-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 503-506
doi:10.5923/j.ajmms.20261602.26
Received: Jan. 3, 2026; Accepted: Jan. 21, 2026; Published: Feb. 3, 2026

The Effect of Carbon Monoxide on the Morphological Parameters of the Thymus
Qodirov Oybek O‘ktam ugli1, Teshayev Shuxrat Jumayevich2
1PhD., Associate Professor, Department of Histology, Cytology and Embryology Bukhara State Medical Institute, Bukhara, Uzbekistan
2Doctor of Medical Sciences, Professor, Department of Anatomy and Clinical Anatomy (OHTA), Bukhara State Medical Institute, Bukhara, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Morphofunctional changes in the thymus were identified as a result of chronic exposure to an isocyanate isotope in laboratory animals under experimental conditions. Chronic exposure to the isocyanate isotope caused specific alterations in each part of the thymus, including the proliferation of connective tissue, cellular hypertrophy, signs of inflammation, and dilation of blood vessels.
Keywords: Сhronic carbon monoxide exposure, Oxidative stress, Apoptosis, Spleen, Morphological changes, Experimental study
Cite this paper: Qodirov Oybek O‘ktam ugli, Teshayev Shuxrat Jumayevich, The Effect of Carbon Monoxide on the Morphological Parameters of the Thymus, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 503-506. doi: 10.5923/j.ajmms.20261602.26.
Article Outline
1. Introduction
- Carbon monoxide poisoning disrupts the metabolism of carbon and protein, resulting in symptoms of acidosis. The balance of potassium and calcium in the blood and the functioning of the central nervous system can be disrupted, which can lead to premature death [15,16,17]. The harmful effects of carbon monoxide include changes in the body, including the morphological characteristics of organs, and the development of therapeutic and preventive measures to reduce the harmful effects of carbon monoxide has not lost its relevance [1,2].In practice, the morphometric changes in the thymus and the negative effects of carbon monoxide on the human body are very diverse, and many scientific studies are being conducted around the world to solve this urgent problem. These works were carried out at the world's leading research centers and universities (Western Kentucky University, Dominican University of California, Harvard University, University of Missouri-Columbia, University of Nebraska–Lincoln, Colby College (USA), Oxford University (Great Britain) [3,4,5]. Living organisms live under a huge number of external influences. It is no secret that carbon monoxide is constantly causing harm to humanity among such external influences. Carbon monoxide affects the body through the respiratory system [6,7,8,9]. According to researchers and scientists from leading scientific centers in the world today, the presence of 0.1% carbon monoxide in the composition of the inhaled air can lead to death [9,10]. This gas forms a compound with hemoglobin that is 300 times stronger than oxygen - carboxyhemoglobin [11,12]. As a result, the ability of hemoglobin to transport oxygen to tissues decreases sharply, leading to hypoxia, and in severe cases, to anoxia. can lead to [13,14].Оbjеctivе оf thе study To identify the morphological and functional changes in spleen tissue under chronic inhalational exposure to carbon monoxide (CO), evaluate their severity and developmental stages, and scientifically substantiate the mechanism of CO influence on spleen immunological reactivity based on morphological, morphometric, histological, and biochemical parameters.
2. Mаtеriаl аnd Mеthоds оf Rеsеаrch
- The experiments were conducted on 208 white female outbred rats born in vivarium conditions. Rats aged 3, 6, 9 and 12 months were involved. The experiments followed the ethical rules for the use of animals and the requirements of the Helsinki Congress. Before the start of the experiments, all sexually mature rats were quarantined for a week and, excluding somatic or infectious diseases, they were transferred to the usual vivarium regime under the same conditions. During the experiment, the behavior and physiological state of animals in the standard and experimental groups were monitored. Rats were divided into 4 groups (n = 208): I-control group (n = 40); II-IV-groups (n = 168) experimental animals were chronically poisoned with a dose of carbon monoxide in the air of 0.01-0.05 mg / l. During the experiment, 4 3-month-old, 1 6-month-old, and 1 12-month-old rats died as a result of chronic carbon monoxide poisoning. After that, we divided 162 outbred white female rats chronically poisoned with carbon monoxide into 3 more groups. Group 2 (n = 54) rats chronically poisoned with carbon monoxide in the experiment; Group 3 (n = 54) rats chronically poisoned with carbon monoxide were administered intragastrically through a metal stomach tube for 14 days with 1 ml of infusion of the plant Gulimansar; In the 4th group (n = 54) experiment, rats chronically poisoned with carbon monoxide were administered intragastrically through a metal gastric tube for 14 days with a volume of 0.1 ml of an alcoholic solution of asparagus oil (in a ratio of 1:9). A total of 208 rats were used in the experiments, of which only 6 died during the experiments.
3. Rеsults аnd Discussiоn
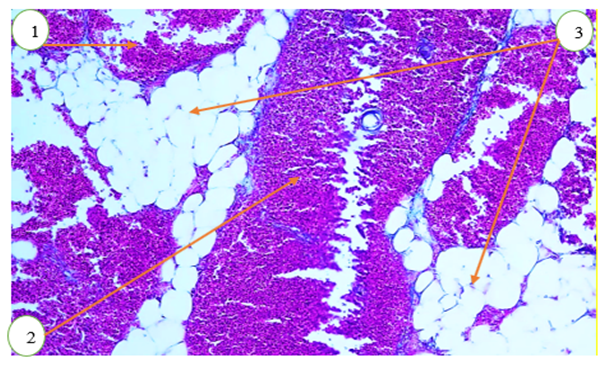
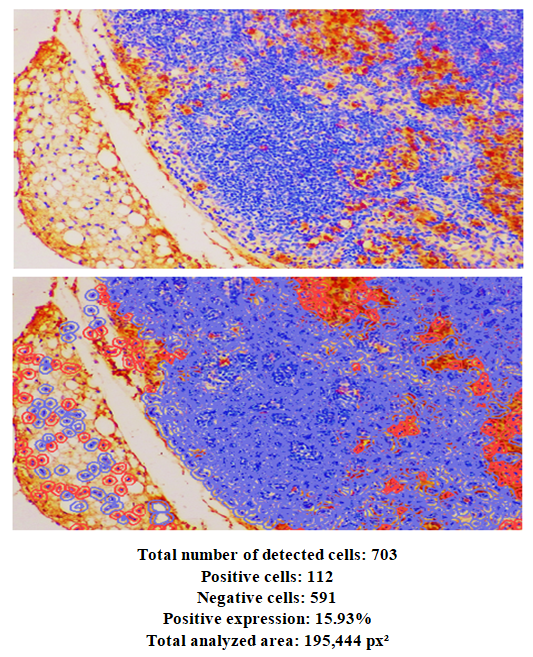
- In 6-month-old rats, the mean thickness of the thymic capsule was 4.6 ± 0.21 µm at the hilum, 7.0 ± 0.10 µm at the anterior pole, and 5.7 ± 0.14 µm at the posterior pole. The mean diameter of the trabecula was 11.1 ± 0.19 µm in the proximal part and 7.7 ± 0.24 µm in the distal part. The average depth of the trabecula was 12.7 ± 0.31 µm.In 6-month-old rats, the mean area of the thymic lobule was 66.3 ± 0.9%. In the control group of 6-month-old rats, the mean area of the cortical region was 64.9 ± 1.3%, the mean area of the medullary region was 25.4 ± 1.6%, and the corticomedullary index was 2.4 ± 0.1%.In 6-month-old rats, the mean wall thickness of trabecular vessels was as follows: arteriole – 16.8 ± 0.32 µm, venule – 15.9 ± 0.24 µm, and capillary – 4.2 ± 0.10 µm. The mean internal diameter of the trabecular arteriole was 18.2 ± 0.15 µm; the mean diameter of the accompanying arteriole was 18.2 ± 0.15 µm; and the mean diameter of the venule was 4.8 ± 0.30 µm.The mean wall thickness of cortical vessels was 15.4 ± 0.17 µm for arterioles, 13.9 ± 0.23 µm for venules, and 3.4 ± 0.10 µm for capillaries. The mean internal diameter of cortical arterioles was 17.2 ± 0.33 µm, venules – 23.1 ± 0.25 µm, and capillaries – 4.5 ± 0.30 µm.In the medullary region, the mean wall thickness of arterioles was 14.1 ± 0.11 µm, venules – 14.6 ± 0.36 µm, and capillaries – 2.6 ± 0.10 µm. The mean internal diameter of medullary arterioles was 15.8 ± 0.17 µm, venules – 15.1 ± 0.21 µm, and capillaries – 3.7 ± 0.30 µm.In this image, we can observe structural disruptions in both the cortical and medullary regions of the thymus. In a normal thymus, the cortex (darker/redder) and medulla (lighter) are clearly distinguishable. In this image, however, their boundaries appear blurred and disrupted. A decrease in the number of cells and their dispersed arrangement in the cortical region indicate signs of lymphoid atrophy.
4. Discussion
- Immunohistochemical changes in the thymus of 6-month-old outbred white rats exposed to chronic carbon monoxide and treated with citric acid for therapeutic and prophylactic purposes were investigated. In 6-month-old rats, the mean thickness of the thymic capsule was 12.7 ± 0.21 µm at the hilum, 15.6 ± 0.10 µm at the anterior pole, and 14.1 ± 0.14 µm at the posterior pole. The mean diameter of the trabecula was 20.1 ± 0.19 µm in the proximal part and 16.7 ± 0.24 µm in the distal part. The average depth of the trabecula was 20.7 ± 0.31 µm. The mean area of thymic lobules in 9-month-old rats was 73.3 ± 0.9%.In the control group of 6-month-old rats, the mean cortical area was 73.9 ± 1.3%, the medullary area was 34.4 ± 1.6%, and the corticomedullary index was 10.5 ± 0.1%.The mean wall thickness of trabecular vessels in 6-month-old rats was 33.8 ± 0.32 µm for arterioles, 32.9 ± 0.24 µm for venules, and 21.2 ± 0.10 µm for capillaries. The mean internal diameter was 32.2 ± 0.15 µm for arterioles, 38.4 ± 0.33 µm for venules, and 21.8 ± 0.30 µm for capillaries.In the cortical layer, the mean wall thickness of arterioles was 32.4 ± 0.17 µm, venules 30.9 ± 0.23 µm, and capillaries 20.4 ± 0.10 µm. The mean internal diameter of cortical arterioles was 34.2 ± 0.33 µm, venules 40.1 ± 0.25 µm, and capillaries 21.5 ± 0.30 µm.In the medullary region, the mean wall thickness of arterioles was 31.1 ± 0.11 µm, venules 31.6 ± 0.36 µm, and capillaries 20.6 ± 0.10 µm. The mean internal diameter of medullary arterioles was 25.8 ± 0.17 µm, venules 25.1 ± 0.21 µm, and capillaries 13.7 ± 0.30 µm.
5. Cоnclusiоns
- Immunohistochemical changes in the thymus of 6-month-old outbred white rats exposed to chronic carbon monoxide and treated with citric acid for therapeutic and prophylactic purposes were investigated. In 6-month-old rats, the mean thickness of the thymic capsule was 12.7 ± 0.21 µm at the hilum, 15.6 ± 0.10 µm at the anterior pole, and 14.1 ± 0.14 µm at the posterior pole. The mean diameter of the trabecula was 20.1 ± 0.19 µm in the proximal part and 16.7 ± 0.24 µm in the distal part. The average depth of the trabecula was 20.7 ± 0.31 µm. The mean area of thymic lobules in 9-month-old rats was 73.3 ± 0.9%.In the control group of 6-month-old rats, the mean cortical area was 73.9 ± 1.3%, the medullary area was 34.4 ± 1.6%, and the corticomedullary index was 10.5 ± 0.1%.The mean wall thickness of trabecular vessels in 6-month-old rats was 33.8 ± 0.32 µm for arterioles, 32.9 ± 0.24 µm for venules, and 21.2 ± 0.10 µm for capillaries. The mean internal diameter was 32.2 ± 0.15 µm for arterioles, 38.4 ± 0.33 µm for venules, and 21.8 ± 0.30 µm for capillaries.In the cortical layer, the mean wall thickness of arterioles was 32.4 ± 0.17 µm, venules 30.9 ± 0.23 µm, and capillaries 20.4 ± 0.10 µm. The mean internal diameter of cortical arterioles was 34.2 ± 0.33 µm, venules 40.1 ± 0.25 µm, and capillaries 21.5 ± 0.30 µm.In the medullary region, the mean wall thickness of arterioles was 31.1 ± 0.11 µm, venules 31.6 ± 0.36 µm, and capillaries 20.6 ± 0.10 µm. The mean internal diameter of medullary arterioles was 25.8 ± 0.17 µm, venules 25.1 ± 0.21 µm, and capillaries 13.7 ± 0.30 µm.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML