Khamidova F. M., Norjigitov A. M.
Department of Pathological Anatomy, Samarkand State Medical University, Samarkand, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
The article presents a comprehensive analysis of a retrospective, diagnostic study of bronchiectasis in the population of the Samarkand region. The inclusion and exclusion criteria of patients were carefully studied, confirming the validity of the selected study groups. Particular attention is paid to the comparative histological and genetic analysis of patients with bronchiectasis. Bronchiectasis is more common in the city of Samarkand, Urgut and Narpay districts. The disease is seasonal, reaching its peak in the spring and autumn seasons. For the study, the results of pathomorphological - general histological, histochemical and morphometric examination of bronchi and lung lobes of 48 patients who underwent lobectomy due to bronchiectasis, as well as the results of laboratory biopsies (blood) of 50 patients with bronchiectasis to study the (dell) mutation of the GSTM1 gene polymorphism were studied.
Keywords:
Bronchiectasis, GSTM1, Morphometry, Patomorfology
Cite this paper: Khamidova F. M., Norjigitov A. M., The Role of GSTM1 Gene Polymorphism in the Development of Bronchiectasis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 489-494. doi: 10.5923/j.ajmms.20261602.23.
1. Introduction
Bronchiectasis is a chronic acquired and, in some cases, congenital disease characterized by irreversible changes (dilation, deformation) of the bronchi, accompanied by their functional impairment, impaired drainage function and chronic purulent-inflammatory processes in the bronchial tree, peribronchial space, the development of atelectasis, emphysema and cirrhosis of the lung parenchyma [1].Respiratory system diseases occupy a leading position among all diseases. The same trend is observed in pediatric practice, where their share is more than 70% [2]. Studies have shown that the prevalence of bronchiectasis detected by computed tomography (CT) in patients with COPD varies from 4% to 72%, depending on the bronchiectasis criteria and the populations studied. In contrast, approximately 20% of cases of COPD are recorded as the primary diagnosis in US and European bronchiectasis registries [3].Genetic defects can also play a role in the development of bronchiectasis. In modern pulmonology, chronic nonspecific lung diseases are considered multifactorial, based on the increased sensitivity of the body to environmental factors. The genetic component, mainly single nucleotide polymorphisms (SNPs), plays an important role in their manifestation. Polymorphisms affecting the semantic parts of genes often lead to the exchange of amino acids and the emergence of proteins with new functional properties [4].GSTT1 (Glutathione S-transferase theta 1) is a phase II detoxification gene (22q11.23), encoding an enzyme that ensures the binding of glutathione to toxic metabolites and protects cells from oxidative stress. The GSTT1 (0) deletion polymorphism leads to the loss of enzymatic activity and is associated with increased inflammation and adverse disease outcomes, while the functional allele is protective, including in pneumonia in children.A number of studies have shown that genetic polymorphisms in genes encoding enzymes responsible for the biotransformation of xenobiotics contribute to the development of bronchopulmonary diseases such as chronic obstructive pulmonary disease (COPD) [5]. Carriers of the dell haplotype of GSTT1 and GSTM1 have an increased risk of developing COPD [6]. However, a number of authors have not identified such associations [7,8].Recently, there is increasing evidence that single nucleotide polymorphisms play an important role in the development of immune responses by shaping certain gene alleles. Genetic predisposition may include specific polymorphisms that determine the level of antioxidant protection. It can increase the risk of developing diseases at any stage: both during pregnancy and after childbirth.Many studies have shown that the risk of developing bronchopulmonary diseases with combined polymorphisms of different genes is higher than with a single gene defect. Homozygotes for the "dell" alleles of two genes at once, GSTM1 0/0 and GSTT1 0/0, are risk factors for the development of asthma. The accumulation of defects in the glutathione-S-transferase family system contributes to the persistent, severe course of asthma in children. In asthma, genetically determined defects in the production of xenobiotic detoxification enzymes GSTT1 and GSTM1, which occur as part of limited allergic damage to the airways in children without a family history of allergic heredity, have been shown to be a leading link in the pathogenesis of the disease [9].A meta-analysis of 12 studies (4484 participants) showed that GSTM1-dell and GSTT1-dell genotypes significantly increased the risk of oral submucosal fibrosis. The associations were strengthened by the large sample size and high exposure to areca nut. The results support the role of GST polymorphisms in the pathogenesis of the disease and their importance in risk stratification and early diagnosis [10].A significant relationship was found between the child's age and the consumption of seafood and canned fish, legumes, cereals and starchy foods with the level of mercury in the blood (p<0.05). A significant interaction was found between the consumption of canned fish and the GSTP1 gene polymorphism: increased mercury levels were observed only in children with the Ile/Ile and Ile/Val genotypes. The data obtained require confirmation in other populations [11].The study was aimed at assessing the influence of environmental factors and polymorphisms of the glutathione-S-transferase (GSTT1, GSTM1, GSTP1) genes on the likelihood of detecting elevated blood aluminum concentrations in children. The sample included 366 children aged 2-8 years with typical development (control group, Jamaica). Consumption of legumes was associated with a higher likelihood of detecting aluminum in the blood, while a higher level of parental education reduced this risk. A significant interaction was found between seafood consumption and GSTP1 genotype: the likelihood of detecting aluminum was observed only in children with the GSTP1 Ile/Ile genotype. The results obtained require confirmation in other populations [12].Therefore, one of the urgent problems in our study is the study of GSTM1 gene polymorphism in patients with bronchiectasis in the Samarkand region using pathomorphological and molecular genetic methods.Research Aim. The aim of the study is to study the GSTM1 gene polymorphism (dell) mutation in patients with bronchiectasis in the Samarkand region using pathomorphological and molecular genetic methods.
2. Materials and Methods of Research
The study examined the results of pathomorphological - general histological, histochemical and morphometric examination of bronchi and lung lobes in patients who underwent lobectomy due to bronchiectasis, as well as laboratory results of biopsy (blood) from patients with bronchiectasis to study the polymorphism (dell) mutation of the GSTM1 gene.For this, the patients in our study were divided into 3 observation groups.Group 1 was a control group, which included 12 cases of bronchial and lung biopsy specimens from patients who died of respiratory diseases, and 20 cases of biopsy specimens (blood) from healthy individuals who did not have bronchiectasis to detect the GSTM1 gene polymorphism (dell) mutation.In 2 groups - 34 cases were included, in which patients who underwent lobectomy due to bronchiectasis were treated with biopsies of bronchus and lung lobes.In group 3, blood from 30 patients was used to study the GSTM1 gene polymorphism (dell) mutation in patients with bronchiectasis.Inclusion criteria: To be included in the study, patients with bronchiectasis must be over 18 years of age, have a diagnosis of bronchiectasis confirmed by radiology and high-resolution computed tomography, and have clinical symptoms consistent with bronchiectasis (cough, sputum production, shortness of breath, or respiratory infections) with a clinical diagnosis of J 47 on the ICU.Exclusion criteria: Patients were excluded from the study if they were unable to provide informed consent or if they had active tuberculosis or lung cancer. Patients with cystic fibrosis or pulmonary fibrosis with secondary bronchiectasis were also excluded.Patients in the bronchiectasis groups were required to be clinically stable and not taking antibiotics or oral corticosteroids for 4 weeks prior to study entry. All relevant medical history (comorbidities, current medications, significant past illnesses, surgeries, and diagnostic procedures) was recorded at screening.Bronchiectasis was defined as bronchial dilatation on high-resolution CT, clinical signs, airway obstruction, and a history of smoking. To be included in the “bronchiectasis” cohort, participants had to have a CT scan within the past 5 years that showed evidence of bronchiectasis. In addition, to be included in the bronchiectasis cohort, participants had to have no clinical diagnosis of COPD and no relevant smoking history (<10 pack-years) and/or no airway obstruction as determined by spirometry at screening.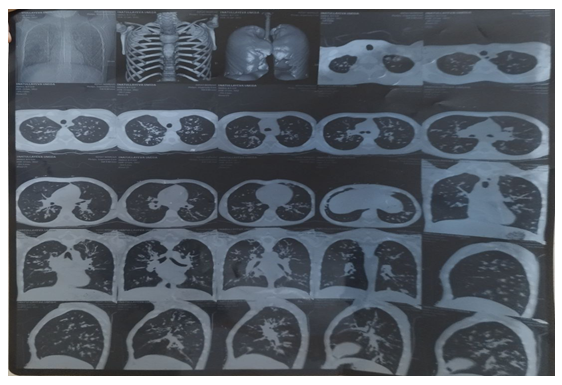 | Figure 1. Patient I.U. is 24 years old. According to the results of high-resolution computed tomography (CT) of the chest, diffuse changes in the bronchial tree are detected in both lungs. In some segmental and subsegmental bronchi, their diameter is larger than the diameter of the adjacent pulmonary arteries. Thickening of the bronchial walls and their clear visualization up to the peripheral sections are noted. |
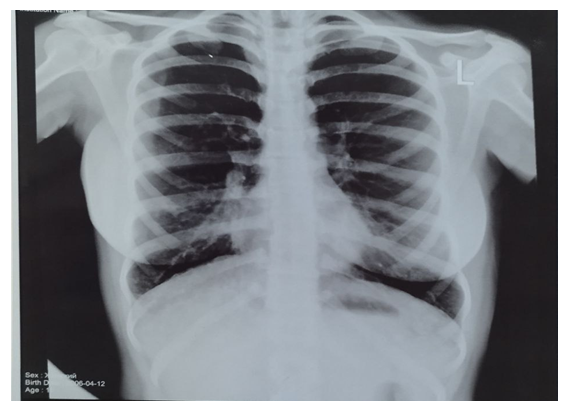 | Figure 2. Patient M.M. is 30 years old. The radiograph shows increased lung volume and deformation in both lung fields, mainly in the lower sections. The coarse and uneven appearance of the bronchovascular pattern indicates a chronic inflammatory process in the bronchial walls |
To confirm the results obtained in the initial group of patients, another 30 patients from the same group who met the same inclusion and exclusion criteria were included in the study. As described below, laboratory results of biopsy (blood) samples from patients with bronchiectasis were analyzed to investigate the presence of the GSTM1 gene polymorphism (dell) mutation in these patients. Patients provided written informed consent.Methods of the study.Pathomorphological studies were conducted using hematoxylin and eosin staining to determine structural changes in the tissues and cells of the bronchi and lung lobes for general histological and histochemical examinations, Van Gieson and Mallory staining to determine the state of their connective tissue collagen fibers and muscle fibers, and Weigert staining to determine the state of elastic fibers.GSTT1 gene polymorphism testing method. GSTT1 gene polymorphism testing was carried out by PCR at “GENMEDICAL” LLC in Samarkand.The first stage involved collecting biological material from patients. Venous blood from patients was collected in EDTA vacuum tubes and stored at a temperature of +4...+8°C for no more than 24 hours before analysis and delivery to the laboratory.EDTA K2 and EDTA K3 are anticoagulants that prevent blood clotting and preserve the formed elements until the time of examination. Purple or light purple tubes containing EDTA are used for general blood analysis, determination of blood group and Rh factor, as well as PCR diagnostics.Stage 2 involved DNA isolation. For this, DNA extraction from whole blood was performed using the "NK-MAGNIT" (Litech) reagent kit according to the manufacturer's instructions. The extracted DNA was stored at +2...+8°C for up to one week or at −20°C for up to one year.Whole blood tubes were centrifuged at 3000 rpm for 5 minutes, then the plasma was removed. The leukocyte fraction was collected from the pellet and subsequently transferred to clean, labeled tubes for nucleic acid isolation.The working mixture was prepared from L2, H and sorbent solutions. 525 μl of the mixture and the sample under test were added to each tube, then incubated at 60°C. Magnetic separation, washing and elution steps with solutions W1 and W2 were performed sequentially. The purified DNA was transferred to a new tube.The DNA amplification was performed in 3 stages. This was done according to the instructions for the 5× ScreenMix, which included water, ScreenMix, and special primers to prepare the master mix.An internal control was performed in stage 4. In this case, the PCR product of cytochrome CYP3A5*3 was used as an internal control (marker) for the GSTT1 gene, and the expected length of the amplicon was 196 bp.In step 5, the gel was prepared using 1× TAE (or TBE) buffer with 2% agarose added. The mixture was heated until completely dissolved, cooled, and ethidium bromide was added. After pouring into the gel box, the comb was installed and left until completely solidified.After the gel was loaded, buffer was added to the electrophoresis chamber, PCR products mixed with loading dye were loaded, and electrophoresis was performed at 150 V for 30-45 minutes.The sixth step was visualization of the results. In this, the electrophoresis results were stained with UView 6× Loading Dye or ethidium bromide and then visualized under ultraviolet light using a gel documentation system.All data were statistically processed. To obtain statistically reliable results, morphometric analysis of histological sections from bronchi and lung sections was performed, for which at least 20 objects were analyzed in several fields of view. Morphometric studies were performed, including measuring the thickness of the bronchial layers and assessing the thickness of their individual layers, measuring the diameter of the bronchial lumen, performing image statistics, and counting cells in different bronchial layers and lung tissue. Histomorphometric analyses were performed using a scanner connected to an Olympus microscope, using the NDP.VIEW2.0, QuPath.0.4.0.url programs. Statistical analysis of the results was performed using the Microsoft Office Excel-2010 program on a Pentium-IV personal computer using Student's t-test.
3. Research Results
When comparing cohorts of patients in all observation groups, bronchiectasis was more common in women, in 11 cases (66.33%), and in men, in 19 cases (36.66%) (Fig. 3).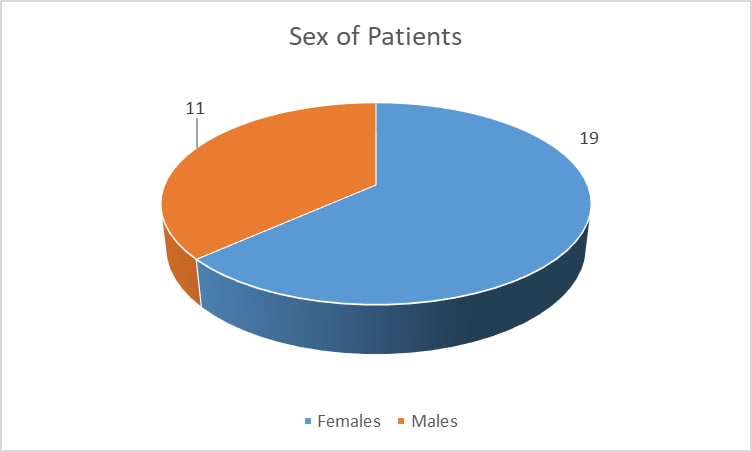 | Figure 3. Sex-related incidence of bronchiectasis in a follow-up group |
When comparing the number of patients who applied in the Samarkand region by place of residence, it was found that the highest number of cases were found in Samarkand city (26.67%), followed by Urgut district (20%), and the lowest number of cases were found in Pakhtachi, Jamboy, Payariq and Pastdargam districts (3.33%).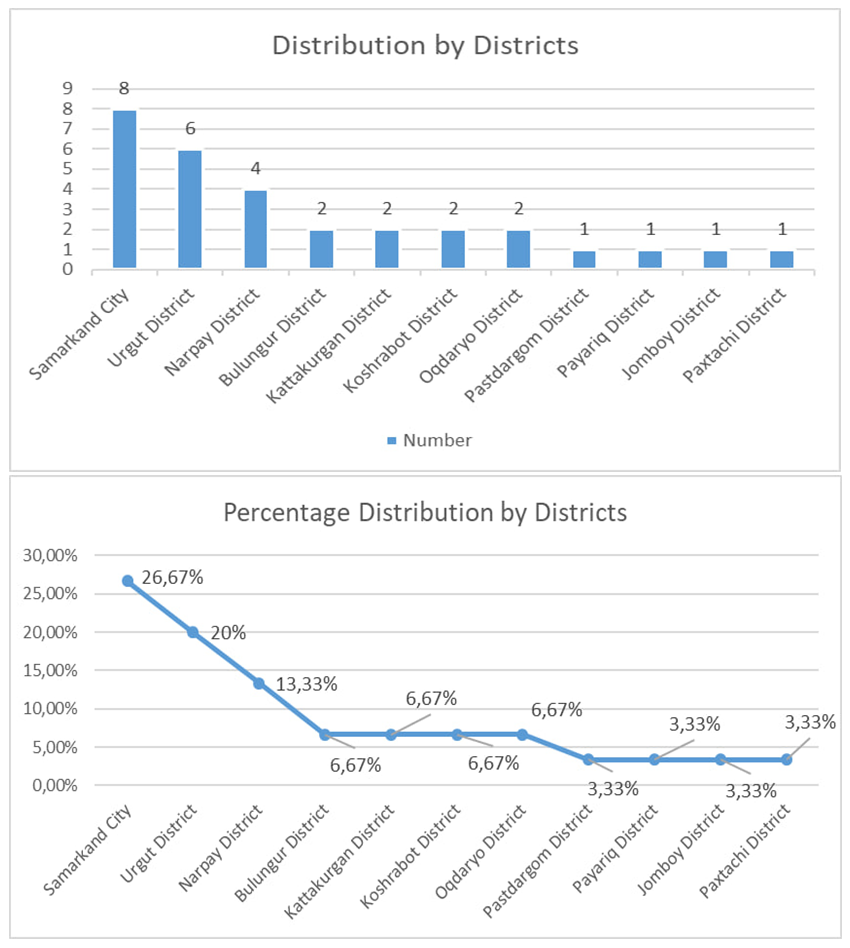 | Figure 4. District-level incidence of bronchiectasis in Samarkand region |
According to the age indicators of the patients, the average age of bronchiectasis in this research group was 53.2 ± 2.1 years.Blood samples were taken from 30 patients for genetic testing. Of these, 19 were women (63.33%) and 11 were men (36.67%). When GSTM1 gene dell mutations were tested, 9 out of 19 (30%) women had the dell mutation, and 10 (33.33%) had a positive (+) result. Among men, the dell mutation was found in 4 (13.33%) patients, and 7 (23,33) had a positive (+) result. This indicates that the prevalence of bronchiectasis is slightly higher in women than in men (Fig. 5).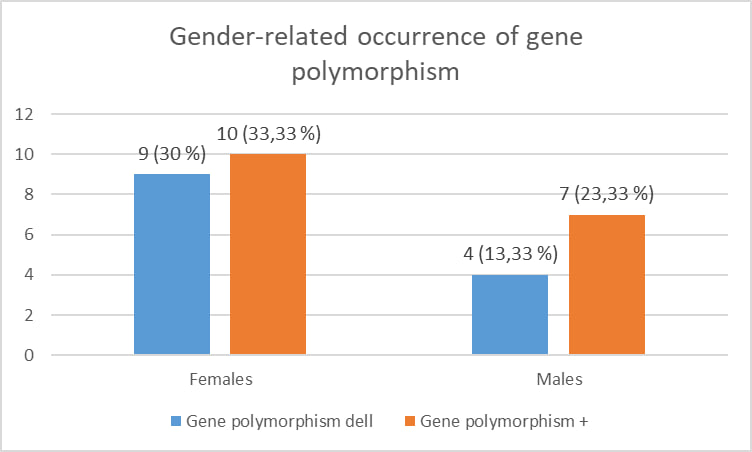 | Figure 5. Gender-related occurrence of gene polymorphism |
The study of histological preparations revealed morphological polymorphism of the bronchial epithelium. The most common type of epithelium was multi-row ciliated epithelium. It is characterized by an increase in multi-row formation due to epithelial cell hyperplasia. The cilia of prismatic epithelial cells are not expressed on the entire surface of the epithelium, but in places where they are present, they are glued together, hyperplasia of goblet cells is observed. Small lymphocytes are also located between the epithelial cells, penetrating the surface of the epithelium. It is noteworthy that the subepithelial membrane is slightly thickened.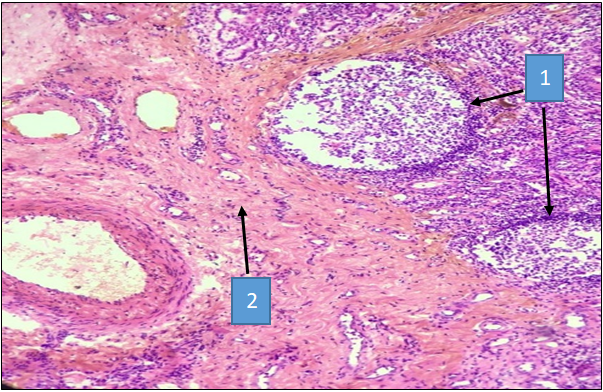 | Figure 6. Bronchiectasis. Lung tissue of a 48-year-old patient. The lung tissue shows large focal lymphocytic infiltration (1), areas of destruction and sclerosis (2). Hematoxylin and eosin staining. Cat. X40 |
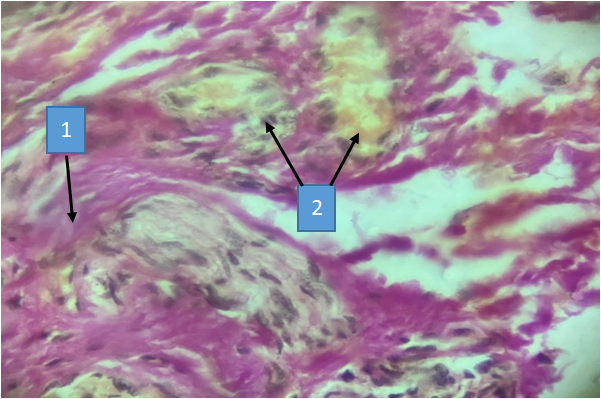 | Figure 7. Bronchiectasis. Lung biopsy of a 52-year-old patient. Sclerosis (1) and vascularization (2). Hematoxylin and eosin staining. Cat. X40 |
The formation of mucosal insufficiency occurs as a result of an active inflammatory process in the bronchi, a violation of the structure of the bronchial mucosa, an increase in the viscosity of bronchial secretions and a decrease in the frequency of ciliary beats in the ciliated epithelium. In chronic obstruction, epithelial desquamation, squamous metaplasia and loss of cilia by ciliated cells are observed (Fig. 8). In areas of epithelial metaplasia, foci of sclerosis and fuchsinophilia of collagen fibers are observed (Fig. 9).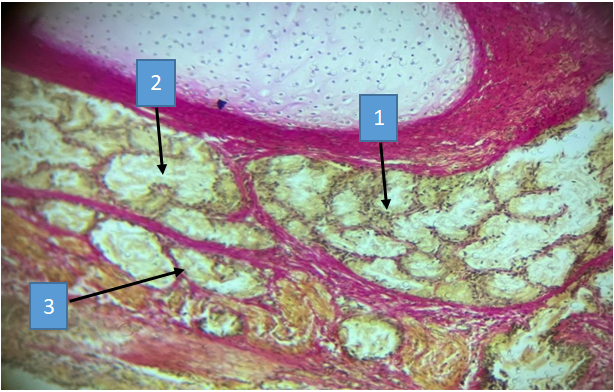 | Figure 8. Bronchiectasis disease. A 47-year-old patient with a small-caliber bronchus fragment. Serous glands in the bronchi are hypertrophied (1), in a state of hypersecretion (2), and most of them show accumulation of secretion and destruction (3). Staining according to Van-Gieson |
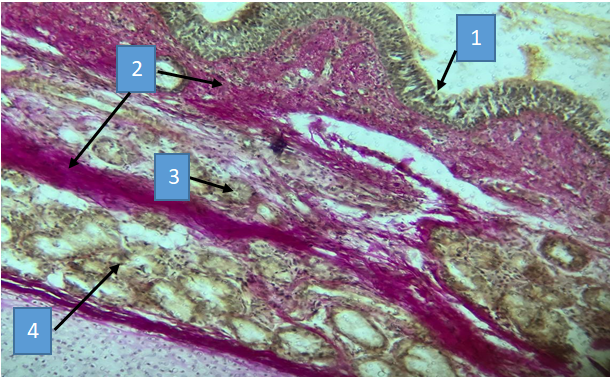 | Figure 9. Bronchiectasis disease. A 47-year-old patient with a small-caliber bronchus fragment. Hyperplasia of the bronchial mucosa (1), sclerosis of connective tissue and muscle fibers of the submucosa (2), destruction and necrosis of serous and protein glands (3), hypersecretion (4). Staining according to Van-Gieson. Cat. X40 |
The second stage of detoxification includes genes GSTM1, GSTT1, GSTP1, which ensure the conversion of xenobiotics into water-soluble compounds, which are excreted from the body through the liver and kidneys. Analysis of polymorphisms in the genes of this system allows us to predict the rate of metabolism of toxic substances.Glutathione-S-transferases (GSTs) are enzymes involved in the neutralization of carcinogens, drugs, and other toxic compounds by binding them to glutathione. They play an important role in cell protection and may contribute to drug resistance in tumors.According to the goals and objectives of our study, the GSTM1 gene polymorphism (dell) mutation was studied in patients with bronchiectasis.The GSTM1 gene in the control group is characterized by the presence of (dell) genotypes, in which the enzymes are not synthesized. Such variants are associated with an increased risk of gynecological, somatic, especially bronchial asthma and chronic bronchitis, as well as oncological and liver diseases. Genetic testing allows for an assessment of individual risks and the selection of optimal treatment tactics.
4. Conclusions
Bronchiectasis is more common in the city of Samarkand, Urgut and Narpay districts. The disease is seasonal, reaching its peak in the spring and autumn seasons.For the study, the results of pathomorphological - general histological, histochemical and morphometric examination of bronchi and lung lobes of 48 patients who underwent lobectomy due to bronchiectasis, as well as the results of laboratory biopsies (blood) of 50 patients with bronchiectasis to study the (dell) mutation of the GSTM1 gene polymorphism were studied.
References
| [1] | Gupta A.K., Lodha R., Kabra S.K. Non Cystic Fibrosis Bronchiectasis. Indian J Pediatr 2015; 82(10): 938–44. DOI: 10.1007/s12098-015-1866-4. |
| [2] | Krasilova E.V., Bashkina O.A., Matsakova D.V., Samotrueva M.A. Respiratory diseases in children // Astrakhan Medical Journal. 2022. Vol. 17, No. 3. pp. 13–22. doi: 10.48612/agmu/2022.17.3.13.22. |
| [3] | Aksamit T.R., O’Donnell A.E., Barker A.F., et al. Adult patients with bronchiectasis: a first look at the US Bronchiectasis Research Registry. Chest. 2017; 151(4): 982-992. doi:10.1016/j.chest.2016.10.062. |
| [4] | Baranov V.S., Aylamazian E.K. (edrs) Evaluation of inherited predisposition to some common ostetritian disorders. ... Genetic Pass – Background of Personal - Predictive Medicine” SPb., “N-L” 2009. |
| [5] | Korytina G. F., Akhmadishina L. Z., Kochetova O. V., Burdyuk O. V., Zagidullin Sh. Z., Viktorova T. V. Association of polymorphic loci of genes encoding toxic compounds and free radicals with the development and progression of chronic obstructive pulmonary disease // Medical Genetics. 2013. Vol. 12, No. 8. Pp. 32–42. |
| [6] | Faramawy M. M., Mohammed T. O., Hossaini A. M., Kashem R. A., Abu Rahma R. M. Genetic polymorphism of GSTT1 and GSTM1 and susceptibility to chronic obstructive pulmonary disease (COPD) // J. Crit. Care. 2009. Vol. 24, No. 3. R. 7−10. https://doi:10.1016/j.jcrc.2009.06.004]. |
| [7] | Mehrotra S., Sharma A., Kumar S., Kar P., Sardana S., J K Sharma. Polymorphism of glutathione S-transferase M1 and T1 gene loci in COPD. Int J Immunogenet. 2010 Aug; 37(4): 263–267. doi:10.1111/j.1744-313X.2010.00918.x. |
| [8] | Lakhdar R.B., Denden S., Knani J., Leban N., Daimi H., Hassine M., Lefranc G., Ben Chibani J., Haj Khelil A. |
| [9] | Baranov V.S., Ivaschenko T.E., Lavrova O.V., Fedoseyev G.B. Some molecular genetic aspects of the etiopathogenesis of atopic bronchial asthma // Medical Genetics. 2008. Vol. 7, No. 10 (76). Pp. 3–13. |
| [10] | Motahari P, Neshanifard N. Glutathione S-transferase M1 and T1 null polymorphisms and risk of oral submucous fibrosis: a meta-analysis of South Asian populations. BMC Oral Health. 2025; 25: 1108. doi:10.1186/s12903-025-06497-y. |
| [11] | Zaman SF, Samms-Vaughan M, Saroukhani S, Bressler J, Hessabi M, Grove ML, Shakspeare-Pellington S, Loveland KA. Factors associated with blood mercury concentrations and their interactions with three glutathione S-transferase genes (GSTT1, GSTM1, and GSTP1): an exposure assessment study of typically developing Jamaican children. BMC Pediatr. 2024; 24: 14. doi:10.1186/s12887-023-04452-w. PMID: 38178055. |
| [12] | Rahbar MH, Samms-Vaughan M, Zhao Y, Saroukhani S, Bressler J, Hessabi M, Grove ML, Shakespeare-Pellington S, Loveland KA. Interactions between Environmental Factors and Glutathione S-Transferase (GST) Genes with Respect to Detectable Blood Aluminum Concentrations in Jamaican Children. Genes. 2022; 13(10): 1907. doi:10.3390/genes13101907. PMID: 36292793. |






 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML



