-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 604-609
doi:10.5923/j.ajmms.20261602.46
Received: Jan. 12, 2026; Accepted: Feb. 6, 2026; Published: Feb. 11, 2026

Use of the Drug "Enterol" in the Comprehensive Treatment of Chronic Viral Hepatitis
Donokhon B. Mirzajonova1, Shakhnosa A. Tashpulatova2, Gulrukh Y. Sultonova3, Ikrom A. Artikov4, Fazliddin I. Kamalov5, Nilufar F. Chulieva6
1Doctor of Medical Sciences, Associate Professor of the Department of Infectious and children’s Infectious Diseases, Tashkent State Medical University, Tashkent, Uzbekistan
2Candidate of Medical Sciences, Associate Professor of the Department of Infectious and Children’s Infectious Diseases, Tashkent State Medical University, Tashkent, Uzbekistan
3PhD, Senior Lecturer of the Department of Infectious and children’s Infectious Diseases, Tashkent State Medical University, Tashkent, Uzbekistan
4Assistant Lecturer at the Department of Infectious Diseases, Urgench Medical Institute
5Head of Department, Koson District Infectious Diseases Hospital, Kashkadarya Region, Republic of Uzbekistan
6Infection doctor at the Tashkent Regional Infectious Diseases Hospital
Correspondence to: Donokhon B. Mirzajonova, Doctor of Medical Sciences, Associate Professor of the Department of Infectious and children’s Infectious Diseases, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Disturbances of the intestinal microbiota are increasingly recognized as an important factor in the persistence of inflammation and progression of chronic viral hepatitis. Modulation of gut microbiota may represent a promising pathogenetic approach. Objective: To assess alterations of the intestinal microbiota in patients with chronic hepatitis B and C and to evaluate the clinical and laboratory effects of adjunctive probiotic therapy containing Saccharomyces boulardii. Methods: This randomized controlled study included 70 patients with chronic viral hepatitis (40 with hepatitis B and 30 with hepatitis C). Patients were randomly assigned to two groups: the probiotic group (n=40), receiving standard therapy plus Saccharomyces boulardii at a dose of 250 mg/day for 30 days, and the control group (n = 30), receiving standard therapy alone. Clinical symptoms, liver biochemical parameters (ALT, AST), and bacteriological stool analyses were assessed before and after treatment. Statistical analysis was performed using standard methods, with results considered significant at p < 0.05. Results: Intestinal dysbiosis was detected in all patients and was significantly associated with disease duration and severity of clinical manifestations (p < 0.05). Patients receiving probiotic therapy demonstrated a significant improvement in intestinal microbiota composition, characterized by an increase in beneficial microorganisms and a reduction in opportunistic flora compared with the control group (p < 0.05). Probiotic supplementation was associated with faster regression of gastrointestinal and systemic symptoms and a significant normalization of liver enzymes, including ALT and AST levels (p < 0.05). The therapy was well tolerated, and no adverse events were reported. Conclusions: Intestinal dysbiosis is a common finding in chronic viral hepatitis and correlates with disease duration and clinical severity. Adjunctive probiotic therapy containing Saccharomyces boulardii improves gut microbiota balance, contributes to the normalization of liver biochemical markers, and enhances clinical outcomes, supporting its role as an effective component of pathogenetic treatment in patients with chronic hepatitis B and C.
Keywords: Chronic viral hepatitis, Gut microbiota, Intestinal dysbiosis, Probiotics, Saccharomyces boulardii, Hepatitis B, Hepatitis C
Cite this paper: Donokhon B. Mirzajonova, Shakhnosa A. Tashpulatova, Gulrukh Y. Sultonova, Ikrom A. Artikov, Fazliddin I. Kamalov, Nilufar F. Chulieva, Use of the Drug "Enterol" in the Comprehensive Treatment of Chronic Viral Hepatitis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 604-609. doi: 10.5923/j.ajmms.20261602.46.
1. Introduction
- In recent years, increasing attention has been directed toward identifying risk factors that contribute to the progression and destabilization of chronic viral hepatitis, as well as toward the development of novel pathogenetic treatment strategies. The progression of chronic viral hepatitis is driven by a complex interplay of pathophysiological mechanisms, among which hepatocyte necrosis and progressive liver fibrosis play a central role [2,4].Recent clinical and experimental studies have demonstrated that intestinal dysbiosis is an important contributor to the persistence of hepatic inflammation in chronic liver diseases. Alterations in the composition and function of the intestinal microbiota are now recognized as a key element of the gut–liver axis, a bidirectional system linking the intestine and the liver through portal circulation, immune signaling, and metabolic pathways [2].The normal intestinal microbiota represents a complex ecological system that performs essential metabolic, immune, and barrier functions. Various endogenous and exogenous factors, including chronic inflammation, antiviral therapy, and impaired bile secretion, may lead to quantitative and qualitative disturbances of this system, resulting in intestinal dysbiosis [1,5].Disruption of the intestinal barrier integrity in dysbiosis increases intestinal permeability and promotes the translocation of bacteria and bacterial products, such as endotoxins, into the portal circulation. This process activates hepatic immune cells, enhances pro-inflammatory cytokine production, and contributes to hepatocellular injury and fibrosis progression. Excessive growth of opportunistic microorganisms further amplifies inflammatory and necrotic processes in the liver, aggravating disease severity [1,6,7].Persistent endotoxemia and systemic inflammation associated with intestinal dysbiosis also play a role in the development of minimal hepatic encephalopathy. Neurotoxic substances, including ammonia and inflammatory mediators derived from intestinal bacteria, impair neurotransmission and cerebral function, leading to cognitive dysfunction and reduced quality of life in patients with chronic viral hepatitis.The relevance of chronic viral hepatitis as a public health problem in the Republic of Uzbekistan is determined by its high prevalence and the heterogeneous circulation of hepatotropic viruses across different regions, influenced by medico-social conditions and lifestyle factors. Despite the availability of modern antiviral agents, the effectiveness of etiological therapy remains limited due to high treatment costs, contraindications, and the risk of adverse effects, underscoring the need for effective adjunctive pathogenetic approaches.In this context, the role of pathogenetic therapy in chronic viral hepatitis aimed at reducing inflammatory activity, protecting hepatocytes, and correcting intestinal dysbiosis becomes increasingly important. Given the central involvement of the gut–liver axis in disease progression, modulation of the intestinal microbiota represents a rational therapeutic target. The inclusion of probiotic preparations in the comprehensive management of patients with chronic viral hepatitis therefore appears promising.Probiotics containing live microorganisms and their biologically active metabolites promote the restoration of intestinal microbial balance, enhance intestinal barrier function, and reduce bacterial translocation and endotoxemia. Through the attenuation of systemic and hepatic inflammatory responses, probiotic therapy may contribute to improved liver function and a more favorable clinical course of chronic viral hepatitis.To evaluate the efficacy of adjunctive probiotic therapy containing Saccharomyces boulardii in patients with chronic hepatitis B and C by assessing its effects on intestinal dysbiosis and liver biochemical activity.The primary endpoints were changes in intestinal microbiota composition, assessed by bacteriological stool analysis, and normalization of liver biochemical parameters, including alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels. The secondary endpoints included regression of gastrointestinal and systemic clinical symptoms and changes in the severity of intestinal dysbiosis. The efficacy of probiotic therapy was operationally defined as statistically significant improvement in microbiological, biochemical, and clinical parameters compared with standard therapy alone.This study was designed as a randomized controlled clinical trial. Eligible patients were randomly allocated into two groups using a simple randomization method. The probiotic group received standard therapy in combination with Saccharomyces boulardii, while the control group received standard therapy alone.Due to the nature of the intervention, blinding of patients and treating physicians was not performed; however, laboratory assessments of biochemical and microbiological parameters were conducted using standardized methods to minimize measurement bias.
2. Materials and Methods
- This study was designed as a prospective randomized controlled clinical trial. A total of 70 patients with chronic viral hepatitis were enrolled and followed during the study period. Chronic viral hepatitis was defined as persistent hepatitis B or C infection with a disease duration of at least 6 months, confirmed by clinical, serological, and molecular criteria.Inclusion criteria were: age 18–60 years. Confirmed diagnosis of chronic hepatitis B or chronic hepatitis C. Presence of biochemical activity (elevated ALT and/or AST levels). Detectable HBV DNA or HCV RNA by PCR. Signed informed consent. Exclusion criteria included: Mixed HBV/HCV infection or co-infection with HIV. Decompensated liver cirrhosis. Severe concomitant somatic diseases (cardiovascular, renal, autoimmune disorders). Alcohol abuse. Use of antibiotics, probiotics, or prebiotics within 4 weeks prior to enrollment. Pregnancy or lactation. Patients receiving antiviral therapy were included only if the treatment regimen had remained unchanged for at least 3 months before enrollment.Eligible patients were randomly assigned to two groups using a simple randomization method in a 1:1 ratio. Allocation concealment was ensured by assigning patients sequentially according to a randomization list. Due to the nature of the intervention, blinding of patients and treating physicians was not performed; however, laboratory and instrumental assessments were conducted by specialists blinded to treatment allocation.The diagnosis of chronic viral hepatitis was established based on medical history, clinical examination, and laboratory parameters, including total protein, total bilirubin, ALT, AST, creatinine, urea, and prothrombin index. Serological markers of viral hepatitis (HBsAg, anti-HBs, anti-HCV) were determined by enzyme-linked immunosorbent assay (ELISA) using certified commercial test systems. Molecular genetic testing was performed using polymerase chain reaction (PCR) to detect HBV DNA and HCV RNA in blood samples with the Rotor-Gene Q system (QIAGEN, Germany). Viral load thresholds corresponding to active infection were defined according to manufacturer recommendations and national clinical guidelines.Abdominal ultrasound examination was performed in all patients. Liver fibrosis was assessed using transient elastography (FibroScan®, Echosens). Fibrosis stages were interpreted according to standard cut-off values (F0–F4). Elastography measurements were performed by trained operators using standardized protocols, with at least ten valid measurements obtained per patient.To evaluate qualitative and quantitative changes in the intestinal microbiota, bacteriological stool examination for intestinal dysbiosis was performed in accordance with approved laboratory diagnostic standards. The analysis included the assessment of major microbial groups, including bifidobacteria, lactobacilli, Escherichia coli (typical and atypical forms), enterococci, opportunistic enterobacteria, and yeast-like fungi.The severity of intestinal dysbiosis was graded based on deviations from reference values and categorized as mild, moderate, or severe. Quality control procedures were applied throughout microbiological analysis. Although stool culture methods primarily detect cultivable microorganisms and do not reflect total microbial diversity, this approach was considered appropriate for routine clinical assessment. All patients received standard basic and pathogenetic therapy according to national clinical guidelines, including hepatoprotective and symptomatic treatment. Patients in the main group additionally received the probiotic preparation Enterol® (Saccharomyces boulardii) at a dose of 250 mg per day for 30 days. The control group received standard therapy without probiotic supplementation. The primary outcome measures were changes in ALT and AST levels and improvement in intestinal dysbiosis severity. Secondary outcomes included regression of gastrointestinal and systemic symptoms and overall clinical course of the disease. Patients were evaluated at baseline and after completion of the treatment course. Treatment efficacy was assessed based on the dynamics of clinical symptoms, laboratory parameters, and intestinal microbiota during the follow-up period.Statistical analysis was performed using standard statistical methods. Quantitative data were expressed as mean ± standard deviation. Differences between groups were assessed using appropriate parametric or non-parametric tests. Results were considered statistically significant at p < 0.05. Missing data were handled by complete case analysis.
3. Results
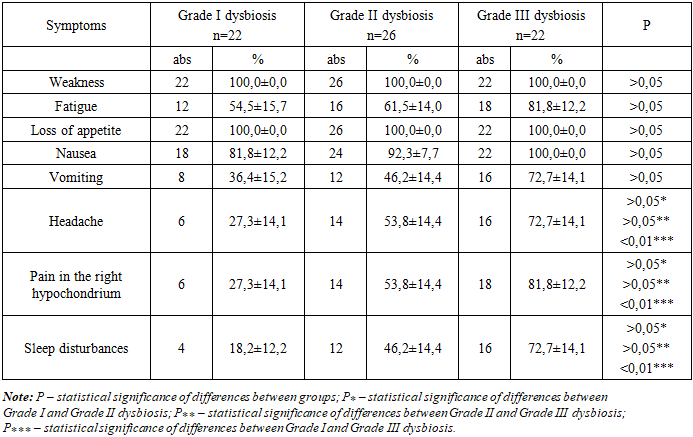
- The study included 70 patients with chronic viral hepatitis, of whom 40 (57.1%) had chronic hepatitis B (CHB) and 30 (42.9%) had chronic hepatitis C (CHC). The mean age of patients was 38 ± 9.7 years. Baseline demographic and clinical characteristics, including viral load, fibrosis stage, and biochemical parameters, were comparable between the two groups (p > 0.05).Intestinal dysbiosis was detected in all patients regardless of viral etiology, although the pattern and severity of microbial alterations varied. Bacteriological analysis revealed reduced levels of beneficial bacteria, particularly bifidobacteria, lactobacilli, and Escherichia coli with normal enzymatic activity. Overgrowth of opportunistic microorganisms was identified in 20 patients (28.6%), predominantly E. coli strains with altered enzymatic activity, Proteus spp., and Klebsiella spp. The presence of Staphylococcus aureus and streptococcal flora was observed in four cases each. Fungal overgrowth (Candida spp.) was detected in 41 stool samples (60%), with no significant difference between CHB and CHC patients.It should be noted that the absence of a healthy control group, as well as missing data on prior antibiotic or proton pump inhibitor use, diet, and comorbidities, represent potential confounding factors. Consequently, while these findings are consistent with intestinal dysbiosis in chronic viral hepatitis, it cannot be conclusively determined that these microbial alterations are solely attributable to the underlying liver disease.Intestinal dysbiosis was graded as follows: grade I in 24 patients (34.2%), grade II in 34 patients (48.5%), and grade III in 14 patients (20.0%). The distribution of dysbiosis grades did not differ significantly between CHB and CHC groups (p > 0.05, chi-square test), suggesting that dysbiosis severity may be independent of viral etiology.Analysis by disease duration revealed a trend toward increased dysbiosis severity with longer chronic viral hepatitis. Among patients with disease duration >10 years (n = 16, mean 13.1 ± 0.5 years), grade III dysbiosis was observed in 10 patients (62.5 ± 18.2%) and grade II in 6 patients (37.5 ± 18.2%). Patients with disease duration of 6–10 years (n = 30, mean 8.13 ± 0.4 years) exhibited grade II dysbiosis in 60.0 ± 13.5% of cases, grade I in 33.3 ± 12.6%, and grade III in 6.7 ± 6.7%. In patients with disease duration <5 years (n = 24, mean 3.6 ± 0.4 years), grade I dysbiosis was significantly more frequent (58.3 ± 14.8%) compared with longer disease duration (p < 0.05, chi-square test), whereas grade II dysbiosis was observed in 41.7 ± 14.8%.Spearman correlation analysis demonstrated a moderate positive association between disease duration and dysbiosis grade (r = 0.46, p < 0.01). Regression analysis adjusting for age and viral etiology indicated a similar trend, although statistical significance was not reached, suggesting potential confounding. These results indicate a descriptive association rather than a causal relationship, with small subgroup sizes potentially contributing to type II error.The frequency of clinical manifestations of chronic viral hepatitis, including gastrointestinal and systemic symptoms, tended to increase with worsening dysbiosis. For instance, general weakness increased numerically across dysbiosis grades but did not reach statistical significance (p > 0.05, chi-square test), indicating appropriate restraint in interpretation. Overall, these trends support the concept of a gut–liver interaction, where intestinal dysbiosis may contribute to the severity of clinical symptoms (Table 1).
|
4. Discussion
- The results of the present study confirm the significant role of intestinal microbiota disturbances in the pathogenesis of chronic viral hepatitis. Dysbiosis was observed in all patients, irrespective of viral etiology, and the severity of microbial imbalance correlated with the duration of disease. These findings underscore the involvement of the gut–liver axis in the maintenance of chronic hepatic inflammation. Alterations in the intestinal microbiota can lead to increased bacterial translocation, endotoxemia, and activation of systemic inflammatory responses, thereby contributing to hepatocellular injury and the progression of fibrosis. This mechanistic link supports the concept that the gut microbiome is not merely a bystander but an active participant in chronic liver disease pathophysiology.The inclusion of Enterol as part of complex therapy in patients with CHB and CHC was associated with a marked positive effect on both clinical manifestations and microbiota composition. After a 30-day course, significant improvements were observed in bifidobacteria and lactobacilli levels, along with a reduction in Candida colonization and overall dysbiosis severity. Clinically, patients receiving the probiotic experienced faster resolution of specific symptoms such as vomiting, general fatigue, and sleep disturbances. These findings suggest that adjunctive probiotic therapy can accelerate recovery and improve patient well-being.Saccharomyces boulardii, the active component of Enterol, exerts several beneficial effects that may explain these clinical outcomes. It is highly resistant to gastric acidity and antibiotics, allowing it to reach the intestine in a viable form. In the gut, S. boulardii inhibits the growth of pathogenic and opportunistic microorganisms through competitive antagonism and secretion of antimicrobial peptides, enhances mucosal barrier integrity, and modulates local and systemic immune responses. These mechanisms collectively contribute to the restoration of intestinal homeostasis and reduction of bacterial translocation, which may, in turn, decrease hepatic inflammatory activity. The observed clinical benefits support the use of probiotics as a pathogenetic intervention targeting the gut–liver axis.Analysis of clinical symptom distribution revealed that more severe degrees of dysbiosis were associated with higher prevalence of headache, right hypochondrial pain, and sleep disturbances. These symptoms, often linked to systemic inflammatory and neurotoxic mediators such as ammonia and pro-inflammatory cytokines, contribute to patient discomfort, reduced quality of life, and decreased functional capacity. Although causality cannot be definitively inferred due to potential confounders, the correlation between dysbiosis severity and symptom burden emphasizes the clinical relevance of intestinal microbiota disturbances.The chosen probiotic regimen (250 mg twice daily for 30 days) proved effective and well tolerated. However, the rationale for the specific dose and duration was not explicitly based on prior dose-finding studies, and adherence to therapy was not formally monitored. Future studies could explore optimal dosing, treatment duration, and long-term effects on both microbiota composition and liver disease outcomes. In addition, the inclusion of placebo-controlled, randomized trials with larger sample sizes and stratification by fibrosis stage, disease duration, and comorbidities would provide more robust evidence regarding efficacy and safety.Overall, these findings indicate that correction of intestinal dysbiosis using probiotics such as Enterol can be considered an important adjunct to baseline therapy in chronic viral hepatitis. Probiotic intervention targets a pathogenetic pathway by modulating the gut–liver axis, reducing inflammatory activity, improving microbiota balance, and enhancing clinical recovery. Given the limited options for etiological therapy in patients with contraindications, high costs, or intolerance to antiviral agents, probiotics represent a safe, accessible, and potentially cost-effective strategy for improving patient outcomes.Future research should aim to integrate microbiome sequencing techniques, such as 16S rRNA profiling or metagenomic analysis, to provide a more comprehensive understanding of the microbial shifts associated with chronic viral hepatitis and the effects of probiotic therapy at the species and functional level.
5. Conclusions
- 1. All patients with chronic hepatitis B and C exhibited intestinal microbiota disturbances of varying severity.2. The severity of intestinal dysbiosis is directly associated with the duration of chronic viral hepatitis.3. Severe forms of dysbiosis are linked to an intensification of clinical manifestations and a reduction in patients’ quality of life.4. The inclusion of Enterol in complex therapy contributes to significant normalization of intestinal microbiota and a reduction in dysbiosis severity.5. Enterol is well tolerated and may be recommended as an effective component of pathogenetic therapy in patients with CHB and CHC.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML