positive change in microbiological parameters was observed. In Group 1 (high efficacy, n=52) of the main group, the frequency of microorganisms decreased as follows: Streptococci – 11 (21.1 ± 5.7%), Enterococci – 8 (15.4 ± 5.5%), Staphylococci – 4 (7.7 ± 3.7%), Enterobacteriaceae– 9 (17.3 ± 5.3%), and Candida – 7 (13.5 ± 4.8%), p < 0.001.In Group 2 (partial efficacy, n=8), a significant reduction in microbial frequency was also observed: Streptococci – 1 (12.5 ± 12.5%), Enterococci – 3 (37.5 ± 18.3%), Staphylococci – 2 (25.0 ± 16.4%), Enterobacteriaceae – 3 (37.5 ± 18.3%), and Candida – 2 (25.0 ± 16.4%), p < 0.05–0.01.Overall analysis indicated that Bioterol reduced pathogenic flora in the vaginal microbiocenosis by 3–7 times, with statistically significant differences for all parameters, p < 0.001.
5. Conclusions
1. Bioterol demonstrated high efficacy in stabilizing the urogenital tract microbiocenosis.o Pathogenic flora (Gardnerella, Candida, Ureaplasma, Mycoplasma) decreased 3–7 times, p < 0.001.o Leukocytes, a marker of inflammation, decreased 3–5 times.o Restoration of acidity and strengthening of the vaginal immunological barrier were observed.2. In patients with CIN I + HPV(+) and CIN I + HPV(–), disruption of vaginal microbiocenosis was highly prevalent, demonstrating the significant role of dysbiotic vaginal processes in dysplasia progression and persistent HPV infection.3. High therapeutic efficacy was observed in 86.7% of patients treated with Bioterol.o In the remaining 13.3% of patients, partial efficacy was noted, indicating the need for:§ An additional course, or§ Adjustment of the treatment strategy.4. Correction of vaginal microflora during the pre-destruction preparation stage is crucial as an adjunctive therapeutic approach.
References
| [1] | Mitra A, MacIntyre DA, Marchesi JR, Lee YS, Bennett PR, Kyrgiou M. The vaginal microbiota, human papillomavirus infection and cervical intraepithelial neoplasia: what do we know and where are we going next? Microbiome. 2016; 4: 58. (SpringerLink) |
| [2] | Wang H, et al. Associations of Cervicovaginal Lactobacilli With High-Risk Human Papillomavirus Infection, Cervical Intraepithelial Neoplasia, and Cancer: A Systematic Review and Meta-Analysis. The Journal of Infectious Diseases. 2019; 220(8): 1243–1254. (OUP Academic) |
| [3] | Norenhag J, Du J, Olovsson M, Verstraelen H, Engstrand L, Brusselaers N. The Vaginal Microbiota, Human Papillomavirus and Cervical Dysplasia: A Systematic Review and Network Meta-Analysis. BJOG.2020; 127: 171–180. (PubMed) |
| [4] | Dysbiosis of vaginal microbiota associated with persistent high-risk human papilloma virus infection. Journal of Translational Medicine. 2022; 20: 12. (SpringerLink) |
| [5] | Disturbances of Vaginal Microbiome Composition in Human Papillomavirus Infection and Cervical Carcinogenesis: A Qualitative Systematic Review. BMC Infectious Diseases. 2022. (PubMed) |
| [6] | Liu Y, Wang S, Liu J, Su M, Diao X, Liang X, Zhang J, Wang Q, Zhan Y. Characteristics of vaginal microbiota in various cervical intraepithelial neoplasia: a cross-sectional study. Journal of Translational Medicine. 2023; 21: 816. (SpringerLink) |
| [7] | Ma Y, Li Y, Liu Y, Cao L, Han X, Gao S, Zhang C. Vaginal Microbiome Dysbiosis is Associated with the Different Cervical Disease Status. Journal of Microbiology. 2023. (MedNews) |
| [8] | Žukienė G, Narutytė R, Rudaitis V. Association Between Vaginal Microbiota and Cervical Dysplasia Due to Persistent Human Papillomavirus Infection: A Systematic Review of Evidence from Shotgun Metagenomic Sequencing Studies. Int. J. Mol. Sci. 2025; 26(9): 4258. (MDPI) |
| [9] | Yang Q, Dai W, Wu D, et al. Inferred bi-directional interactions between vaginal microbiota, metabolome and persistent HPV infection accompanied by high-grade cervical intraepithelial neoplasia. BMC Microbiology. 2025; 25: 404. (SpringerLink) |
| [10] | Li Y, Wu X. Vaginal microbiome distinction in women with HPV+, cervical intraepithelial neoplasia, and cervical cancer, a retrospective study. Frontiers in Cellular and Infection Microbiology. 2025; 14: 1483544. (PMC) |






 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML