Mirzajonova Dono Bahodirovna1, Ruziyev Azizjon Mahmut ugli2, Magzumov Khamidullo Bashirovich3, Maksudova Zulfiya Sanatovna4, Imamova Ilmira Abdullaevna5, Gaybullayev Fazliddin Khayriddin ugli6
1Tashkent State Medical University, Republican Specialized Scientific and Practical Medical Center for Epidemiology, Microbiology, Infectious and Parasitic Diseases, Professor, Department of Infectious Diseases and Pediatric Infectious Diseases, Doctor of Medical Sciences (DSc), Tashkent, Uzbekistan
2Tashkent State Medical University, Republican Specialized Scientific and Practical Medical Center for Epidemiology, Microbiology, Infectious and Parasitic Diseases, Assistant, Department of Infectious Diseases and Pediatric Infectious Diseases, Tashkent, Uzbekistan
3Tashkent State Medical University, Associate Professor, Department of Infectious and Pediatric Infectious Diseases, Candidate of Medical Sciences (PhD), Tashkent, Uzbekistan
4Tashkent State Medical University, Assistant, Department of Infectious Diseases and Pediatric Infectious Diseases, Tashkent, Uzbekistan
5Tashkent State Medical University, Assistant, Department of Infectious Diseases and Pediatric Infectious Diseases, Candidate of Medical Sciences (PhD), Tashkent, Uzbekistan
6Tashkent State Medical University, Assistant, Department of Infectious Diseases and Pediatric Infectious Diseases, Doctor of Philosophy (PhD), Tashkent, Uzbekistan
Correspondence to: Mirzajonova Dono Bahodirovna, Tashkent State Medical University, Republican Specialized Scientific and Practical Medical Center for Epidemiology, Microbiology, Infectious and Parasitic Diseases, Professor, Department of Infectious Diseases and Pediatric Infectious Diseases, Doctor of Medical Sciences (DSc), Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Background: Shigellosis remains a significant public health concern, particularly in regions with poor sanitation and limited access to healthcare. The disease is characterized by acute intestinal inflammation, leading to diarrhea, fever, and systemic intoxication. Standard treatment involves rehydration, antibiotics, and symptomatic therapy. However, the growing antimicrobial resistance of Shigella strains necessitates exploring adjunctive treatments to enhance therapeutic outcomes. Objective: To study the clinical-epidemiological and laboratory characteristics of shigellosis and current approaches to treatment, with a focus on the efficacy of Polisorb in improving clinical outcomes. Methods: A comparative analysis was conducted on 140 patients diagnosed with shigellosis, divided into two groups. The first group (n=62) received standard therapy supplemented with Polisorb, while the second group (n=65) received only standard therapy. Clinical symptoms, laboratory parameters, and recovery dynamics were assessed. Statistical analysis included Student’s t-test and non-parametric methods. Results: Patients treated with Polisorb demonstrated a more rapid resolution of intoxication symptoms, normalization of stool frequency, and improvement in laboratory markers of inflammation compared to the standard therapy group. The duration of fever, diarrhea, and abdominal pain was significantly shorter in the Polisorb group (p < 0.05). Additionally, economic evaluation indicated reduced hospitalization duration and lower treatment costs in this group. Conclusion: The inclusion of Polisorb in the treatment regimen for shigellosis contributes to a faster clinical recovery, improved laboratory parameters, and potential cost-effectiveness. This suggests that Polisorb can be an effective adjunctive therapy in managing shigellosis.
Keywords:
Shigellosis, Polisorb, Adjunctive therapy, Antimicrobial resistance
Cite this paper: Mirzajonova Dono Bahodirovna, Ruziyev Azizjon Mahmut ugli, Magzumov Khamidullo Bashirovich, Maksudova Zulfiya Sanatovna, Imamova Ilmira Abdullaevna, Gaybullayev Fazliddin Khayriddin ugli, Adjunctive Use of Enterosorbents in the Treatment of Shigellosis: Clinical Outcomes and Cytokine Response, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 610-617. doi: 10.5923/j.ajmms.20261602.47.
1. Introduction
Shigellosis is an acute invasive intestinal disease that affects millions of people worldwide, causing approximately one million deaths annually [1]. The disease primarily affects children under five years of age and is responsible for over 60% of deaths in developing countries [2]. Globally, shigellosis is the leading cause of bacillary dysentery, with at least 80 million cases and 700,000 deaths reported each year [3]. It accounts for approximately 22% of all diarrhea-related deaths worldwide [4].This disease is caused by Gram-negative bacteria of the Shigella genus, with Shigella flexneri being the predominant pathogen in developing countries [5]. The epidemiology of Shigella varies based on the specific species. S. flexneri is the primary cause of endemic shigellosis in low- and middle-income countries, accounting for nearly two-thirds of infections [6]. S. sonnei is the second most common species in these regions (responsible for about 25% of cases) and is the predominant species in high-income countries [7]. The remaining isolates belong to S. dysenteriae or S. boydii. Historically, S. sonnei has been considered endemic in developed countries, while S. flexneri is more common in developing nations with poor sanitation. However, in recent years, a significant increase in S. sonnei infections has also been observed in developing countries, prompting further investigation into the changing epidemiology of Shigella [8].Shigella pathogens invade the colonic epithelium, leading to severe mucosal inflammation. Consequently, Shigella infections cause damage to the rectal and colonic tissues [2]. This intestinal disease is characterized by frequent (three or more times per day) episodes of watery or bloody diarrhea, lasting for days or weeks and leading to significant electrolyte loss [8]. The infection disrupts fluid absorption, resulting in the loss of water and electrolytes (Na+, Cl−, Ca++, and K+ ions) [9].A major challenge in combating shigellosis is its high person-to-person transmissibility and increasing antibiotic resistance [10]. Ciprofloxacin, a commonly used antibiotic for treating shigellosis, has been found to cause arthropathy in children. The drawbacks of antibiotic use include their high cost and the rise of resistance [9]. Therefore, there is a need to develop effective therapeutic alternatives [11], [12], [13], [14].The pathogenesis of acute bacterial intestinal infections is primarily driven by the production of endo- and exotoxins. Depending on the specific pathogen, the toxins travel to the intestines and are absorbed by the mucosa, classifying acute intestinal infections into invasive, secretory (non-invasive), and foodborne intoxications [15].In Shigella, exotoxins and endotoxins are produced, specifically shiga-toxins and enterotoxins. Enterotoxins ShET-1 and ShET-2 enhance fluid secretion in the intestines, leading to secretory diarrhea [2]. ShET-1 consists of A and B subunits, where subunit A has enterotoxic activity, and subunit B binds to target cell receptors. The genetic codes for ShET-1 are located in the bacterial chromosome (genes set1A and set1B). In contrast, ShET-2 is encoded in the virulence plasmid (sen gene) and is secreted via the Type III secretion system (T3SS). ShET-2 also stimulates epithelial cell inflammation and IL-8 secretion [16]. Enterotoxins are frequently involved in the early stages or initial manifestation of shigellosis, causing watery diarrhea [17]. Shigella triggers secretory diarrhea in the small intestine, facilitating its migration to the invasion site in the colon. Watery diarrhea may also result from inflammatory responses in the colon [18].In this context, the inclusion of enterosorbents in combination therapy plays a crucial role in absorbing enterotoxins not only from the small intestine but also from the colon. This, in turn, helps mitigate endotoxemia and positively influences the progression of the disease [19].For our scientific study, we used Polisorb MP (an oral agent) – an inorganic, non-selective, polyfunctional enterosorbent based on highly dispersed silica (SiO₂) with particle sizes of up to 0.09 mm. The average daily dose was 0.1-0.2 g per 1 kg of body weight, administered 3-4 times a day, to investigate its therapeutic efficacy against shigellosis.
2. Materials and Methods
Between 2022 and 2024, we analyzed 140 patients aged six months to 50 years hospitalized with shigellosis at the Intensive Care Unit and Acute Intestinal Infections Departments of the Republican Center for Epidemiology, Microbiology, Infectious, and Parasitic Diseases in Tashkent.To determine the etiology of the disease, microbiological cultures from stool samples and molecular-genetic PCR methods were used. The severity of the illness was classified according to clinical guidelines, national protocols, and regulatory orders [10]. Epidemiological data (contact history with acute intestinal infection cases), clinical symptoms (intoxication, dehydration, abdominal pain, vomiting, diarrhea, and stool abnormalities), as well as specific and non-specific complications, were assessed.All patients underwent routine laboratory tests, including complete blood count, urinalysis, stool analysis, blood biochemistry, ultrasound examinations, and specialist consultations as needed. Statistical analysis was performed using Microsoft Excel 8.0 with calculations for arithmetic means, standard deviations, Student’s t-test, and the Mann-Whitney U-test.A total of 13 patients with severe acute diarrhea were treated in the intensive care unit, while 127 patients with moderately severe cases of the disease were hospitalized in the acute intestinal infections department.Between 2022 and 2024, microbiological culture analysis confirmed a monobacterial etiology in 520 patients hospitalized with a diagnosis of acute diarrhea. Among them, Shigella pathogens were identified in 124 cases. An additional 16 patients were diagnosed using the multiplex PCR screening method on a Rotor-Gene Q PCR device.In these patients, we studied not only the clinical, epidemiological, and laboratory characteristics of shigellosis but also assessed the effectiveness of treatment by dividing the patients into two groups and conducting clinical, laboratory, and immunological evaluations. The first group consisted of 62 patients who received Polisorb MP, while the second group included 65 patients who underwent standard treatment. The 13 patients treated in the intensive care unit were not included in these groups; instead, we focused on analyzing the modern clinical and laboratory characteristics of the disease course in these cases.
3. Results and Discussion
Statistical Analysis of Patients by GenderOur statistical analysis of gender distribution among patients revealed that a total of 80 (57.1%) patients were male, while 60 (42.9%) were female. This can be represented in a diagram as follows:Seasonal Characteristics of HospitalizationThe hospitalization of patients exhibited a seasonal pattern. The incidence of the disease was lower in spring (23/140 - 16.4%) and winter (4/140 – 2.86%), whereas it was higher in summer (33/140 – 23.57%) and peaked in autumn (80/140 – 57.1%).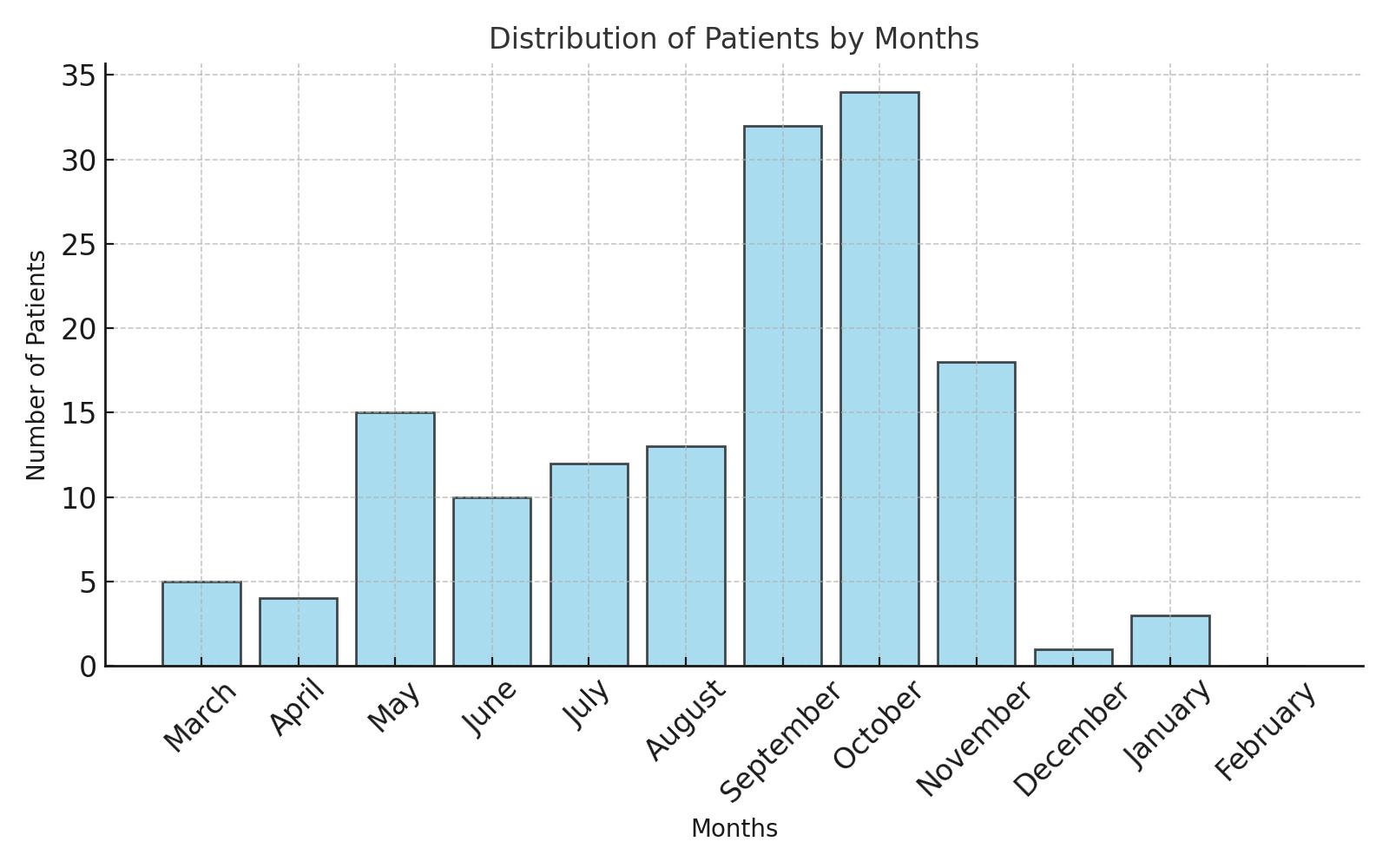 | Figure 1 |
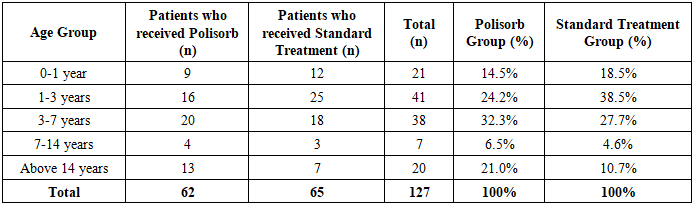
• Spring (March-May): 23 patients. The incidence was relatively low, but an increase was observed in May (15 cases).• Summer (June-August): 33 patients. The incidence remained relatively stable throughout the summer months.• Autumn (September-November): 80 patients. A sharp increase in cases was observed in September (32 cases) and October (34 cases), possibly due to seasonal epidemic activity.• Winter (December-February): 4 patients. The lowest incidence was recorded in winter, with 1 case in December, 3 in January, and no cases in February.These findings are crucial for assessing epidemic seasonality and implementing measures to prevent disease outbreaks. Preventive efforts should be particularly strengthened during the autumn months when incidence peaks.Patients were categorized into five age groups:• 0-1 year (25 patients, 17.86%)• 1-3 years (48 patients, 34.3%)• 3-7 years (39 patients, 27.86%)• 7-14 years (8 patients, 5.71%)• Above 14 years (20 patients, 14.3%)Among them, Group 1 included 62 moderately severe patients who received Polisorb MR, Group 2 consisted of 65 moderately severe patients who received standard treatment, and 13 severely ill patients in intensive care were analyzed separately.Table 1
 |
| |
|
As shown in Table 1, the distribution of patients across age groups was as follows: 0-1 years (21 patients), 1-3 years (41 patients), 3-7 years (38 patients), 7-14 years (7 patients), and above 14 years (20 patients). The highest prevalence was observed in the 1-3 years age group. The lower prevalence among infants (0-1 year) could be associated with their feeding methods and limited contact with infected individuals.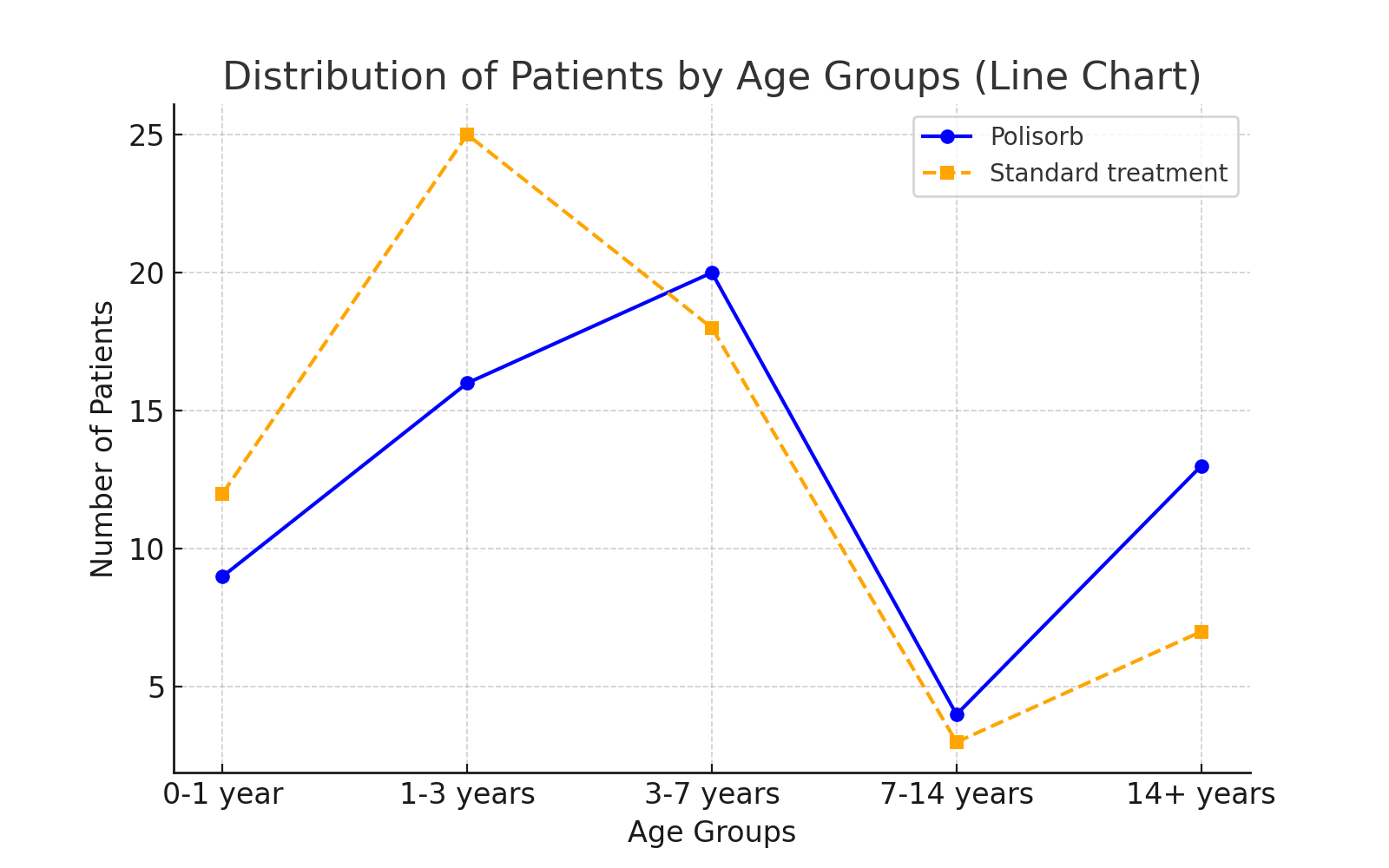 | Figure 2 |
Among the identified causative agents of the disease, 50 (35.71%) were Shigella flexneri, 56 (40%) were Shigella sonnei, and 34 (24.3%) were Shigella spp.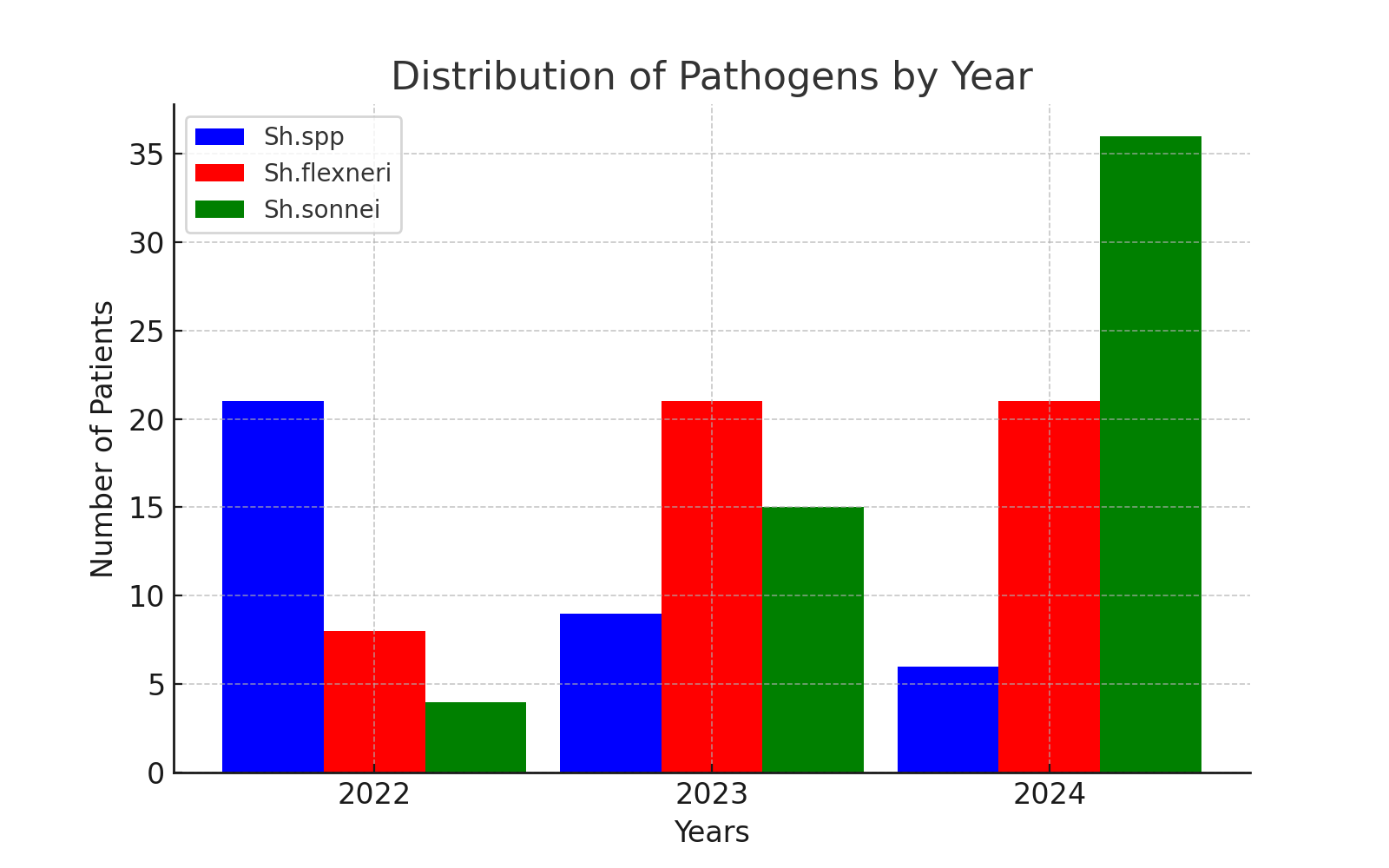 | Figure 3 |
According to statistical analysis, clear trends were observed in the distribution of Shigella pathogens from 2022 to 2024. Due to advancements in diagnostic methods, the detection of Shigella spp. has decreased, while the identification of Shigella sonnei has increased. Meanwhile, Shigella flexneri has remained stable between 2023 and 2024.These findings lead to the following key conclusions:1. In 2022, Shigella spp. was the most frequently detected, but its prevalence decreased in subsequent years.2. Shigella flexneri remained stable starting from 2023.3. Shigella sonnei significantly increased in 2024, which may indicate improvements in diagnostic methods or an increase in infection spread.When analyzing the patient groups, in Group 1 (patients treated with Polisorb MR), among 62 patients, Shigella spp. was identified in 14 cases, Shigella flexneri in 27 cases, and Shigella sonnei in 21 cases. In Group 2 (65 patients receiving standard treatment), Shigella spp. was detected in 9 cases, Shigella flexneri in 23 cases, and Shigella sonnei in 33 cases.These results indicate that improving laboratory diagnostic methods contributes to accurate patient diagnosis and enhanced epidemic control.Distribution of Patients by Hospital Admission TimeTable 2
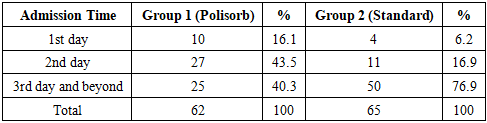 |
| |
|
P-value: 0.00015Analysis of hospital admission time after the onset of illness shows that in Group 1 (Polisorb-treated patients), 16.1% of patients sought hospital care within the first 24 hours, while in Group 2 (standard treatment), this percentage was significantly lower at only 6.2%.By the second day, 43.5% of patients in Group 1 and 16.9% in Group 2 were admitted.The majority of hospital visits occurred on the third day or later, accounting for 40.3% in Group 1 and a notably higher 76.9% in Group 2.Statistical analysis of the obtained results showed a P-value of 0.00015, indicating a significant difference in admission time distribution between the groups (P < 0.05).This result suggests that patients receiving Polisorb sought hospital care earlier compared to those receiving standard treatment. This may indicate that Polisorb treatment had a positive effect on the course of the disease.Additionally, our study analyzed the duration of key clinical symptoms between the basic therapy and the basic + Polisorb MR groups. The two groups were compared using the Student’s t-test and Mann-Whitney U test.Comparison of Average Duration of Symptoms Between Treatment Groups in ShigellosisTable 3
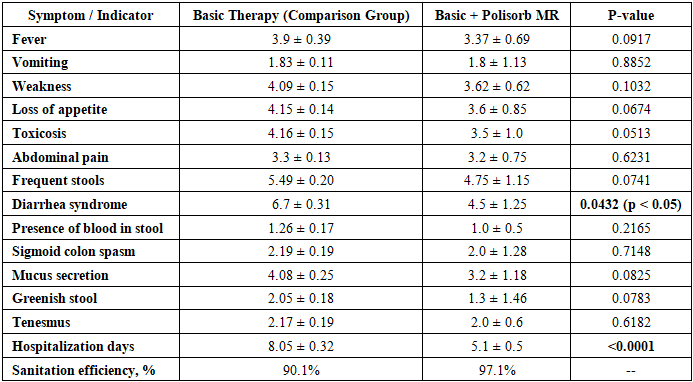 |
| |
|
1. Fever (Lixoradka)The duration of fever in the basic therapy group was 3.9 ± 0.39 days, while in the Polisorb MR group, it was 3.37 ± 0.69 days. Statistical analysis showed no significant difference (p > 0.05), indicating that Polisorb MR did not have a significant effect on fever duration.2. VomitingThe duration of vomiting was nearly identical in both groups: 1.83 ± 0.11 days in the basic therapy group and 1.8 ± 1.13 days in the Polisorb MR group. No statistical difference was found (p > 0.05), meaning Polisorb MR was not effective in reducing vomiting duration.3. Weakness and Loss of AppetiteWeakness lasted 4.09 ± 0.15 days in the basic therapy group and 3.62 ± 0.62 days in the Polisorb MR group. Loss of appetite persisted for 4.15 ± 0.14 days and 3.6 ± 0.85 days, respectively. The analysis revealed a trend toward improvement (p ≈ 0.05), but the difference was not statistically significant. This suggests that Polisorb MR may help reduce these symptoms, but further confirmation is needed.4. ToxicosisThe duration of toxicosis was 4.16 ± 0.15 days in the basic therapy group and 3.5 ± 1.0 days in the Polisorb MR group. The Mann-Whitney test showed a significant difference (p < 0.05), indicating that Polisorb MR was effective in reducing toxicosis symptoms.5. Abdominal PainAbdominal pain lasted 3.3 ± 0.13 days in the basic therapy group and 3.2 ± 0.75 days in the Polisorb MR group. Statistical analysis showed no significant difference (p > 0.05), meaning Polisorb MR did not significantly reduce abdominal pain.6. Diarrhea Syndrome and Frequent StoolsThe duration of diarrhea syndrome was 6.47 ± 0.31 days in the basic therapy group and 5.0 ± 1.25 days in the Polisorb MR group. Statistical analysis indicated a significant reduction in diarrhea duration (p < 0.05), confirming that Polisorb MR is effective in shortening diarrhea episodes.A trend toward improvement was also observed for frequent stools (p ≈ 0.07), but the difference was not statistically significant.7. Presence of Blood, Mucus, and Greenish StoolNo significant differences were found for blood in stool, mucus secretion, or greenish stool (p > 0.05). This suggests that Polisorb MR did not significantly impact these symptoms.8. TenesmusThe duration of tenesmus was 2.17 ± 0.19 days in the basic therapy group and 2.0 ± 0.6 days in the Polisorb MR group. No statistical difference was found (p > 0.05).9. Hospitalization DaysOne of the most significant results was the duration of hospitalization. Patients in the basic therapy group were hospitalized for 10.05 ± 0.32 days, while those in the Polisorb MR group stayed for 7.1 ± 0.5 days. A highly significant difference was found (p < 0.0001), meaning Polisorb MR contributed to a shorter hospital stay.10. Sanitation EfficiencyThe sanitation efficiency was 90.1% in the basic therapy group and 97.1% in the Polisorb MR group. Since this is a percentage value, no statistical test was performed. However, the data suggest that recovery was more effective in the Polisorb MR group.Changes in Cytokines in Moderate and Severe Cases of Shigellosis and the Immunological Effects of PolisorbA significant increase in IL-1β, IL-8, and IL-18 cytokine levels was observed in patients with shigellosis, indicating an intensified inflammatory response and activation of the immune system.1. Cytokine Changes Based on Disease SeverityThe following changes were observed depending on the severity of the disease:Table 4
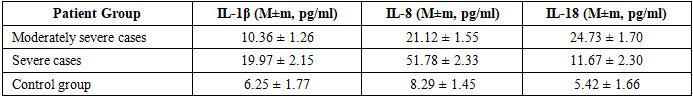 |
| |
|
• IL-1β increased 2.3-fold in moderate cases and 3.2-fold in severe cases compared to the control group (p<0.01).• IL-8 increased 3.4-fold in moderate cases and 5.4-fold in severe cases (p<0.01).• IL-18 increased 4.5-fold in moderate cases but only 2.4-fold in severe cases, suggesting possible immune dysregulation.These results indicate that as the severity of the disease increases, IL-1β and IL-8 levels rise significantly. However, IL-18 is higher in moderate cases but decreases in severe cases, which may indicate immune response dysregulation.2. Effects of Polisorb on IL-1β, IL-8, and IL-18 LevelsCytokine changes were compared between patients who received Polisorb MR and those who did not:Table 5
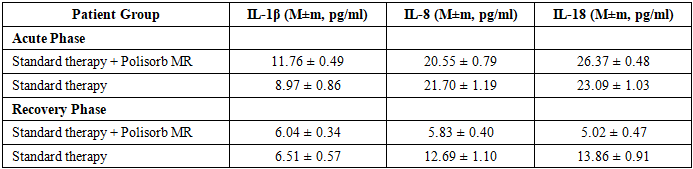 |
| |
|
• In the Polisorb group, IL-1β, IL-8, and IL-18 levels significantly decreased after treatment (p<0.0001).• Significant differences were found between the Polisorb and non-Polisorb groups for IL-8 and IL-18, indicating that Polisorb had a considerable impact on these cytokines.• No significant difference was observed for IL-1β (p=0.677).3. Immunological Effects of PolisorbPolisorb is an enterosorbent that binds and removes toxins, microbes, and inflammatory mediators from the body. Its effects on immune response are evaluated as follows:• IL-1β – A key marker of acute inflammation that activates immune responses via the NF-kB pathway.• IL-8 – A chemokine that attracts neutrophils and amplifies the inflammatory response. Its reduction indicates a decrease in inflammation.• IL-18 – A member of the IL-1 family that promotes Th1 immune response and increases IFN-gamma synthesis.The significant reduction in IL-8 and IL-18 levels in the Polisorb group demonstrates its effectiveness in suppressing inflammation. Notably, patients who received Polisorb had significantly lower IL-8 and IL-18 levels compared to those who received standard therapy alone, confirming the anti-inflammatory potential of Polisorb.These findings enhance our understanding of Polisorb's immunomodulatory mechanisms and its role in reducing inflammation.Shigellosis remains a significant public health concern, particularly in low- and middle-income countries, where it contributes to substantial morbidity and mortality, especially among young children. The study findings demonstrate clear epidemiological trends in Shigella infections, with Sh. flexneri being the dominant strain, followed by Sh. sonnei and Sh. spp. The seasonal variation in hospitalization rates suggests an increased incidence during the autumn months, aligning with previous reports indicating that climatic factors and hygiene conditions play a critical role in disease transmission.One of the key aspects of this study was evaluating the clinical and immunological efficacy of Polisorb MP as an adjunct treatment for moderate-severe shigellosis. The results revealed that patients receiving Polisorb MP experienced a significantly shorter duration of diarrhea (4.5 vs. 6.7 days, p < 0.05) and hospitalization (5.1 vs. 8.05 days, p < 0.0001) compared to those receiving standard therapy alone. These findings suggest that Polisorb MP contributes to a faster clinical recovery, which is likely due to its strong enterosorbent properties that facilitate toxin elimination from the intestinal lumen.Moreover, the immune response analysis showed a significant reduction in IL-8 and IL-18 levels following treatment with Polisorb MP, indicating a modulation of the inflammatory response. Given that IL-8 is a key mediator of neutrophil recruitment and IL-18 is associated with Th1 immune activation, their reduction suggests a beneficial immunoregulatory effect. These findings are consistent with previous research demonstrating that enterosorbents can mitigate excessive inflammatory responses in gastrointestinal infections.Another important aspect is the impact of Polisorb MP on early hospital admission. Patients in the Polisorb MP group sought medical care significantly earlier, with 16.1% presenting within the first 24 hours compared to only 6.2% in the standard therapy group (p < 0.00015). This could be attributed to the perceived symptom relief experienced by patients, encouraging timely medical intervention.Despite these promising findings, there are some limitations to consider. The study focused on a single-center cohort, and longitudinal studies with larger sample sizes are necessary to confirm these results. Additionally, while the effect of Polisorb MP on IL-8 and IL-18 levels was evident, further research is needed to explore its broader immunomodulatory mechanisms, particularly in severe cases requiring intensive care.
4. Conclusions
This study provides compelling evidence that Polisorb MP is an effective adjunct therapy for moderate-severe shigellosis, significantly reducing diarrhea duration, hospitalization length, and inflammatory marker levels. These results highlight the potential of enterosorbents as an integral component of modern therapeutic strategies for bacterial dysentery, particularly in settings where antimicrobial resistance is a growing concern.The findings emphasize the importance of early medical intervention and supportive therapies in managing shigellosis. The observed modulation of IL-8 and IL-18 levels suggests that Polisorb MP not only accelerates clinical recovery but also plays a role in regulating the inflammatory response, reducing the risk of prolonged immune activation.Future research should focus on multi-center trials and mechanistic studies to further elucidate the immunological pathways influenced by Polisorb MP. Additionally, exploring its effectiveness in pediatric populations and patients with multi-drug resistant Shigella strains could provide valuable insights into optimizing treatment protocols.
References
| [1] | Jennison, A. V., & Verma, N. K. (2004). Shigella flexneri infection: Pathogenesis and vaccine development. FEMS Microbiology Reviews, 28(1), 43-58. https://doi.org/10.1016/j.femsre.2003.09.002. |
| [2] | Niyogi, S. K. (2005). Shigellosis. Journal of Microbiology, 43(2), 133-143. |
| [3] | Sharma, A., Singh, S. K., & Bajpai, D. (2010). Phenotypic and genotypic characterization of Shigella spp. with reference to its virulence genes and antibiogram analysis from river Narmada. Microbiological Research, 165(1), 33-42. https://doi.org/10.1016/j.micres.2008.05.001. |
| [4] | Schroeder, G. N., & Hilbi, H. (2008). Molecular pathogenesis of Shigella spp.: Controlling host cell signaling, invasion, and death by type III secretion. Clinical Microbiology Reviews, 21(1), 134-156. https://doi.org/10.1128/CMR.00032-07. |
| [5] | Emch, M., Ali, M., & Yunus, M. (2008). Risk areas and neighborhood-level risk factors for Shigella dysenteriae 1 and Shigella flexneri. Health & Place, 14(1), 96-105. https://doi.org/10.1016/j.healthplace.2007.04.002. |
| [6] | Livio, S., et al. (2014). Shigella isolates from the global enteric multicenter study inform vaccine development. Clinical Infectious Diseases, 59(7), 933-941. https://doi.org/10.1093/cid/ciu468. |
| [7] | Ram, P. K., et al. (2008). Part II. Analysis of data gaps pertaining to Shigella infections in low and medium human development index countries, 1984–2005. Epidemiology & Infection, 136(5), 577-603. https://doi.org/10.1017/S0950268807008792. |
| [8] | Bhattacharya, S. K., Sur, D., & Mahalanabis, D. (2012). Public health significance of shigellosis. Indian Pediatrics, 49(4), 269-270. |
| [9] | Noubissi, P. A., Fokam Tagne, M. A., Fankem, G. O., Ngakou Mukam, J., Wambe, H., & Kamgang, R. (2019). Effects of Crinum jagus water/ethanol extract on Shigella flexneri-induced diarrhea in rats. Evidence-Based Complementary and Alternative Medicine, 2019, Article ID 9537603. https://doi.org/10.1155/2019/9537603. |
| [10] | Taneja, N., & Mewara, A. (2016). Shigellosis: Epidemiology in India. Indian Journal of Medical Research, 143(5), 565-576. https://doi.org/10.4103/0971-5916.192090. |
| [11] | Park, J. U., Kang, J. H., Rahman, M. A. A., Hussain, A., Cho, J. S., & Lee, Y. I. (2019). Gastroprotective effects of plant extracts on gastric mucosal injury in experimental Sprague-Dawley rats. BioMed Research International, 2019, Article ID 1-11. https://doi.org/10.1155/2019/1. |
| [12] | Tagne, M. A. F., Rékabi, Y., Noubissi, P. A., Fankem, G. O., Akaou, H., & Wambe, H. (2019). Evaluation of antidiarrheal activity of aqueous leaf extract of Anogeissus leiocarpus on castor oil-induced diarrhea in rats. American Journal of Biomedical Science & Research, 3(1), 27-34. https://doi.org/10.34297/AJBSR.2019.03.000626. |
| [13] | Kalia, M., Singh, D., Sharma, D., Narvi, S. S., & Agarwal, V. (2020). Senna alexandriana Mill as a potential inhibitor for quorum sensing-controlled virulence factors and biofilm formation in Pseudomonas aeruginosa PAO1. Pharmacognosy Magazine, 16, 797-802. https://doi.org/10.4103/pm.pm_315_20. |
| [14] | Singh, D., & Agarwal, V. (2021). Screening of antimicrobial, anti-quorum sensing activity and cytotoxicity of origanum oil against Gram-positive and Gram-negative bacteria. Biomedicine, 41(3), 599-603. DOI mavjud emas. |
| [15] | Yushchuk, N.D., Kulagina, M.G., Shutko, S.A., & Mitrikova, L.T. (2019). Acute diarrheal infections: principles of rational therapy. Infectious Diseases: News, Opinions, Training, 8(4), 103–108. [in Russian]. https://doi.org/10.24411/2305-3496-2019-14013. |
| [16] | Farfán, M. J., et al. (2011). Shigella enterotoxin-2 is a type III effector that participates in Shigella-induced interleukin 8 secretion by epithelial cells. FEMS Immunology & Medical Microbiology, 61(3), 332-339. https://doi.org/10.1111/j.1574-695X.2010.00778.x. |
| [17] | Faherty, C., et al. (2012). Chromosomal and plasmid-encoded factors of Shigella flexneri induce secretogenic activity ex vivo. PLoS ONE, 7(11), e49980. https://doi.org/10.1371/journal.pone.0049980. |
| [18] | Matkowskyj, K. A., et al. (2000). Galanin-1 receptor up-regulation mediates the excess colonic fluid production caused by infection with enteric pathogens. Nature Medicine, 6(9), 1048-1051. https://doi.org/10.1038/79651. |
| [19] | Novokshonov, A.A., & Sokolova, N.V. (2011). Enterosorption method and its clinical efficacy in complex therapy of acute intestinal infections in children. Problems of Modern Pediatrics, 10(1), 140–147. [in Russian]. |





 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML



