-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 622-626
doi:10.5923/j.ajmms.20261602.49
Received: Jan. 14, 2026; Accepted: Feb. 6, 2026; Published: Feb. 11, 2026

Immunological Determinants of Postoperative Enterocolitis in Children with Hirschsprung's Disease
Abdurahmonov Isroiljon Ibrohimjon ugli1, Umarov Bakhtiyorjon Yatgarovich2
1DSc, Director of the Children's National Medical Center
2Free Applicant, Children's National Medical Center
Correspondence to: Abdurahmonov Isroiljon Ibrohimjon ugli, DSc, Director of the Children's National Medical Center.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Hirschsprung’s disease remains one of the most challenging congenital disorders of the gastrointestinal tract in children despite significant advances in pediatric surgery. Hirschsprung-associated postoperative enterocolitis is the most severe complication and develops in 25–42% of operated patients, leading to recurrent hospitalizations, prolonged treatment, and increased mortality risk. Growing evidence suggests that immunological mechanisms play a decisive role in the development and severity of this complication. The aim of the present study was to identify immunological determinants associated with the development of postoperative enterocolitis in children with hirschsprung’s disease and to assess their prognostic significance. A total of 153 children with histologically confirmed hirschsprung’s disease were enrolled. Postoperative enterocolitis developed in 75 patients, while 78 children demonstrated an uncomplicated postoperative course.Comprehensive evaluation of innate, adaptive, and mucosal immune parameters was performed before surgery and during the early postoperative period. The development of postoperative enterocolitis was associated with a significant reduction in cd4⁺ t lymphocytes, a decreased cd4/cd8 ratio, imbalance of il-6/il-10 cytokines, reduced secretory iga levels, and increased expression of tlr4, scd14, and zonulin. These findings enabled the development of a clinic-immunological risk prediction model with high diagnostic accuracy. The proposed approach provides a basis for early risk stratification and personalized prevention strategies in children undergoing surgical treatment for hirschsprung’s disease.
Keywords: Hirschsprung’s disease, Postoperative enterocolitis, Children, Immunological determinants, Innate immunity, Mucosal immunity, Cytokines, Risk prediction
Cite this paper: Abdurahmonov Isroiljon Ibrohimjon ugli, Umarov Bakhtiyorjon Yatgarovich, Immunological Determinants of Postoperative Enterocolitis in Children with Hirschsprung's Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 622-626. doi: 10.5923/j.ajmms.20261602.49.
1. Relevance
- Hirschsprung’s disease is a congenital disorder characterized by the absence of enteric ganglion cells in variable segments of the intestine, resulting in severe motility impairment and functional bowel obstruction [1]. The incidence of the disease is estimated at 1 per 4,500–5,000 live births and remains stable worldwide, while postoperative complications continue to pose a significant clinical challenge [4]. Postoperative enterocolitis is the most serious complication associated with hirschsprung’s disease, occurring in 25–42% of patients even after technically successful surgery [5]. This condition is characterized by severe intestinal inflammation, impaired epithelial barrier function, bacterial translocation, and a high risk of systemic inflammatory response and sepsis [6]. Mortality rates in severe cases range from 5% to 10%, emphasizing the high medical and social burden of this complication. Traditionally, the pathogenesis of postoperative enterocolitis has been attributed to mechanical obstruction, residual aganglionosis, and intestinal dysbiosis. However, recent studies have demonstrated that immune dysregulation plays a pivotal role in triggering and maintaining inflammatory processes [3,7]. Children with hirschsprung’s disease often exhibit preoperative immune abnormalities, including impaired t-cell regulation, decreased secretory iga levels, and excessive activation of innate immune receptors. Of particular interest is the imbalance between pro-inflammatory and anti-inflammatory cytokines, especially elevated il-6 and tnf-α levels combined with insufficient il-10 production [2,9]. Additionally, increased expression of toll-like receptor 4 and elevated circulating markers of microbial translocation, such as scd14 and zonulin, reflect compromised intestinal barrier integrity and exaggerated innate immune activation [3,5]. Despite accumulating evidence, clinically applicable prognostic systems incorporating immunological markers are still lacking. Most existing models rely solely on clinical and surgical variables and fail to account for immune-mediated mechanisms. Therefore, comprehensive investigation of immune parameters and their integration into predictive models is highly relevant [1,4]. Early identification of children at high risk of postoperative enterocolitis would allow targeted monitoring, timely preventive measures, and personalized postoperative management, ultimately improving surgical outcomes and quality of life [7,10].Aim of the study The aim of this study was to identify immunological determinants associated with the development of postoperative enterocolitis in children with hirschsprung’s disease and to evaluate their prognostic value for developing a clinic-immunological risk stratification model to optimize postoperative monitoring and preventive strategies.
2. Materials and Methods
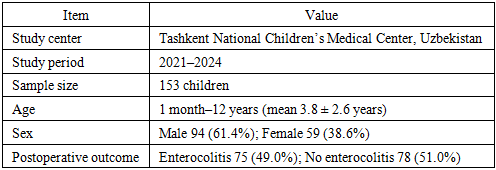
- This prospective observational study was conducted at the Tashkent National Children’s Medical Center (TNCMC), Tashkent, Republic of Uzbekistan, a tertiary referral center providing specialized surgical care for children with congenital gastrointestinal disorders. The study was carried out in the departments of pediatric surgery and pediatric gastroenterology between January 2021 and December 2024.A total of 153 children with histologically confirmed Hirschsprung’s disease were consecutively enrolled. The age of the patients ranged from 1 month to 12 years, with a mean age of 3.8 ± 2.6 years. The study population included 94 boys (61.4%) and 59 girls (38.6%). All patients underwent definitive surgical treatment according to standardized institutional protocols.Based on postoperative clinical outcomes, the patients were divided into two groups. The enterocolitis group consisted of 75 children (49.0%) who developed postoperative Hirschsprung-associated enterocolitis within the first 30 days after surgery. The control group included 78 children (51.0%) with an uncomplicated postoperative course and no clinical or laboratory signs of enterocolitis. The diagnosis of postoperative enterocolitis was established using accepted clinical criteria, including abdominal distension, diarrhea, fever, systemic inflammatory response, and laboratory evidence of inflammation.Immunological assessment was performed at two time points: preoperatively and on postoperative days 3–5. Peripheral venous blood samples were collected under sterile conditions in the morning hours. Cellular immunity was evaluated by flow cytometry and included determination of CD3⁺, CD4⁺, and CD8⁺ T lymphocytes, calculation of the CD4/CD8 ratio, and assessment of CD16⁺56⁺ natural killer (NK) cells.Humoral immunity was assessed by measuring serum concentrations of IgA, IgM, and IgG using immunoturbidimetric assays. Mucosal immunity was evaluated by determining secretory IgA levels. Cytokine profiling included measurement of interleukin-6 (IL-6), interleukin-10 (IL-10), and tumor necrosis factor-α (TNF-α) using enzyme-linked immunosorbent assay (ELISA). Markers of innate immune activation and intestinal barrier dysfunction, including Toll-like receptor 4 (TLR4), soluble CD14 (sCD14), and zonulin, were also measured by ELISA.The study was conducted in accordance with the principles of the Declaration of Helsinki. The research protocol was approved by the Local Ethics Committee of the Tashkent National Children’s Medical Center (Protocol No. 07/21, dated March 15, 2021). Written informed consent was obtained from the parents or legal guardians of all participating children prior to inclusion in the study.Statistical analysis was performed using standard methods of variational statistics. Quantitative data are presented as mean ± standard deviation (M ± SD). Intergroup comparisons were performed using the Mann–Whitney U test and χ² test, with statistical significance set at p < 0.05.
|
3. Results
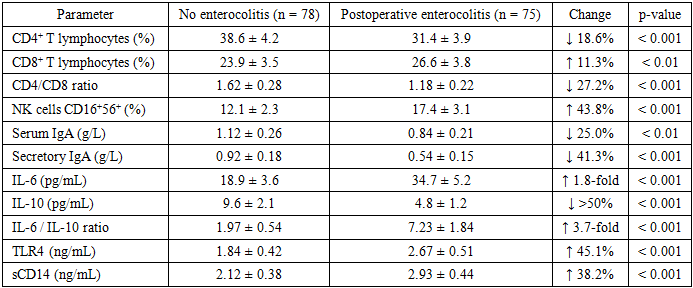
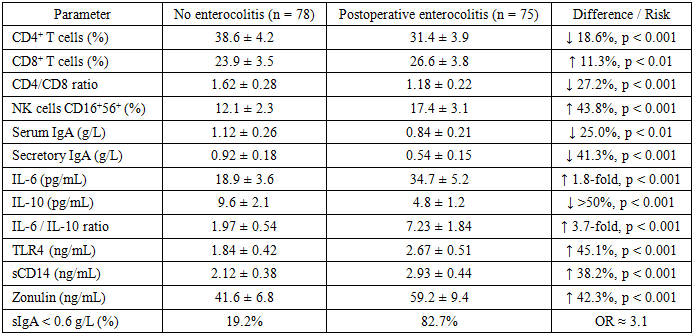
- A total of 153 children with histologically confirmed Hirschsprung’s disease were included in the analysis. During the first 30 postoperative days, postoperative enterocolitis developed in 75 patients (49.0%), while 78 children (51.0%) had an uncomplicated postoperative course. Comparative analysis revealed marked and statistically significant differences in immune parameters between the two groups, affecting adaptive, innate, and mucosal immunity. Assessment of cellular immunity demonstrated a significant reduction in CD4⁺ T lymphocytes in children who developed postoperative enterocolitis. The mean proportion of CD4⁺ cells in the enterocolitis group was 31.4 ± 3.9%, which was 18.6% lower than in children without complications (38.6 ± 4.2%, p < 0.001). In contrast, the percentage of CD8⁺ T lymphocytes was moderately increased (26.6 ± 3.8% vs. 23.9 ± 3.5%, p < 0.01). As a result, the CD4/CD8 ratio declined from 1.62 ± 0.28 in the control group to 1.18 ± 0.22 in the enterocolitis group, representing a 27.2% reduction (p < 0.001), indicative of impaired T-cell immunoregulatory balance. Markers of innate cellular immunity also differed significantly. The proportion of CD16⁺56⁺ natural killer (NK) cells was significantly higher in children with postoperative enterocolitis (17.4 ± 3.1%) compared with those without complications (12.1 ± 2.3%), corresponding to a 43.8% increase (p < 0.001). This finding reflects excessive activation of innate immune mechanisms in the postoperative period. Analysis of humoral immunity revealed a selective IgA deficiency. Serum IgA concentrations were significantly lower in the enterocolitis group (0.84 ± 0.21 g/L) than in the control group (1.12 ± 0.26 g/L), representing a 25.0% decrease (p < 0.01). In contrast, serum IgG and IgM levels did not differ significantly between groups (p > 0.05). More pronounced changes were observed in mucosal immunity. Levels of secretory IgA were markedly reduced in children with postoperative enterocolitis (0.54 ± 0.15 g/L) compared with controls (0.92 ± 0.18 g/L), corresponding to a 41.3% reduction (p < 0.001). Cytokine profiling demonstrated a strong pro-inflammatory shift. Mean IL-6 concentrations in the enterocolitis group were almost 1.8-fold higher than in children without complications (34.7 ± 5.2 pg/mL vs. 18.9 ± 3.6 pg/mL, p < 0.001). Conversely, IL-10 levels were reduced by more than 50% (4.8 ± 1.2 pg/mL vs. 9.6 ± 2.1 pg/mL, p < 0.001). Consequently, the IL-6/IL-10 ratio increased from 1.97 ± 0.54 in the control group to 7.23 ± 1.84 in the enterocolitis group, representing a 3.7-fold increase (p < 0.001). Significant differences were also observed in markers of innate immune activation and intestinal barrier function. Mean TLR4 levels were 45.1% higher in children with enterocolitis (2.67 ± 0.51 ng/mL) than in those without complications (1.84 ± 0.42 ng/mL, p < 0.001). Similarly, sCD14 concentrations increased by 38.2% (2.93 ± 0.44 ng/mL vs. 2.12 ± 0.38 ng/mL, p < 0.001). Levels of zonulin, a marker of intestinal permeability, were elevated by 42.3% in the enterocolitis group (59.2 ± 9.4 ng/mL) compared with controls (41.6 ± 6.8 ng/mL, p < 0.001). Overall, the results demonstrate that postoperative enterocolitis in children with Hirschsprung’s disease is associated with profound immune dysregulation characterized by impaired T-cell regulation, marked mucosal IgA deficiency, excessive innate immune activation, and compromised intestinal barrier integrity.
|
|
4. Conclusions
- The present study demonstrates that postoperative enterocolitis in children with Hirschsprung’s disease is closely associated with a complex and measurable immune imbalance involving adaptive, innate, and mucosal immunity. Postoperative enterocolitis developed in 49.0% of the studied patients, emphasizing the high clinical relevance of early risk identification.Children who developed enterocolitis showed a pronounced reduction in adaptive immune regulation, with CD4⁺ T lymphocytes decreased by 18.6% and the CD4/CD8 ratio reduced by 27.2% compared with patients without complications. Mucosal immune defense was markedly impaired, as reflected by a 41.3% decrease in secretory IgA, with critically low levels (<0.6 g/L) detected in 82.7% of affected children. In contrast, only 19.2% of patients without enterocolitis demonstrated similar deficiency.The inflammatory profile was characterized by a strong pro-inflammatory shift. IL-6 concentrations were increased nearly 1.8-fold, while IL-10 levels were reduced by more than 50%, resulting in a 3.7-fold elevation of the IL-6/IL-10 ratio. Markers of innate immune activation and intestinal barrier dysfunction were also significantly elevated, including TLR4 (+45.1%), sCD14 (+38.2%), and zonulin (+42.3%), indicating increased intestinal permeability and microbial translocation.Importantly, the presence of three or more abnormal immune markers was associated with a 4.5-fold higher risk of postoperative enterocolitis, with an incidence of 78.4% in this high-risk subgroup. These findings confirm that postoperative enterocolitis is not merely a surgical complication but an immune-mediated condition. Integration of immunological profiling into routine perioperative assessment may enable early risk stratification, targeted monitoring, and personalized preventive strategies, ultimately reducing the incidence and severity of postoperative enterocolitis in children with Hirschsprung’s disease.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML