-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 485-488
doi:10.5923/j.ajmms.20261602.22
Received: Jan. 9, 2026; Accepted: Feb. 1, 2026; Published: Feb. 3, 2026

The Role of GSTT1 Gene Polymorphism in Pneumonia with Unfavorable Outcomes in Children
Aminova N. A.
Department of 1st Pediatrics and Neonatology, Samarkand State Medical University, Samarkand, Uzbekistan
Correspondence to: Aminova N. A., Department of 1st Pediatrics and Neonatology, Samarkand State Medical University, Samarkand, Uzbekistan.
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Pneumonia remains one of the leading causes of severe morbidity and adverse outcomes in children. Oxidative stress and the effectiveness of antioxidant defense systems play a significant role in the pathogenesis of complicated disease progression. The aim of the study was to determine the role of GSTT1 gene polymorphism in the development of adverse outcomes of pneumonia in children. A total of 133 children aged 1 to 12 years were examined, including patients with pneumonia with unfavorable and favorable outcomes, as well as a group of healthy children. GSTT1 gene polymorphism was determined by polymerase chain reaction. It was found that the GSTT1 deletion variant (null) was significantly more common in children with an unfavorable outcome of pneumonia and was associated with an increased risk of severe disease. The data obtained confirm the prognostic significance of GSTT1 in pneumonia in children.
Keywords: Pneumonia, Children, Adverse outcome, Polymorphism, GSTT1, Oxidative stress
Cite this paper: Aminova N. A., The Role of GSTT1 Gene Polymorphism in Pneumonia with Unfavorable Outcomes in Children, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 485-488. doi: 10.5923/j.ajmms.20261602.22.
Article Outline
1. Introduction
- Pneumonia remains one of the leading causes of preventable child mortality and severe morbidity worldwide. According to WHO data, in 2019, pneumonia claimed the lives of 740,180 children under the age of 5, accounting for 14% of all causes of death in this age group and remaining the leading infectious cause of death in children [1]. UNICEF estimates that pneumonia kills more than 700,000 children under the age of 5 every year, with the greatest burden falling on countries with limited health resources [2]. Current clinical guidelines emphasize that the risk of severe disease and hospitalization is highest in young children, and clinical assessment of severity requires a comprehensive approach that takes into account respiratory disorders, systemic inflammatory response, and complications [3]. Even when following the standards of care outlined in national and international guidelines, the outcome of the disease in individual children remains difficult to predict [4,5]. This is due to the polyetiology of pneumonia, variability in immune response, differences in the spectrum of pathogens and comorbidity, as well as the contribution of host factors, including genetically determined susceptibility to inflammatory lung damage.Clinical data in children with severe community-acquired pneumonia show signs of pro-oxidant activation and biomolecule damage, which is considered one of the links in the formation of an unfavorable course [6]. Consequently, antioxidant defense systems (including glutathione-dependent detoxification pathways) are pathogenetically significant, and individual differences in their activity may potentially determine the outcome. In this regard, the family of glutathione S-transferase (GST) enzymes is of particular interest, as they ensure the conjugation of reduced glutathione with reactive metabolites and products of lipid peroxidation, reducing toxicity and limiting tissue damage. The topic is further relevant because the role of GST-dependent mechanisms is significant not only for the host organism, but also in the host-pathogen interaction system. For example, it has been shown that Streptococcus pneumoniae uses glutathione metabolism to protect against oxidative stress and maintain virulence, which highlights the importance of glutathione-dependent processes in the context of pneumococcal infection [7]. Consequently, “bottlenecks” in glutathione metabolism and detoxification can influence the dynamics of inflammation and the likelihood of an unfavorable outcome.Clinical and genetic studies conducted over the years indicate that polymorphisms in detoxification genes (including GSTM1/GSTT1/GSTP1) contribute to susceptibility to pneumonia and the risk of complications. It has been shown that variability in phase I–II detoxification genes, including GSTT1, may be associated with the frequency of complications in pneumonia of various origins, supporting the concept of a significant contribution of host genetic factors [8]. Individual studies also consider xenobiotic detoxification genes as risk factors for the development of pneumonia in hospitalized patients, emphasizing the role of “detox genes” in susceptibility to inflammatory lung damage [9]. Although some of these studies were conducted in adult cohorts, they are important from a methodological point of view: they demonstrate the fundamental possibility of identifying genetic determinants of the course of pneumonia and set the direction for pediatric studies with the endpoints of “adverse outcome” and “severity.”Thus, studying the role of GSTT1 polymorphism in pneumonia with adverse outcomes in children is relevant for a number of reasons. Obtaining evidence of a link between GSTT1 polymorphism with an unfavorable outcome can broaden our understanding of the mechanisms of severe pneumonia in children and lay the foundation for the introduction of genetic predictors into a comprehensive system for predicting and preventing complications.Research Objective: to determine the role of GSTT1 gene polymorphism in the development of adverse outcomes of pneumonia in children.
2. Materials and Methods
- The study was conducted at the Samarkand Regional Children's Multidisciplinary Medical Center and the 3rd Family Polyclinic. Children aged 1 to 12 years were included in the study.Depending on the clinical course and outcome of pneumonia, all examined patients were divided into three groups. Group I (n=31) included children with pneumonia with an unfavorable outcome. Group II (n=83) consisted of children with pneumonia with a favorable outcome. The control group consisted of 19 practically healthy children of comparable age.The inclusion criteria were clinically and radiologically confirmed pneumonia and informed consent from the parents or legal representatives of the child. Children with congenital malformations, primary immunodeficiencies, and severe concomitant pathology were excluded from the study.Venous blood collected in vacuum tubes with EDTA was used for molecular genetic analysis. GSTT1 gene polymorphism was determined by polymerase chain reaction. DNA was extracted from whole blood using a standard reagent kit, and the target gene region was amplified using a commercial PCR mix and specific primers. The correctness of the reaction was checked using an internal amplification control.
3. Research Results
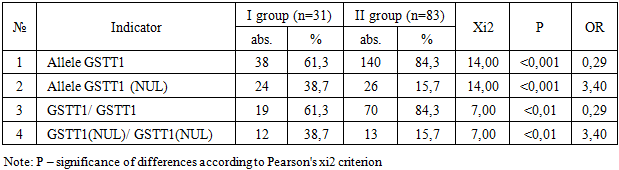
- Table 1 presents a comparative analysis of the distribution of alleles and genotypes of the GSTT1 gene in children with pneumonia with unfavorable (group I, n=31) and favorable outcomes (group II, n=83).
|
|
4. Discussion
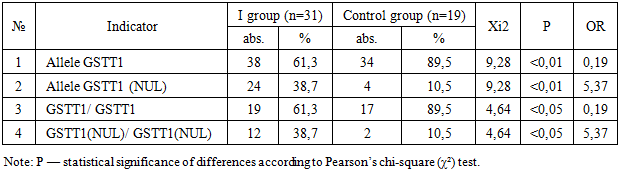
- The data obtained in this study indicate the significant role of GSTT1 gene polymorphism in the development of adverse outcomes of pneumonia in children. It was found that the deletion variant of the GSTT1 gene (null) was significantly more common in patients with an unfavorable outcome of the disease compared to both the group of children with a favorable course of pneumonia and the control group of practically healthy children, which is confirmed by statistically significant differences according to the χ² criterion and high odds ratios.In group I, the frequency of the GSTT1 (null) allele and the corresponding homozygous genotype was significantly higher than in group II (OR=3.38; P<0.001) and in the control group (OR=5.37; P<0.01). At the same time, the presence of the functional GSTT1 allele was associated with a favorable outcome of pneumonia and was considered a relative protective factor. These results are consistent with the concept of the key role of glutathione-S-transferases in protecting tissues from oxidative damage during infectious and inflammatory processes [10,11].It is known that severe pneumonia is accompanied by pronounced oxidative stress caused by the activation of neutrophils and macrophages, increased production of active forms of oxygen and nitrogen, and an imbalance in antioxidant systems [12]. Glutathione S-transferases, including GSTT1, are involved in the inactivation of lipid peroxidation products and toxic metabolites, thereby limiting damage to cell membranes and the endothelium of the alveolar-capillary barrier [13]. In the absence of a functional GSTT1 gene, these protective mechanisms are weakened, which can contribute to a more severe inflammatory response, the development of respiratory failure, and other complications. The results of this study are consistent with data from other clinical and genetic studies showing an association between detoxification gene polymorphisms (GSTM1, GSTT1, GSTP1) and the risk of complicated pneumonia and adverse outcomes of infectious diseases of the respiratory system [14]. Thus, a number of studies have demonstrated that carriage of GST deletion variants is associated with a more pronounced inflammatory response, increased duration of illness, and increased frequency of complications, especially in young children [15].It is important to note that the identified association has not only pathogenetic but also clinical significance. Genetic markers, unlike traditional laboratory indicators, are stable and do not depend on the stage of the disease. This allows GSTT1 polymorphism to be considered as a potential component of an early risk stratification system in children with pneumonia, especially upon admission to the hospital [16]. In the future, combining clinical and laboratory criteria for severity with molecular genetic markers may improve the accuracy of prognosis and contribute to the personalization of treatment tactics.At the same time, it should be borne in mind that the development of an unfavorable outcome of pneumonia is a multifactorial process in which, along with genetic predisposition, age, premorbid background, pathogen virulence, and the timeliness and adequacy of therapy play an important role. Therefore, it is advisable to consider GSTT1 polymorphism not as an isolated predictor, but as one of the elements of a comprehensive prognostic model.
5. Conclusions
- Thus, the results of the study confirm the pathogenetic significance of the GSTT1 gene in the formation of adverse outcomes of pneumonia in children and justify the feasibility of further research in this direction with an expanded sample size and the inclusion of additional genetic and biochemical markers of oxidative stress.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML