-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 513-520
doi:10.5923/j.ajmms.20261602.28
Received: Dec. 9, 2025; Accepted: Jan. 2, 2026; Published: Feb. 3, 2026

Application of a Minimally Invasive Surgical Treatment Method for Osteoarthritis of the Knee Joints
Asilova Saodat Ubaevna1, Abdullaev Maxsur Maqsudjon o’g’li2, Tursunov Vosit Khakimbaevich2, Aliev Kholbek Erkinovich2, Babakulov Abduaziz Shadiyevich2, Nazirova Muyassar Ubaevna3
1Kimyo International University in Tashkent, Uzbekistan
2Akfa Medline University Hospital, Uzbekistan
3Tashkent State Medical University, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The purpose of the article is to consider the use of multipotent adipose tissue stem cells with platelet-rich plasma together after arthroscopic interventions in female patients to reduce the development of osteoarthritis of the knee joint. In 5 patients, the results were compared with the initial version within 6 months. The results were evaluated using clinical methods, ultrasound and MRI studies. The views of modern scientists on this issue are given. The results are presented, discussed and recommended for use in osteoarthritis of the knee joint.
Keywords: Knee arthroscopy, Stromal vascular fraction, Platelet-rich plasma, Cell therapy
Cite this paper: Asilova Saodat Ubaevna, Abdullaev Maxsur Maqsudjon o’g’li, Tursunov Vosit Khakimbaevich, Aliev Kholbek Erkinovich, Babakulov Abduaziz Shadiyevich, Nazirova Muyassar Ubaevna, Application of a Minimally Invasive Surgical Treatment Method for Osteoarthritis of the Knee Joints, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 513-520. doi: 10.5923/j.ajmms.20261602.28.
1. Introduction
- Osteoarthritis (OA) is the most common disease of the musculoskeletal system, which is characterized by pain, short–term stiffness, impaired function, degeneration of articular cartilage, sclerosis of the subchondral bone and the formation of osteophytes. Knee joint OA affects 250 million (3.8%) of the world's inhabitants. Among all nosological forms of joint pathology, OA is 55%. According to epidemiological studies, OA of various localization affects 1-2% of the population under 45 years of age and 15-85% of older people [1,2]. According to WHO, OA occurs in men in an average of 9.6%, in women in 18%. Therefore, when searching for new ways to prevent and treat OA, it is necessary to take into account the gender disparity (in 2015, there were 1,854 women per 1,000 men) and a higher incidence of this disease in women [3].The main clinical manifestations of knee joint OA are pain and limitation of function, as well as pathological changes in the structure of the joint, which causes a decrease in the quality of life. Despite the multifactorial nature of the disease, the most important etiopathogenetic factor of degenerative knee joint damage is inflammation. Signs of inflammation are observed both in the early and late stages of OA. At the same time, inflammation of the synovial membrane is accompanied by an increase in the level of proinflammatory mediators, including cytokines (TNF, IL-12, IL-6, IL-15, IL-17, IL-18, IL-21, LIF, IL-8), nitric oxide, PGE2, and is associated with damage to adjacent cartilage tissue [4,5].The low ability of articular cartilage to self-repair significantly limits the treatment options for patients with osteoarthritis of the knee joint [6]. Traditionally used nonsteroidal anti-inflammatory drugs and glucocorticosteroids suppress inflammation and are quite effective in relieving pain, but they are not able to stimulate regenerative processes in damaged cartilage. In addition, drugs of these groups, even when used in therapeutic doses, often cause the development of serious adverse reactions. Joint replacement surgeries are also relatively often associated with serious and life-threatening complications, including an increased risk of infection, thromboembolism, myocardial infarction, stroke, as well as a limited-service life of the endoprosthesis.One of the new directions in the treatment of knee osteoarthritis is associated with the use of cellular technologies based on the use of stem cells. The most promising candidates are multipotent mesenchymal stromal/stem cells (MSCs), which have pronounced anti-inflammatory and regenerative potential [7,8,9,10,11,12]. Common sources of these cells are bone marrow and adipose tissue, and adipose tissue has a number of advantages [13]. Thus, the number of MSCs that can be isolated from adipose tissue is 100-1000 times higher than their amount contained in an equivalent volume of bone marrow [14]. In addition, MSCs isolated from adipose tissue are genetically more stable over a long period of cultivation, have a lower aging coefficient and high proliferative activity [15,16]. Adipose tissue can be easily obtained by performing a standard liposuction procedure under local anesthesia, and lipotransfer methods are widely used in modern plastic surgery. In addition, the stromal vascular fraction (SVF) of lipoaspirate is a rich source not only of MSCs, but also of other cell types involved in tissue regeneration, for example, precursors of endothelial cells, preadipocytes, fibroblasts, mast cells, macrophages, T and B lymphocytes [16, 17]. SVF cells have anti-inflammatory and immunomodulatory effects; in addition, the MSCs contained in SVF are able to differentiate into connective tissue cells, including cartilage, tendons and ligaments [18]. A significant advantage can also be considered the possibility of using freshly isolated SVF cells that have not undergone cultural expansion in the form of minimally manipulated cells, since in vitro manipulations can lead to genetic and epigenetic changes that can affect the functional and biological properties of cells. Preclinical animal studies have shown the safety and effectiveness of SVF cells in the treatment of OA, cartilage defects or other orthopedic diseases [15,19,20]. Few pilot studies have demonstrated good tolerability, safety, and clinical efficacy of SVF in patients with early stages of OA [17,21,22,23].The purpose of the scientific workThe present study was undertaken with the aim of clinical approbation of autologous SVF cells with platelet-rich plasma in female patients with long–term knee osteoarthritis (gonarthrosis), grade II-III according to Kellgren-Lawrence after arthroscopic debridement.
2. Materials and Methods
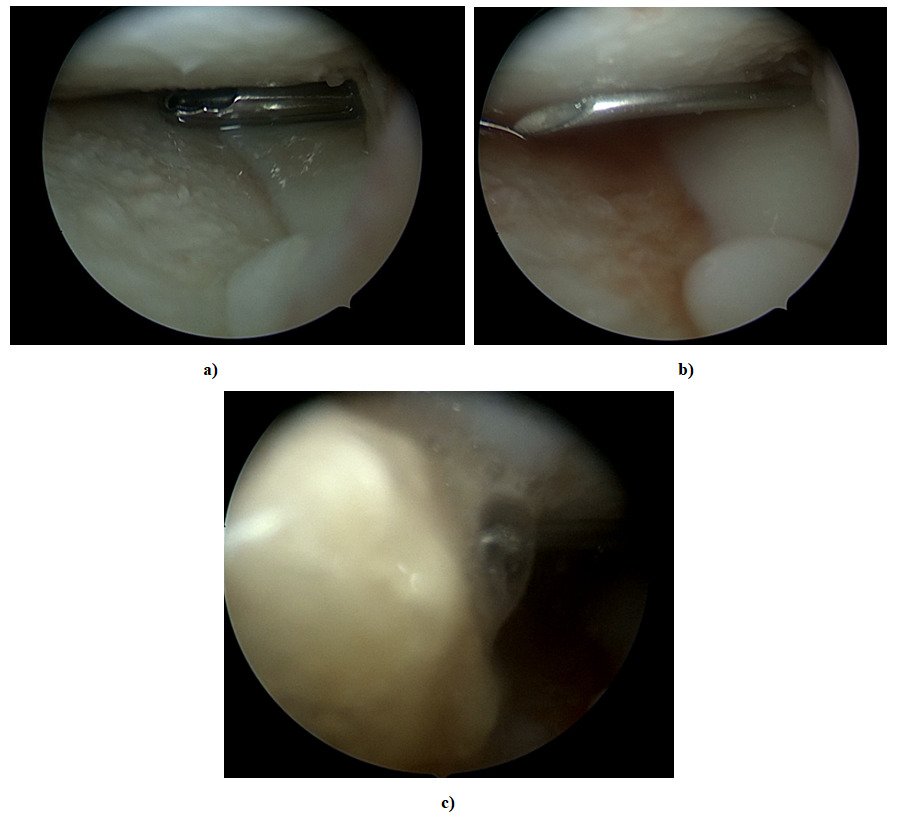
- Patients were recruited into the study in accordance with the protocol approved by the local ethical committee. Inclusion criteria: 1) women aged 40 to 60 years; 2) the presence of primary OA of the knee joints (Rg stage 2-3 according to Kellgren–Lawrence), verified clinically, radiologically, by magnetic resonance; 3) the possibility of liposuction; 4) the presence of written informed consent. Exclusion criteria: 1) blood diseases associated with a decrease in shaped elements; 2) autoimmune diseases; 3) acute infectious diseases; 4) mental illnesses; 5) oncological diseases 6) allergy to drugs for local anesthesia; 7) use of corticosteroid drugs in the previous 4 weeks; 8) contraindications for joint puncture, including joint replacement.Obtaining SVF cells Under local anesthesia, 2.0 ml of 0.5% novocaine, in the operating room, a group of surgeons simultaneously draw venous blood to obtain platelet-rich plasma (PRP), the resulting venous blood in a volume of 15 ml is treated with platelet release in a volume of about 5 ml by single centrifugation at 3200 rpm for 5 minutes in a 20 ml test tube with the addition of 1 ml of 5% sodium citrate. When taking adipose tissue, a Klein solution and 1 ml of dexamethasone are injected into subcutaneous adipose tissue through an infusion cannula, and fiber infiltration is performed. 30 minutes after the injection of the specified solution, about 40 ml of adipose tissue is taken using a special cannula for injection into the knee joint. By double centrifugation for 4 minutes at 2400 rpm and filtration in specialized syringes "Arthrex", about 5 ml of the stromal vascular fraction is obtained. The resulting stromal vascular fraction is mixed with pre-prepared platelet-rich plasma (PRP) from the patient's blood. The finished mass in a volume of 10 ml is transplanted into the affected joint, under local anesthesia 2.0 ml of 0.5% novocaine. The place of adipose tissue intake and the injection area are sealed with alcohol aseptic dressings. The stage of the SVF sampling operation is performed within 45-50 minutes.The research procedure SVF cells together with platelet-enriched plasma were injected once intraarticularly under the control of an arthroscope in a volume of 10 ml of cellular suspension per joint after arthroscopic debridement of the knee joints. The examination of patients was carried out before the introduction of cells and 1, 3, 6 months after the introduction of cells. The safety assessment included physical examination and analysis of adverse events (allergic, toxic, inflammatory reactions). In the analysis of clinical efficacy, an assessment of the severity of pain syndrome using a visual analog pain scale (VAS) and an assessment of the condition of affected joints and quality of life according to KOOS (Knee injury and Osteoarthritis Outcome Score; www.koos.nu), as well as ultrasound and MRI examination of the joints.
3. Results
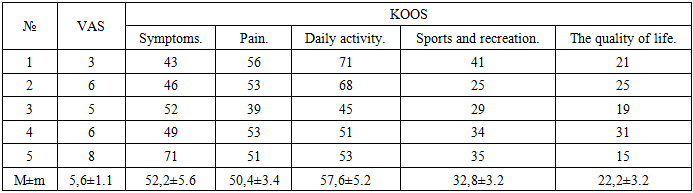
- Characteristics of patients The study recruited 5 patients, all women aged 40 to 60 years; median 50.4 years) with primary knee joint OA, Rg stage II (n = 2) and II-III (n = 3), NSF II ct (n=5). The duration of the disease is 5.6±3.2 years (Iu 7, IQR 4-8) (Table 1). To assess the functional state of the affected joints and the quality of life, a specialized 100-point scale for the knee joint KOOS was used, which allows you to assess pain, other symptoms of the disease (swelling, limitation of movement), daily activity, the possibility of sports/recreation, as well as the level of quality of life associated with the affected joint. The calculation of points on the KOOS scale was carried out on the basis of a patient questionnaire, with 100 points indicating the absence of symptoms, 0 points indicating their sharp severity. The data obtained are summarized in Table 2.
|
|
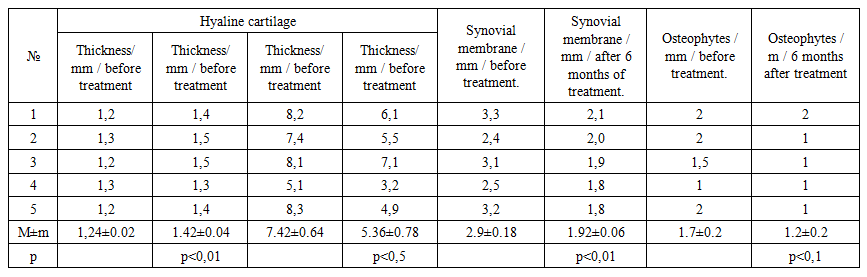 | Table 3. MRI of knee joints in patients with OA |
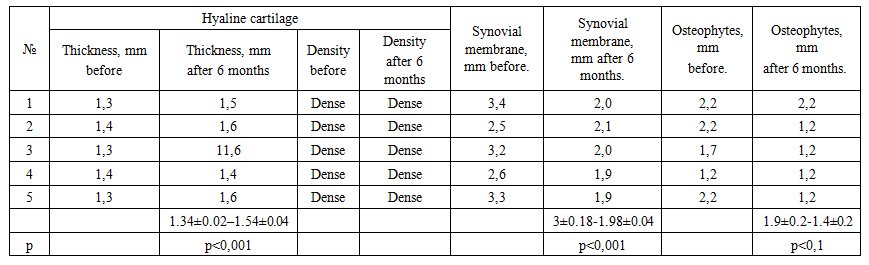 | Table 4. Ultrasound of the knee joints in patients with OA |
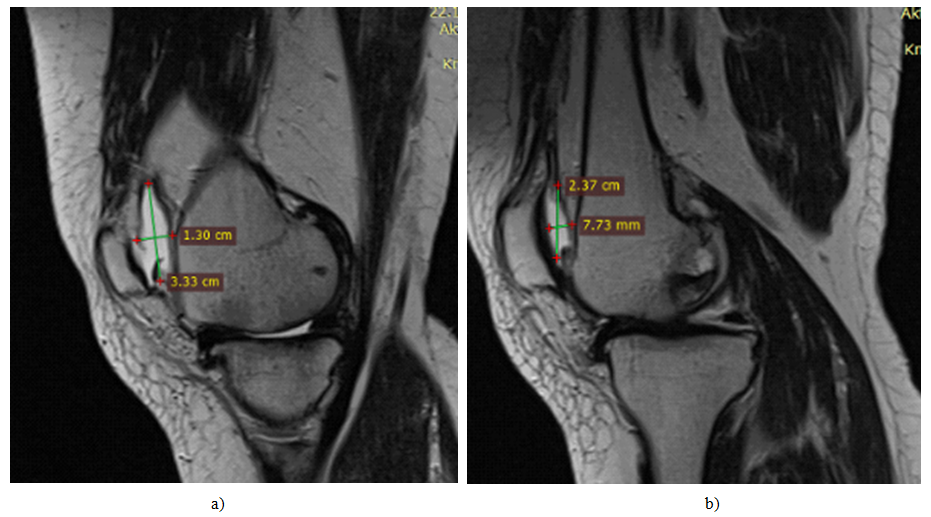 | Figure 2. MRI picture of synovial fluid volume. a)-before surgical intervention. b)-after 6 months of surgical intervention. |
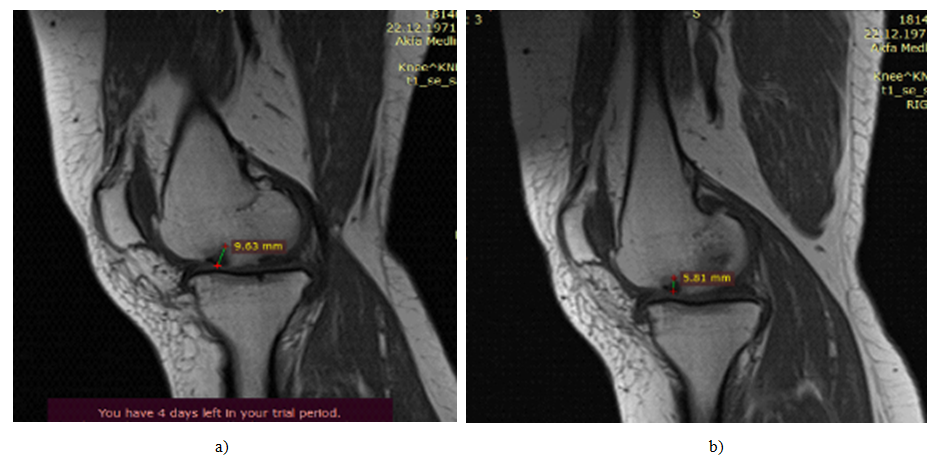 | Figure 3. MRI picture of cartilage defect, osteophyte volume. a)-before surgical intervention. b)-after 6 months of surgical intervention |
4. Discussion
- Nucleated SVF cells isolated from adipose tissue are finding increasing use in clinical practice. The therapeutic potential of SVF cells is associated primarily with a high content of stem/stromal cells, in particular MSCs, which have a pronounced trophic effect due to the secretion of a wide range of growth factors, cytokines and chemokines necessary for repair. A significant part of clinical studies is based on the use of cultured in vitro MSCs of adipose tissue [24], however, native SVF – as a "therapeutic agent", and not as a source for obtaining MSCs – has a number of advantages. One of the main advantages of SVF in comparison with "pure" stem/stromal cell populations obtained as a result of in vitro cultivation is the heterogeneity of this fraction. Indeed, along with MSCs, SVF samples contain various types of cells that have a stimulating effect on regeneration and revascularization processes and have anti-inflammatory and immunomodulatory properties (preadipocytes, precursors of endothelial cells, macrophages of the second type and T-regulatory cells) [25,26,27]. According to the criteria developed by the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT), SVF cells should have a viability of at least 70% and express the following immunophenotype: CD13+, CD29+, CD44+, CD73+, CD90+ (> 40%), CD34+ (> 20%), CD31+ (< 20%) and CD45+ (< 50%) [25]. In our study, lipoaspirates were obtained in all patients from the same anatomical region (anterior abdominal wall), SVF cells were isolated using a standard technique using a centrifuge and syringes from Arthrex, while the final population of nucleated SVF cells corresponded to generally accepted criteria in their characteristics: high viability along with a high content of MSCs precursors and variable CD34 expression (data not provided). It should be noted that it is in the treatment of patients with degenerative joint diseases that the therapeutic "value" of adipose tissue cells is higher compared, for example, with bone marrow, since adipose tissue MSCs differ from bone marrow MSCs by a higher chondrogenic potential [28]. At the same time, safety and good tolerability have been demonstrated for various routes of administration, including systemic [29,30] and local (intraarticular) [31,24]. Nevertheless, in patients with knee joint OA, intra–articular administration is preferable - not only because of the "targeted" cell delivery, but also because of the ability of patients' synovial fluid in contact with MSCs to enhance the chondrogenic potential of the latter [28]. A feature of our study can be considered the recruitment of patients with long-term knee osteoarthritis, mainly of the III radiological stage; at the same time, we did not exclude patients with a high body mass index (> 30) from the study, as is done by a number of authors. The message Correa and co-authors are close to us in terms of execution, presenting a description of a clinical case in which the nature of the changes and their dynamics are similar to the results we obtained [31]. In a clinical study by Jo et al. No adverse reactions and significant improvement on the WOMAC scale were reported 6 months after administration of 1×10 (grade 8) cells in 18 patients with knee joint OA [22]. Pak et al. Significant clinical improvement was demonstrated in several patients 2 years after the introduction of cells compared with data at 12 months [23]. In order to enhance the effects of SVF, some authors supplement SVF cells with various factors. For example, Bui et al. used SVF cells in combination with platelet-rich plasma (platelet-rich plasma, PRP) to treat 21 patients with OA II-III [21]. The authors reported two main positive effects – pain reduction after 3-6 months and stimulation of cartilage growth. At the same time, both of these effects are attributed to PRP, since, firstly, PRP suppresses the NF-kB cascade activated in chondrocytes of patients with OA [32] and, secondly, PRP suppresses the production of VEGF by stromal/stem cells of adipose tissue, which is an inhibitor of cartilage growth [33], thereby facilitating prochondrogenic effect of adipose tissue stem cells. It is important to note that in our study, pain relief was also achieved by transplantation of SVF cells using PRP, and statistically significant changes were recorded after 1 month. after the introduction of cells.There are also data in the literature from a multicenter non-randomized clinical trial in which 1,128 patients with osteoarthritis of large joints were recruited [34]. According to the presented data, the treatment of osteoarthritis using autologous SVF adipose tissue led to significant clinical improvement in the vast majority of patients. Moreover, with long–term follow–up of patients (up to 4.5 years), in none of the cases were such severe undesirable consequences or side effects associated with treatment as infectious complications, oncological or autoimmune diseases recorded. Interestingly, the therapeutic effect of SVF cells is not limited to positive changes in the target organ, but is broader. Thus, in the Bright study, it was found that intravenous administration of SVF to patients with osteoarthritis was accompanied by an unexpected decrease in the frequency and intensity of migraine attacks [35]. The authors suggest that this effect is associated with the anti-inflammatory and immunomodulatory activity of MSCs and M2 macrophages. The results of our pilot study indicate the safety and good tolerability of intraarticular injection of autologous SVF cells of adipose tissue in patients with osteoarthritis of the knee joints. This approach can be considered as one of the methods that prevent further progression of the disease, and in the case of severe injuries, such therapy can help reduce pain and other manifestations of arthritis and thus facilitate the patient's period before implantation of an artificial joint. However, further studies involving controlled, randomized clinical trials with long-term prospective follow-up are needed to verify these assumptions. In addition, in order to prove the direct anti-inflammatory effect of SVF cells, it is planned to further study biomarkers of the inflammatory process both locally (in synovial fluid) and at the systemic level (in the blood serum of patients with osteoarthritis of the knee joint).
5. Conclusions
- Cell therapy can be used immediately after arthroscopic interventions in the knee joints. The use of platelet-rich plasma (PRP) in combination with the stromal vascular fraction represents a safe and effective treatment of osteoarthritis of the knee joints in the early stages after arthroscopic interventions. By stopping the progression of osteoarthritis, this method can prevent the need for a complete knee replacement.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML