-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 495-499
doi:10.5923/j.ajmms.20261602.24
Received: Jan. 3, 2026; Accepted: Jan. 21, 2026; Published: Feb. 3, 2026

Integrated Hormonal and Doppler Ultrasound Predictors of Diminished Ovarian Reserve in Women
Ibragimova Hakimaxon Ravshanjon qizi1, Yuldasheva Surayya Zaripovna2
1Phd Student, Fergana Medical Institute of Public Health, Uzbekistan
2DSc, Associate Professor, Tashkent State Medical University, Uzbekistan
Correspondence to: Ibragimova Hakimaxon Ravshanjon qizi, Phd Student, Fergana Medical Institute of Public Health, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The study assessed integrated hormonal, ultrasound and Doppler predictors of diminished ovarian reserve in 244 women of reproductive age. Women with low ovarian reserve were characterized by a higher burden of infertility and miscarriages, increased prevalence of ovarian surgery, pelvic inflammatory disease, chronic stress and metabolic disorders. Endocrine changes (elevated FSH, prolactin, free testosterone, TSH and anti-TPO) were accompanied by a marked reduction in ovarian volume and AFC. Doppler findings demonstrated increased vascular resistance in uterine and ovarian arteries. Thresholds RI>0.65 and PI>1.8 were strongly associated with low ovarian reserve. An integrated outpatient pathway combining hormonal tests, ultrasound morphology and Doppler indices may improve early detection and clinical risk stratification.
Keywords: Diminished ovarian reserve, Doppler ultrasound, Uterine artery, Ovarian artery, AMH, FSH, Antral follicle count
Cite this paper: Ibragimova Hakimaxon Ravshanjon qizi, Yuldasheva Surayya Zaripovna, Integrated Hormonal and Doppler Ultrasound Predictors of Diminished Ovarian Reserve in Women, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 495-499. doi: 10.5923/j.ajmms.20261602.24.
1. Introduction
- Diminished ovarian reserve (DOR) is among the leading causes of reduced fertility and early reproductive aging and remains a key challenge for both outpatient gynecology and reproductive medicine. [2,13,15] Clinically, DOR is associated with a lower probability of spontaneous conception, reduced ovarian responsiveness to stimulation, poorer outcomes of assisted reproductive technologies, and a narrowed timeframe for fertility planning. [13,15] In recent years, an increasing proportion of women have presented with signs of premature follicular pool depletion, which is not explained by chronological age alone. [15] Accumulating evidence suggests that ovarian reserve decline may be accelerated by chronic pelvic inflammatory disease, previous ovarian surgery, endometriosis, metabolic disturbances (including obesity and insulin resistance), environmental influences, and sustained psychoemotional stress; additionally, immune-mediated mechanisms and thyroid autoimmunity may contribute to ovarian dysfunction in susceptible women. [2,9,12,13,15]Current clinical assessment of ovarian reserve relies primarily on endocrine markers and ultrasound morphology. Anti-Müllerian hormone (AMH) and follicle-stimulating hormone (FSH) are widely used as the most informative laboratory indicators, while antral follicle count (AFC) and ovarian volume represent the core ultrasound parameters. [3,6,13] Nevertheless, these measures do not always capture the earliest functional alterations of the ovarian microenvironment. Endocrine shifts may become apparent after a period of subclinical impairment, and structural ultrasound changes may reflect comparatively late stages of ongoing functional decline. [5,14] This diagnostic gap is particularly relevant in outpatient practice, where women often seek evaluation only after unsuccessful attempts to conceive, and where timely risk identification could improve counseling, surveillance, and preventive reproductive planning. [3,6,13,15]A central concept in the contemporary pathogenesis of DOR is the contribution of microcirculatory disturbances and reduced intrastromal perfusion. Impaired uterine–ovarian blood flow can compromise granulosa-cell trophism, disturb angiogenic regulation, and accelerate follicular atresia, thereby promoting earlier depletion of the follicular pool. [5,14] Doppler ultrasonography offers a non-invasive functional assessment of uterine and ovarian perfusion and enables quantitative characterization of vascular impedance using resistance index (RI), pulsatility index (PI), and related velocity parameters. [1,5,10,11] Importantly, multiple studies have demonstrated associations between ovarian stromal blood flow indices and ovarian reserve markers or ovarian response in assisted reproduction, supporting the clinical relevance of Doppler assessment as a complementary diagnostic dimension. [5,8,11,14] However, despite these advances, Doppler indices are not consistently incorporated into routine ovarian reserve assessment algorithms, and the added value of integrating Doppler hemodynamic markers with endocrine and ultrasound parameters remains insufficiently standardized across clinical settings. [1,5,10,11,14]Accordingly, there is a need for clinically practical integrated models that combine endocrine biomarkers with ultrasound morphology and Doppler hemodynamic indices to strengthen early detection and outpatient risk stratification of DOR. [14,15] The novelty of the present work lies in evaluating an integrated hormonal–ultrasound–Doppler approach within a single clinical framework and examining its potential utility for early identification of women at risk of diminished ovarian reserve in routine practice. [14,15] Therefore, the aim of this study was to evaluate the diagnostic value of an integrated hormonal and Doppler ultrasound approach for early identification of women at risk of diminished ovarian reserve. [14,15]The aim of this study was to evaluate the diagnostic value of an integrated hormonal and Doppler ultrasound approach for early identification of women at risk of diminished ovarian reserve.
2. Materials and Methods
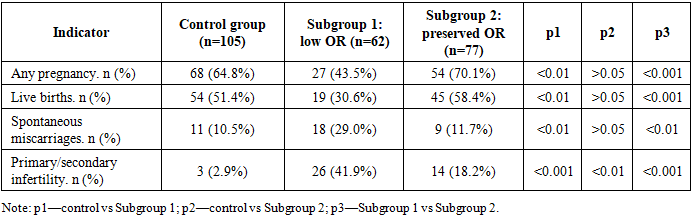
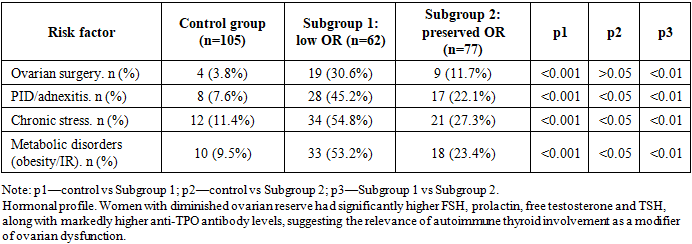
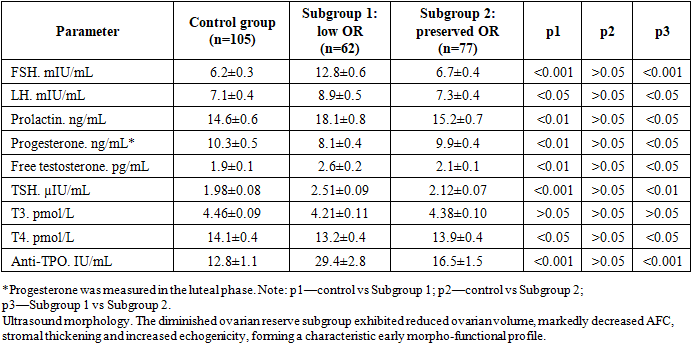
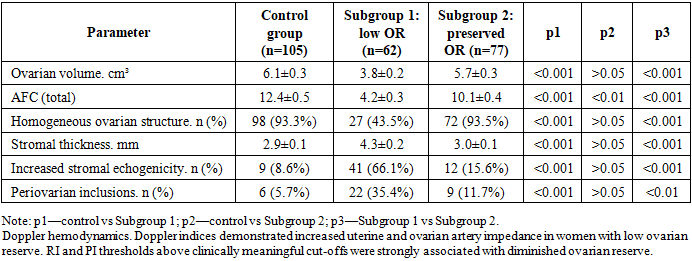
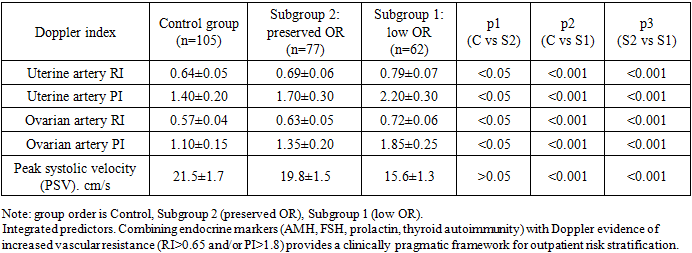
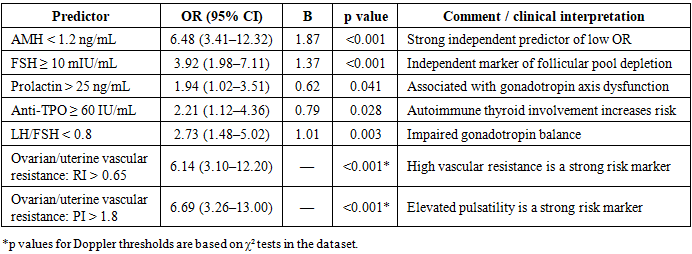
- Study design and participants. A cross-sectional analytical study included 244 women of reproductive age who underwent comprehensive clinical, laboratory, ultrasound and Doppler examination. The cohort was divided into a control group (n=105) and two clinical subgroups: Subgroup 1—women with diminished ovarian reserve (n=62) and Subgroup 2—women with preserved ovarian reserve (n=77). Subgroup allocation was based on ovarian reserve assessment (AMH cut-off, reduced antral follicle count and decreased ovarian volume) and clinical evaluation. Doppler indices were analyzed as additional functional hemodynamic parameters and were not used as stand‑alone criteria for subgroup assignment.Inclusion criteria: reproductive age; availability of complete clinical, laboratory and ultrasound data; informed consent. Exclusion criteria: current pregnancy; ovarian malignancy; acute pelvic infection at the time of examination; endocrine therapy within the previous 3 months; severe systemic disease potentially affecting gonadotropin secretion or ovarian perfusion.Hormonal assessment. Blood samples were obtained in the early follicular phase (cycle days 2–5). The following parameters were assessed: FSH, LH, prolactin, progesterone (luteal phase), free testosterone, thyroid-stimulating hormone (TSH), free thyroid hormones (T3, T4), and anti-thyroid peroxidase antibodies (anti-TPO).Ultrasound and Doppler protocol. Transvaginal ultrasound included ovarian volume, AFC (total), stromal thickness and echogenicity. Spectral Doppler evaluation of uterine and ovarian arteries was performed on cycle days 5–7. The resistance index (RI), pulsatility index (PI) and peak systolic velocity (PSV) were recorded.Statistical analysis. Quantitative variables were summarized as mean ± SD (or median [IQR] when non‑normally distributed). Normality was assessed using the Shapiro–Wilk test. Between‑group comparisons were performed using Student’s t‑test/one‑way ANOVA or the Mann–Whitney U/Kruskal–Wallis tests, as appropriate. Categorical variables were compared using χ² or Fisher’s exact test. For multiple pairwise comparisons (p1, p2, p3), adjusted p values were interpreted with attention to multiplicity. Associations were quantified using odds ratios (OR) with 95% confidence intervals (CI). Where applicable, multivariable logistic regression was used to explore independent associations.
3. Results
- Reproductive history and risk factors. The diminished ovarian reserve subgroup demonstrated a higher frequency of infertility and spontaneous miscarriages. A history of ovarian surgery, pelvic inflammatory disease, chronic stress and metabolic disorders occurred significantly more often in women with low ovarian reserve.
|
|
|
|
|
|
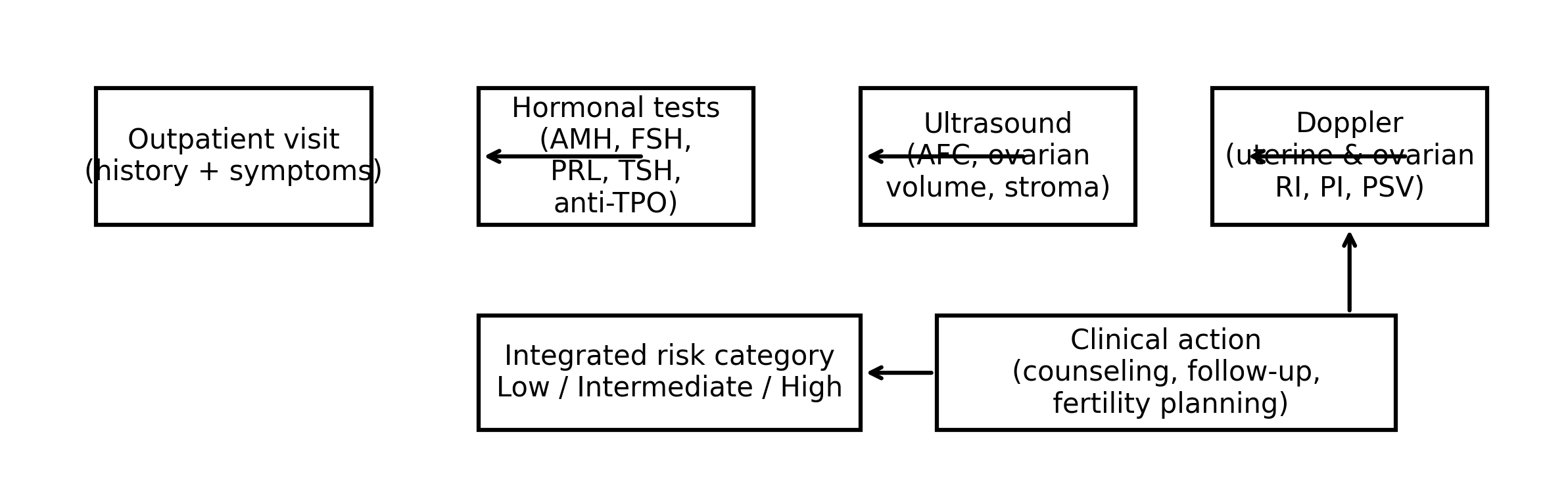 | Figure 1. Outpatient diagnostic pathway for integrated risk stratification (schematic). |
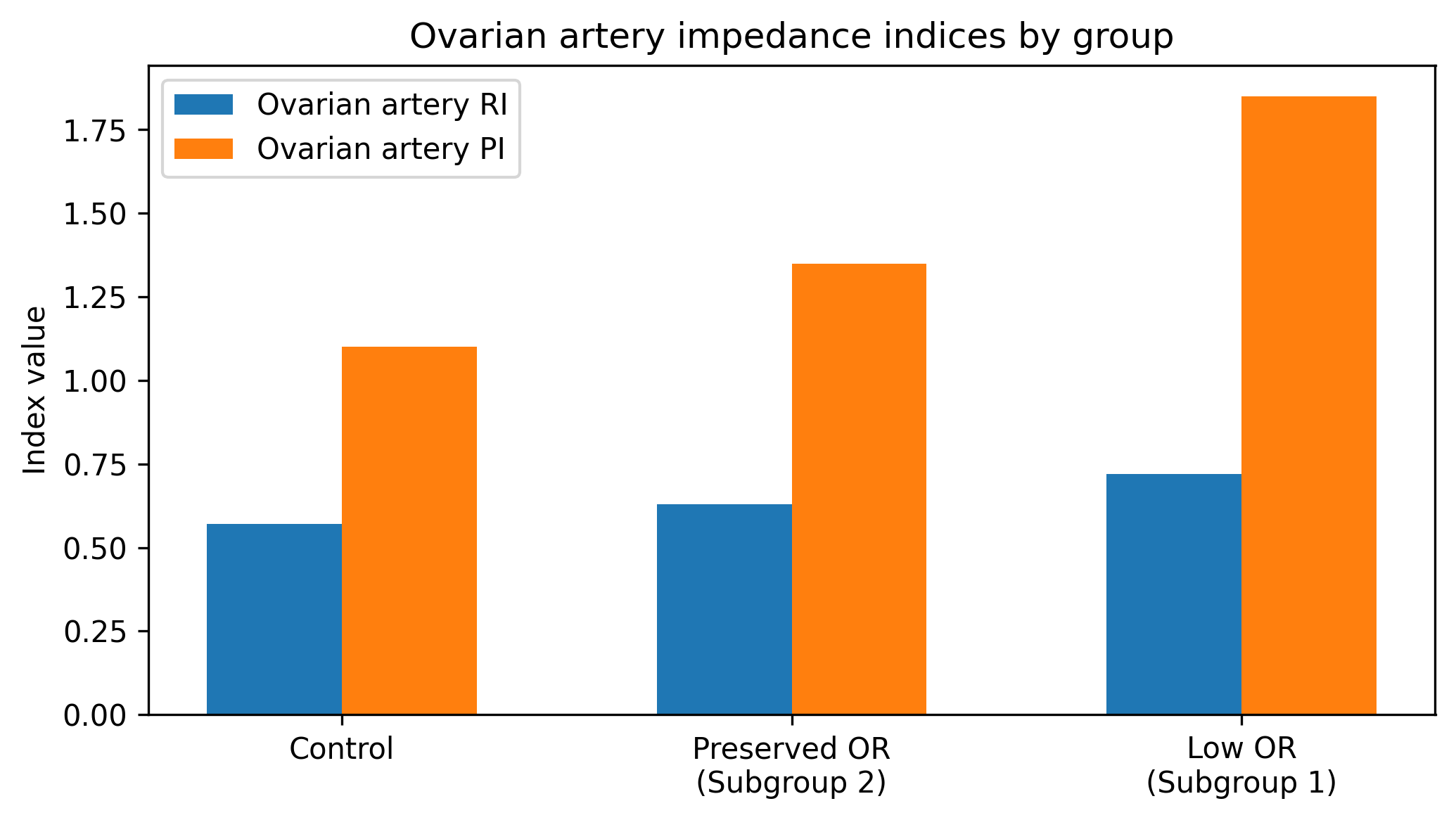 | Figure 2. Ovarian artery RI and PI across groups (based on group means) |
4. Discussion
- This study demonstrates that diminished ovarian reserve is associated with a combined pattern of endocrine, morphologic and hemodynamic alterations. FSH elevation, prolactin increase, androgen excess and thyroid autoimmune markers were accompanied by reductions in ovarian volume and AFC and by increased impedance in uterine and ovarian arteries.The hemodynamic component is clinically relevant because impaired perfusion may precede pronounced endocrine deterioration. Increased RI and PI reflect higher downstream resistance and reduced diastolic flow, which may compromise follicular trophism and granulosa cell function.From a practical perspective, isolated biomarkers have limited performance in heterogeneous outpatient populations. Therefore, an integrated diagnostic approach—hormonal profile + ultrasound morphology + Doppler indices—can strengthen early detection and targeted counseling. The proposed integrated model aligns with the concept of early functional impairment preceding irreversible follicular depletion.Limitations include the cross-sectional design and absence of prospective reproductive outcomes. Future work should validate the integrated model prospectively and derive a calibrated risk score with ROC analysis using individual-level data.
5. Conclusions
- 1. Diminished ovarian reserve in women of reproductive age is associated with a combination of reproductive history burden, risk factors, endocrine dysregulation, ovarian morphological changes, and impaired uterine–ovarian hemodynamics.2. Key endocrine predictors include AMH <1.2 ng/mL and FSH ≥10 mIU/mL, with additional contributions from hyperprolactinemia, thyroid autoimmunity (anti-TPO), and disturbed LH/FSH balance.3. Hemodynamic markers (RI >0.65 and PI >1.8) in uterine and ovarian arteries are strongly associated with diminished ovarian reserve and provide clinically useful early functional signals.4. An integrated hormonal–ultrasound–Doppler approach may improve outpatient risk stratification and support timely fertility counseling and preventive reproductive planning.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML