-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 618-621
doi:10.5923/j.ajmms.20261602.48
Received: Jan. 14, 2026; Accepted: Feb. 2, 2026; Published: Feb. 11, 2026

Clinical, Echocardiographic, and Genetic Markers for Early Detection of Chronic Heart Failure in Patients with Coronary Heart Disease
Kenjayev Majid Latypovich1, Khudaykulova Visola Dilmurodovna2
1Chief Medical Officer of the Tashkent Regional Specialized Somatic Hospital
2Basic Doctoral Student, Department of Faculty and Hospital Therapy, Nephrology and Hemodialysis No. 2, Tashkent Medical Academy
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Chronic heart failure (CHF) is one of the most severe complications of coronary heart disease (CHD) and remains a leading cause of morbidity and mortality worldwide. Early identification of CHF in patients with CHD is essential for improving prognosis and optimizing individualized treatment strategies. This study aimed to evaluate the diagnostic value of combined clinical, echocardiographic, laboratory, and genetic markers for early detection of CHF in patients with CHD.A total of 190 individuals were enrolled, including patients with CHD complicated by CHF (n = 69), patients with CHD without CHF (n = 33), and healthy controls (n = 88). All participants underwent clinical assessment, electrocardiography, Doppler echocardiography with speckle-tracking analysis of global longitudinal strain (GLS), laboratory testing (NT-proBNP, CRP, lipid profile), and molecular-genetic analysis of the MMP9 (Gln279Arg) polymorphism.Patients with CHF demonstrated significantly reduced left ventricular ejection fraction and GLS, along with markedly elevated NT-proBNP levels (1620 ± 430 pg/ml) compared to CHD patients without CHF and healthy individuals (p < 0.001). The Arg allele and Arg/Arg genotype of the MMP9 gene were associated with a 2–3-fold increased risk of CHF development. The combined diagnostic approach enables early detection of subclinical myocardial dysfunction and improves risk stratification in CHD patients.
Keywords: Coronary heart disease, Chronic heart failure, NT-proBNP, Global longitudinal strain, MMP9 polymorphism
Cite this paper: Kenjayev Majid Latypovich, Khudaykulova Visola Dilmurodovna, Clinical, Echocardiographic, and Genetic Markers for Early Detection of Chronic Heart Failure in Patients with Coronary Heart Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 618-621. doi: 10.5923/j.ajmms.20261602.48.
1. Relevance
- Coronary heart disease remains the leading cause of cardiovascular mortality, and chronic heart failure represents its most frequent and disabling complication. According to international data, approximately 30–40% of patients with CHD eventually develop CHF, which significantly worsens quality of life and survival [2]. Despite advances in cardiology, early diagnosis of CHF remains challenging due to the absence of clear clinical symptoms in the initial stages [4]. Traditional diagnostic tools, such as standard echocardiographic parameters and clinical assessment, often fail to detect early myocardial dysfunction. Therefore, the identification of sensitive markers capable of revealing subclinical cardiac impairment is of paramount importance. In recent years, speckle-tracking echocardiography and measurement of global longitudinal strain have demonstrated superior sensitivity compared to conventional ejection fraction in detecting early systolic dysfunction [6]. Biomarkers such as NT-proBNP provide valuable information about myocardial wall stress and hemodynamic overload and are widely recommended for CHF diagnosis and risk assessment. Elevated NT-proBNP levels have been consistently associated with disease severity, adverse outcomes, and hospitalization rates [1,8]. Genetic predisposition also plays a crucial role in the progression of CHF. Matrix metalloproteinase-9 (MMP-9) is involved in extracellular matrix remodeling, myocardial fibrosis, and ventricular dilation. Polymorphisms of the MMP9 gene, particularly Gln279Arg, may influence enzyme activity and contribute to accelerated myocardial remodeling. International studies suggest that the Arg allele is associated with increased cardiovascular risk; however, data in Central Asian populations remain limited [3,10].In Uzbekistan, most studies on CHF have focused on clinical and instrumental diagnostics, while molecular-genetic factors have not been sufficiently integrated into routine evaluation. The lack of comprehensive studies combining echocardiographic deformation imaging, biomarkers, and genetic markers underscores the relevance of this research [4]. This study addresses an important gap by integrating clinical, echocardiographic, biochemical, and genetic parameters for early CHF detection. Such a multimodal approach may improve diagnostic accuracy, allow timely initiation of therapy, reduce healthcare costs, and ultimately decrease morbidity and mortality associated with CHD-related heart failure [2,6].Aim of the Study The aim of this study was to evaluate the diagnostic and prognostic significance of clinical indicators, echocardiographic parameters (including global longitudinal strain), laboratory biomarkers (NT-proBNP), and the MMP9 (Gln279Arg) gene polymorphism for early detection of chronic heart failure in patients with coronary heart disease, as well as to identify high-risk individuals for targeted preventive and therapeutic interventions.
2. Materials and Methods
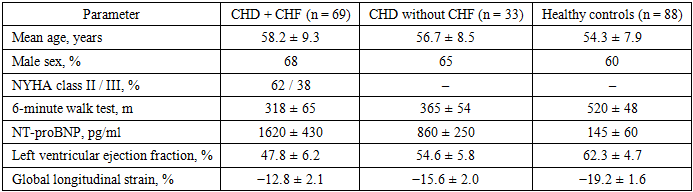
- This prospective clinical, instrumental, and genetic study was carried out between 2023 and 2025 at the Tashkent Regional Multidisciplinary Somatic Hospital. A total of 190 individuals were included in the study and divided into three groups. The first group consisted of patients with coronary heart disease complicated by chronic heart failure (CHD + CHF, n = 69, 34.5%). The second group included patients with confirmed coronary heart disease without clinical signs of chronic heart failure (CHD, n = 33, 16.5%). The control group comprised 88 apparently healthy volunteers (44.0%) without a history or clinical evidence of cardiovascular disease.The mean age of patients in the CHD + CHF group was 58.2 ± 9.3 years, in the CHD group 56.7 ± 8.5 years, and in the control group 54.3 ± 7.9 years. Male participants predominated in all groups, accounting for 68%, 65%, and 60%, respectively. Inclusion criteria were a documented diagnosis of coronary heart disease and informed consent to participate in the study. Exclusion criteria included severe valvular heart disease, symptomatic hypotension, advanced chronic kidney disease (estimated glomerular filtration rate <30 ml/min/1.73 m²), significant arrhythmias, oncological or autoimmune disorders, pregnancy, and refusal to undergo genetic testing. All participants underwent a comprehensive clinical examination, 12-lead electrocardiography, and transthoracic Doppler echocardiography using a Vivid S70 ultrasound system (GE Healthcare, USA) in accordance with the recommendations of the American Society of Echocardiography. Left ventricular systolic and diastolic functions were assessed by measuring ejection fraction, end-diastolic and end-systolic volumes, transmitral flow parameters (E/A ratio, E/e′), isovolumetric relaxation time, and left atrial volume index. Speckle-tracking echocardiography was performed to determine global longitudinal strain from three apical projections using EchoPAC software. Functional capacity was assessed using the 6-minute walk test. Venous blood samples were collected to measure NT-proBNP, C-reactive protein, glucose, lipid profile, creatinine, homocysteine, and lipoprotein(a). NT-proBNP concentration was determined by immunoassay, and values above 125 pg/ml were considered elevated in patients younger than 75 years.
|
3. Results
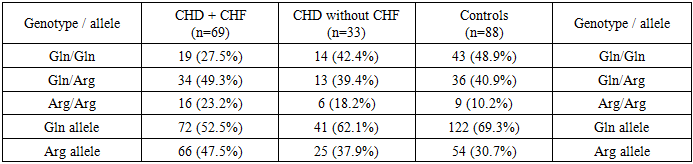
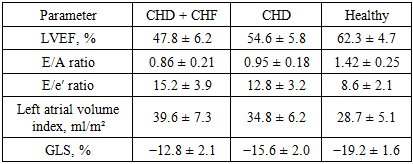
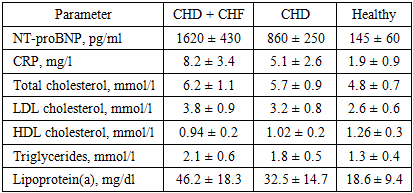
- Clinical and functional characteristics of the study groups were analyzed and compared between patients with coronary heart disease complicated by chronic heart failure (CHD + CHF), patients with coronary heart disease without heart failure (CHD), and healthy controls. The mean age of patients in the CHD + CHF group was 58.2 ± 9.3 years, which did not differ significantly from the CHD group (56.7 ± 8.5 years) and the healthy control group (54.3 ± 7.9 years; p > 0.05). Male patients predominated in all groups, accounting for 68% in the CHD + CHF group, 65% in the CHD group, and 60% among healthy controls. According to NYHA classification, all patients in the CHD + CHF group were categorized as class II (62%) or class III (38%), whereas no symptoms of heart failure were observed in the CHD-only group and controls. Functional capacity assessed by the 6-minute walk test was significantly reduced in patients with CHF. The mean walking distance was 318 ± 65 m in the CHD + CHF group, compared to 365 ± 54 m in the CHD group and 520 ± 48 m in healthy individuals (p < 0.001), indicating marked limitation of physical performance in CHF patients. Echocardiographic evaluation revealed pronounced impairment of left ventricular systolic and diastolic function in the CHD + CHF group. Left ventricular ejection fraction was significantly lower in CHD + CHF patients (47.8 ± 6.2%) compared with CHD patients without CHF (54.6 ± 5.8%) and healthy controls (62.3 ± 4.7%; p < 0.05). Global longitudinal strain showed even more pronounced differences, with values of −12.8 ± 2.1% in the CHD + CHF group, −15.6 ± 2.0% in the CHD group, and −19.2 ± 1.6% in healthy subjects (p < 0.001). These findings indicate the presence of subclinical myocardial dysfunction in CHD patients and advanced systolic impairment in CHF patients. Diastolic function parameters were also significantly altered in the CHD + CHF group. The E/A ratio was reduced to 0.86 ± 0.21 compared with 0.95 ± 0.18 in the CHD group and 1.42 ± 0.25 in controls. The E/e′ ratio was elevated in CHF patients (15.2 ± 3.9), reflecting increased left ventricular filling pressure. Left atrial volume index was significantly higher in the CHD + CHF group (39.6 ± 7.3 ml/m²) compared to CHD patients (34.8 ± 6.2 ml/m²) and healthy individuals (28.7 ± 5.1 ml/m²). Laboratory analysis demonstrated marked neurohormonal and inflammatory activation in CHF patients. NT-proBNP levels were highest in the CHD + CHF group (1620 ± 430 pg/ml), moderately elevated in the CHD group (860 ± 250 pg/ml), and within normal range in healthy controls (145 ± 60 pg/ml; p < 0.001). C-reactive protein was significantly elevated in CHF patients (8.2 ± 3.4 mg/l), indicating active inflammatory processes. Lipid metabolism disorders were more pronounced in the CHD + CHF group, with increased total cholesterol, LDL cholesterol, triglycerides, lipoprotein(a), and homocysteine levels, along with reduced HDL cholesterol. Genetic analysis of the MMP9 Gln279Arg polymorphism revealed a significantly higher frequency of the Arg allele in patients compared to controls. The Arg allele was detected in 47.5% of alleles in the patient group versus 30.7% in healthy individuals (χ² = 11.2, p = 0.01), increasing the risk of CHF development (OR = 2.0, 95% CI 1.35–3.12). The Arg/Arg genotype was more common in patients (23.5%) than in controls (10.2%), with an odds ratio of 2.7 (95% CI 1.21–6.05), indicating a strong association with disease progression.
|
|
|
4. Conclusions
- The results of this study demonstrate that chronic heart failure develops in a substantial proportion of patients with coronary heart disease and is associated with pronounced clinical, functional, biochemical, and genetic changes. In the studied cohort, patients with CHD complicated by CHF accounted for 34.5% of all examined individuals, highlighting the high burden of heart failure among patients with ischemic heart disease. Clinical assessment showed that all patients with CHF belonged to NYHA functional class II or III, with 62% classified as class II and 38% as class III. Reduced functional capacity was observed in 72% of CHF patients, as reflected by a significantly shortened 6-minute walk distance (318 ± 65 m), compared with patients without CHF and healthy individuals. Signs of pulmonary congestion were present in 36% of CHF patients, and peripheral edema was detected in 41%, indicating clinically significant volume overload. Echocardiographic evaluation revealed that systolic dysfunction was present in the majority of CHF patients. Left ventricular ejection fraction was reduced below 50% in approximately 58% of patients in the CHD + CHF group, while subclinical systolic dysfunction assessed by global longitudinal strain was detected in more than 80% of these patients. GLS values above −15% were observed in 83% of CHF patients and in 46% of CHD patients without overt heart failure, confirming the high sensitivity of strain imaging for early myocardial dysfunction. Diastolic dysfunction markers, including reduced E/A ratio and elevated E/e′ ratio, were present in over 70% of CHF patients, reflecting increased left ventricular filling pressures. Laboratory findings further supported the presence of advanced myocardial stress and systemic inflammation. Elevated NT-proBNP levels above diagnostic thresholds were detected in 100% of CHF patients, with mean values exceeding those of CHD patients without CHF by 88%. Increased C-reactive protein levels were observed in 78% of CHF patients, indicating active inflammatory processes. Lipid metabolism disorders were highly prevalent, with elevated LDL cholesterol in 70%, increased triglycerides in 64%, and reduced HDL cholesterol in 72% of CHF patients. Lipoprotein(a) concentrations above reference values were detected in 68% of CHF patients, underscoring its role as an independent cardiovascular risk factor. Genetic analysis demonstrated a significant association between the MMP9 Gln279Arg polymorphism and disease severity. The Arg allele was present in 47.5% of patients compared with 30.7% of healthy controls, increasing the risk of heart failure development by approximately 2-fold. The Arg/Arg genotype was identified in 23.5% of patients versus 10.2% of controls, indicating a 2–3-fold higher genetic risk. In conclusion, the integration of clinical assessment, advanced echocardiography, biomarker analysis, and genetic testing enables early identification of high-risk patients, improves risk stratification, and provides a foundation for personalized management strategies in coronary heart disease.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML