Raimova Malika Muhamedjanovna, Muradova Malika Saidaxrorovna
Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Raimova Malika Muhamedjanovna, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In recent years, the problem of cognitive impairment (CI) has become increasingly important from a medical and socioeconomic perspective. This is due to the increasing life expectancy of people in economically developed countries, resulting in population aging and an increased prevalence of both mild cognitive impairment (MCI) and dementia. Severe cognitive impairment is associated with disability and social maladjustment, a significant reduction in the quality of life of patients and their caregivers, poor treatment adherence, an increased prevalence of severe comorbidities (infections, physical injuries, food aspiration) and death, as well as significant healthcare costs associated with the treatment and care of this group of patients. The use of modern approaches to the diagnosis and treatment of MCI makes it possible to identify patients at high risk of developing dementia in the early stages of the disease and, thus, initiate pathogenetic therapy earlier, aimed at maintaining adequate functioning and a satisfactory quality of life. The medical and social significance of chronic kidney disease (CKD) is determined not only by the progression of renal failure but also by cardiovascular complications, which develop in these patients significantly more frequently than in individuals without signs of renal damage and at a significantly younger age.
Keywords:
Chronic kidney disease, Cognitive impairment, Diabetes mellitus, Cerebrovascular insufficiency, Neuropsychological assessment
Cite this paper: Raimova Malika Muhamedjanovna, Muradova Malika Saidaxrorovna, Clinical and Pathogenetic Features of Cognitive Impairment Formation in Patients with Stage III–IV Chronic Kidney Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 679-683. doi: 10.5923/j.ajmms.20261602.60.
1. Introduction
Today, the concept of CKD as a model of accelerated vascular aging has become generally accepted _, one of the prognostically unfavorable consequences of which can be considered the development of cerebrovascular insufficiency in patients, often leading to the development of dementia. In recent years, a number of studies have demonstrated the role of CKD as an independent risk factor for the development and progression of CR in the general population. It is assumed that cerebrovascular insufficiency, which occurs under the influence of cardiovascular risk factors, underlies the development of cognitive dysfunction in patients with CKD at pre-dialysis stages. Risk factors directly related to kidney damage (hyperhomocysteinemia, anemia, hyperphosphatemia, proteinuria) may also play a role; however, the pathogenetic and prognostic role of each has not been adequately studied [1,2,3].The increasing incidence and prevalence of CKD worldwide, along with the steadily increasing prevalence of vascular cognitive impairment, necessitates the development of methods for the early diagnosis of cognitive dysfunction and assessment of the risk of its progression, as well as a thorough study of modifiable risk factors for cognitive impairment in patients with CKD. This will not only optimize treatment and diagnostic strategies but also improve approaches to primary and secondary prevention of dementia [4].Cognitive impairment is one of the most significant medical and socioeconomic problems, associated with severe disability and a significant decline in the quality of life of patients and their caregivers. In kidney disease, CR not only increases the risk of mortality but also complicates informed patient consent for hemodialysis or kidney transplantation. Several studies have shown that the prevalence of cognitive dysfunction increases with the progression of CKD and occurs in approximately 80% of patients in its late stages. Currently, there is no doubt that cognitive impairment in CKD is caused by both toxic (uremic encephalopathy) mechanisms and vascular remodeling [5,11].Uremic encephalopathy occurs with severe renal failure and usually develops slowly. Its development is largely due to the toxic effects of organic acids, the excretion and metabolism of which are impaired in kidney disease. Among the first symptoms of uremic encephalopathy are increased anxiety, slowed mental processes, fatigue, apathy, and decreased concentration. As the disease progresses, cognitive decline to the point of dementia, severe behavioral disturbances, and sleep inversion are observed. Patients may exhibit pronounced symptoms of frontal lobe damage, such as impaired abstract thinking, paratonia, and axial reflexes. Further progression of the pathological process is accompanied by the occurrence of seizures (usually generalized clonic-tonic), multifocal myoclonus, asterixis, meningismus (in approximately a third of cases), agitation, delirium, and visual hallucinations, which are followed by a coma and death [7,8,9,10].CR in uremic encephalopathy manifests primarily as memory and attention impairment. Given that the level of azotemia only slightly correlates with the severity of neurological impairment, laboratory examination results do not allow one to exclude other causes of encephalopathy and are of little value in assessing the prognosis of neurological disorders.
2. Purpose of the Research
To investigate the clinical and pathogenetic features of cognitive impairment formation in patients with stage III–IV chronic kidney disease.
3. Materials and Methods
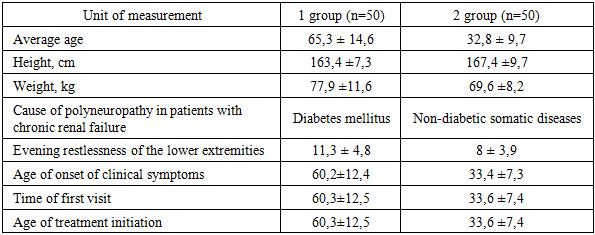
The study design was comprehensive and based on a combination of neurological, immunological, psychological, and neuroimaging studies. A total of 100 participants were enrolled in the study, including 50 patients with chronic kidney disease (CKD) associated with diabetes, 50 patients with CKD without diabetes, and 20 patients with CKD without neurological complications. All participants provided verbal and written consent. The study was conducted in accordance with the standards approved by the Health Ethics Committee. Patients included in the study were selected from among those undergoing inpatient treatment at a branch of the multidisciplinary clinic of the Tashkent Medical Academy and the Republican Specialized Center for Nephrology and Kidney Transplantation, followed by outpatient follow-up, with a diagnosis of CKD (chronic renal failure), and who voluntarily presented to a medical facility.Our clinical study included 100 patients with CKD, with an average age of 23-80 years. Differences in patients by age and gender are presented in Table 1. All patients included in our study were divided into two groups: 50 patients with CRF with diabetic polyneuropathy and 50 patients with CRF with non-diabetic polyneuropathy.Table 1. Clinical characteristics of the study groups
 |
| |
|
According to Table 1, men accounted for 40% of the patients and women for 60%, which is 1.5 times more than men. Seventy percent of the patients were adults and elderly patients; the average age of men was 50.8±19.05 years, and that of women was 55.5±13.03 years.The average age of patients in group 1 was 65.3±14.6 years (range, 55 to 80 years) and in group 2 the average age was 32.8±9.7 years (range, 23 to 55 years). 50 participants in our study suffered from diabetic polyneuropathy and 50 from non-diabetic polyneuropathy. Restlessness in the legs was observed 11.3±4.8 times a day in group 1 and 8±3.9 times in group 2. Clinical signs appeared at the age of 60.2±12.4 years in group 1 and 33.4±7.3 years in group 2. In group 1, patients in group 1 consulted a doctor and started treatment at the age of 60.3±12.5 years, while patients in group 2 started treatment at the age of 33.6±7.4 years. Before joining our study group, patients in the first group had been experiencing polyneuropathy for 5.4±2.6 years, while in the second group this figure was 1.1±0.8 years.Clinical neurological examinations were used as one of the primary outcome measures in the study.The study included 100 patients with stages III-IV chronic kidney disease (CKD). The frequency, severity, and nature of cognitive impairment (CI) were examined depending on the stage of CKD. CI was detected in 68% of all patients examined. The frequency and severity of CI detection in patients with stages III-IV CKD were significantly higher than in the group with stages I-II CKD (p<0.001). Stages III-IV CKD were shown to be an independent predictor of CI development (OR = 27.32, 95% CI 4.3-72.9, p<0.001). Dysfunction of the anterior cerebral cortex and neurodynamic disturbances in patients with early stages of kidney damage play a key role in the development of CI in patients with CKD stages III-IV. The development of CI in CKD is associated with signs of cerebrovascular insufficiency detected by brain MRI (focal white matter changes were detected in 30% of individuals, leukoaraiosis in 23.3%, dilation of the lateral ventricles in 50%, and marked widening of the sulci of the hemispheres in 10%). An increased incidence and severity of CI is associated with risk factors such as male gender, abdominal obesity, anemia, hyperhomocysteinemia, hyperphosphatemia, and target organ damage (left ventricular hypertrophy). An independent predictor of the development of coronary heart disease in CKD is older age (OR = 1.12, 95% CI 1.02-1.22, p = 0.016).An association has been demonstrated with vascular wall remodeling features such as intima-media thickening, the presence of atherosclerotic plaques in the common carotid arteries, and increased vascular wall stiffness, as assessed by pulse wave velocity and augmentation index. In the group of patients with stages III-IV CKD (n = 100), coronary heart disease was observed in 7 (35%) patients, compared with 28 (90.3%) in the group of patients with stages PCV (n = 31). The incidence of coronary heart disease in patients with stages III-IV CKD was significantly higher than in the group with stages I-II CKD (p < 0.001). Among all CKD patients examined, CI was detected in 35 (68%) patients. The incidence of CI in patients with stages III-IV CKD was significantly higher than in the group with stages I-II CKD (p < 0.001).Among all CKD patients examined, CI was detected in 35 (68%) patients. All patients with CI complained of cognitive issues, primarily difficulty memorizing new information, forgetting names, remembering action plans, difficulty remembering objects, attention deficit, difficulty concentrating, and sleep disturbances. However, none of the patients reported significant limitations in their daily activities based on questionnaires assessing their primary and instrumental daily activities.
4. Results
A total of 100 patients with stage III-IV CKD were included in the study. Fifty of them were diagnosed with CKD associated with diabetes (Group 1), while the remaining 50 had CKD without diabetes (Group 2). The patients' cognitive functions were assessed using specialized neuropsychological tests, and the data obtained were analyzed using diagrams. Cognitive impairment is common in patients with advanced CKD and increases with disease progression. Concomitant diabetes mellitus significantly aggravates cognitive dysfunction, highlighting the need for early cognitive screening and comprehensive management of modifiable risk factors.Сognitive impairment in patients of group 1 with diabetes mellitus + chronic kidney disease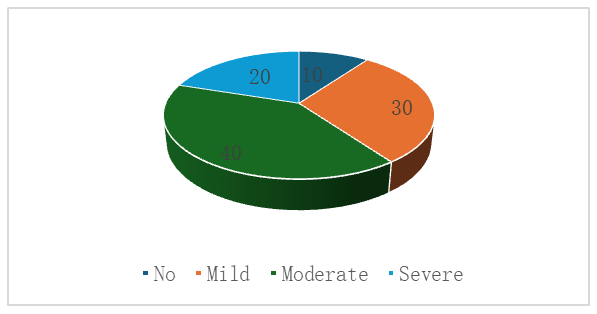 | Figure 1. Cognitive impairment in patients in Group I (DM + CKD) |
These data confirm that the combination of diabetic microangiopathy, uremic toxicity, electrolyte disturbances, and chronic inflammation creates an unfavorable neurometabolic environment that contributes to the accelerated development of cognitive impairment. The high prevalence of moderate and severe forms of cognitive impairment highlights the need for mandatory cognitive screening of patients with DM + CKD, as well as timely, comprehensive correction of systemic risk factors.Cognitive impairment in patients of group 2 (CKD without diabetes mellitus)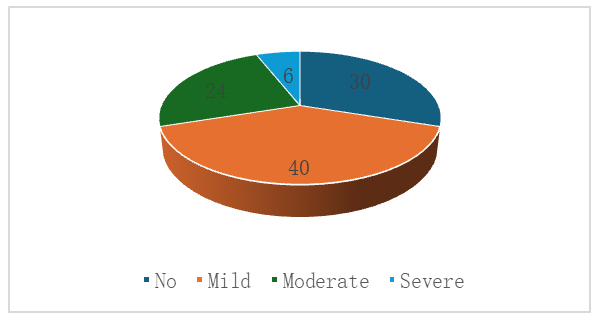 | Figure 2. Cognitive impairment in patients in Group II (CKD without diabetes mellitus) |
Data analysis revealed that among patients in Group II with stages III–IV CKD without concomitant diabetes mellitus, cognitive impairment was recorded in 70% of those examined. The cognitive deficits were predominantly mild (40%) and moderate (24%), indicating a predominantly subclinical or moderate nature of cognitive decline in this group.Severe cognitive impairment was detected in only 6% of patients, which is significantly lower than in patients with a combination of diabetes and CKD. This confirms the role of diabetes mellitus as an additional factor exacerbating neurocognitive impairment due to diabetic microangiopathy, chronic hyperglycemia, and metabolic instability.Preservation of cognitive function was observed in 30% of patients, which is significantly higher than in the group with diabetes and CKD. The results indicate that in the absence of diabetes, cognitive impairment is less frequent and generally less severe. Thus, CKD is an independent risk factor for the development of cognitive impairment, but concomitant diabetes significantly exacerbates the severity of cognitive dysfunction.Comparative analysis of cognitive impairment between groups in stage III-IV CKD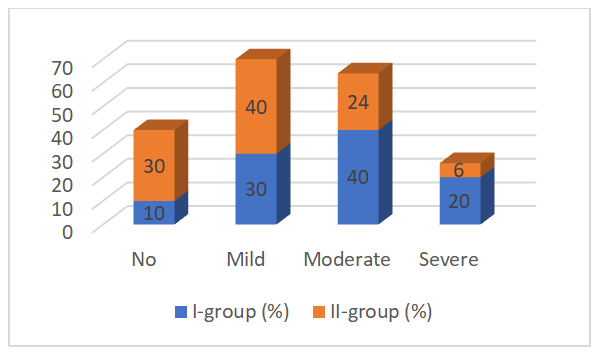 | Figure 3. Comparative analysis of cognitive impairment between groups in CKD stages III–IV |
A comparative analysis of cognitive impairment in patients in the two study groups revealed significant differences in the structure and severity of cognitive deficits. In patients in Group I (DM + CKD), cognitive impairment was significantly more frequent and more severe compared to Group II (CKD without diabetes). Moderate (40%) and severe (20%) impairments predominated in Group I, while in Group II, the proportion of severe impairments was minimal (6%), with mild cognitive impairment predominating (40%).Normal cognitive status was observed in only 10% of patients in Group I, compared to 30% of patients with isolated CKD. This indicates that diabetes mellitus is a significant independent risk factor for cognitive decline, exacerbating the effects of uremic intoxication, dysmetabolic processes, and vascular disorders characteristic of CKD.Thus, these results confirm that the combination of CKD and diabetes mellitus has a significant negative impact on patients' cognitive status and requires earlier screening, monitoring, and a comprehensive approach to risk factor correction. The identified differences substantiate the need for individualized neuroprotection and metabolic stabilization programs in patients with comorbid diabetes and CKD.When assessing the severity of cognitive impairment in patients with different stages of CKD, a deterioration in Mini-Mental State Examination, Frontal Lobe Function Test, and Executive Function Test scores was noted with increasing CKD stage. It is worth noting that no patients exhibited severe cognitive impairment based on testing results combined with limitations in daily activity, ruling out dementia and suggesting the development of MCI. In the structure of cognitive dysfunction in the examined patients, the polyfunctional type of UCI was predominant, both with memory impairment (in 14 (40.0%) patients) and without them (15 (42.9%) patients). Monofunctional types of UCI were significantly less common - amnestic in 4 (11.4%) patients, non-amnestic (with executive function impairment) - in 2 (5.7%).Of all the examined CKD patients, depression was detected in 26 (51%) people, of which 21 (41.2%) had a mild degree and 5 (9.8%) had a severe degree. Among patients with depression, UCI was observed in 18 (69.2%), of which 15 (83.3%) had a mild degree of depression, and 3 (16.7%) had a severe degree. Among the 25 patients without depression, UCI was detected in 17 (68%). That is, the incidence of MCI in patients with depression did not differ from that in patients without depression (Figure 3). There were also no statistically significant differences in the severity of MCI between the groups of patients with and without depression.Our study found that MCI in patients with CKD are detected already at the early stages of kidney disease and are significantly more common in patients with CKD stages III-IV than in CKD stages I-II. It was confirmed that CKD stages III-IV is an independent predictor of MCI, which is consistent with the results of similar studies. For example, the large REGARDS (Reasons for Geographic and Racial Differences in Stroke) study demonstrated an increased risk of developing MCI in patients with chronic kidney disease compared to those without it (OR = 1.23, 95% CI 1.06-1.43) [2]. Another study showed that in patients with chronic renal failure, a decrease in SCF for every 10 ml/min/1.73 m2 is associated with an 11–27% increase in the risk of developing cognitive impairment [5]. A study by S. Seliger et al. demonstrated a 37% increase in the risk of developing dementia in elderly patients with CKD stages III–IV compared to individuals with an SCF> 60 ml/min/1.73 m2 [8].Our study revealed an increase in the severity of cognitive impairment with increasing CKD stage, meaning that patients with a higher stage of CKD demonstrated significantly lower neuropsychological testing results. It is important to note that cognitive impairment in none of the patients reached the level of dementia according to testing results. These data, along with the absence of limitations in daily activity in the examined patients with cognitive impairment in the presence of cognitive complaints, suggest the development of mild cognitive impairment in them. However, this does not exclude the possibility of the development of severe CP in this group of patients at later pre-dialysis stages of CKD, since it should be taken into account that the selection of patients for this study was carried out in a therapeutic hospital, where patients with dementia are rarely admitted.
5. Discussion
The results of this study demonstrate that cognitive impairment develops already at early stages of kidney damage and occurs significantly more frequently in patients with CKD stages III–IV. The severity of cognitive dysfunction increases with disease progression.The absence of severe cognitive impairment accompanied by limitations in daily activities suggests that most patients had mild cognitive impairment rather than dementia. However, this does not exclude the possibility of further progression at later predialysis stages.The findings are consistent with previous studies, including the REGARDS study, which demonstrated an increased risk of cognitive impairment in patients with chronic kidney disease. The synergistic effect of diabetes mellitus and CKD appears to accelerate cerebrovascular and neurodegenerative processes through mechanisms such as microangiopathy, chronic inflammation, metabolic instability, and uremic toxicity.
6. Conclusions
1. Cognitive impairment is significantly more common in patients with CKD stages III–IV than in those with earlier stages, and its severity increases with disease progression.2. Dysfunction of the frontal brain regions plays a key role in the development of cognitive impairment at advanced stages of CKD.3. Diabetes mellitus significantly aggravates cognitive dysfunction in patients with CKD and should be considered a major independent risk factor.4. Early cognitive screening and comprehensive management of cardiovascular and metabolic risk factors are essential to prevent progression of cognitive impairment in CKD patients.
References
| [1] | Aidarov Z. A. et al. Medical and social aspects of renal and cerebrovascular diseases // The Scientific Heritage. - 2020. - No. 48-2. - P. 64-70. |
| [2] | Vasilyeva I. A. Quality of life of patients with chronic renal failure II Nephrology. - 2003. - No. 1. - P. 26-40. |
| [3] | Damulin I. V., Voskresenskaya O. N. Neurological disorders in chronic kidney disease // Neurological Bulletin. - 2017. - Vol. 49. - No. 1. - P. 34-39. |
| [4] | Zinovieva O. E. Antioxidant therapy in the treatment of dysmetabolic polyneuropathy. Department of Nervous Diseases, M. A. Sechenov Moscow Medical Academy, Moscow. Neurological Journal, Vol. 8, No. 8, 2006. |
| [5] | Murkamilov, I. T., Aitbaev, K. A., Fomin, V. V., Murkamilova, Zh. A., Yusupov, F. A., Raimzhanov, Z. R., & Schastlivenko, A. I. (2020). Risk factors for stroke in patients with end-stage chronic kidney disease: current state of the problem. Kazan Medical Journal, 101(6), 825-833. |
| [6] | Nikitina, A. A., Khrulyov, A. E. Cerebrovascular disorders of the pre-dialysis period of chronic kidney disease and mechanisms of their development // Medical Almanac. - 2018. - No. 5 (56). |
| [7] | Sinyukhin V. N. et al. Neurological disorders in chronic kidney disease // Experimental and clinical urology. - 2017. - No. 2. - P. 92-101. |
| [8] | Khudaiarova S. M., Rakhmatullaeva G. K. Clinical and neurological features and pathogenesis of cerebrovascular diseases in chronic kidney disease // Journal of Biomedicine and Practice. - Tashkent, 2020. - No. 4. - P. 165-173. |
| [9] | Khudaiarova S. M., Rakhmatullaeva G. K. Clinical and neurological features of nervous system damage in chronic kidney disease // Journal of Biomedicine and Practice. - Tashkent, 2020. - No. 6. - P. 233-237. |
| [10] | Kobayashi M, Hirawa N, Yatsu K, Kobayashi Y, Yamamoto Y, Saka S, et al. Relationship between silent brain infarction and chronic kidney disease. Nephrol Dial Transplant 2009; 24: 201–207. DOI:10.1093/ndt/gfn419. |
| [11] | Watanabe K, Watanabe T, Nakayama M. Cerebrorenal interactions: impact of uremic toxins on cognitive function. Neurotoxicology 2014; 44: 184–193. DOI:10.1016/j.neuro.2014.06.014. |





 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML