-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 475-478
doi:10.5923/j.ajmms.20261602.19
Received: Jan. 6, 2026; Accepted: Jan. 22, 2026; Published: Feb. 3, 2026

Morphometric Characteristics and Morphological Types of Pterygium
Rajabov Hamid Rashid ugli, Karimov Rasulbek Xasanovich
Urgench Branch of the Tashkent Medical Academy, Khorezm Regional Center of Eye Microsurgery, Urgench, Uzbekistan
Correspondence to: Rajabov Hamid Rashid ugli, Urgench Branch of the Tashkent Medical Academy, Khorezm Regional Center of Eye Microsurgery, Urgench, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Pterygium is a degenerative–proliferative ocular surface disorder characterized by fibrovascular tissue growth onto the cornea and a high rate of postoperative recurrence. Despite advances in surgical management, recurrence remains a major clinical challenge, underscoring the importance of detailed morphometric evaluation of pterygium tissue. To investigate and compare the morphometric characteristics of fibrovascular tissue components in different morphological variants of pterygium and to assess their potential clinical significance. This retrospective morphometric study included biopsy specimens from 78 patients surgically treated for pterygium at the Khorezm Regional Center of Eye Microsurgery between 2022 and 2024. Histological sections were stained with hematoxylin and eosin and digitized using a NanoZoomer scanner. Quantitative morphometric analysis was performed with QuPath-0.5.0 software. The assessed parameters included the area occupied by fibroblasts, collagen fibers, blood vessels, inflammatory infiltrates, and interstitial tissue. Statistical analysis was conducted using variation statistics, with significance set at P < 0.05.
Keywords: Pterygium, Morphometry, Fibrovascular tissue, Collagen fibers, Fibroblasts, Recurrence
Cite this paper: Rajabov Hamid Rashid ugli, Karimov Rasulbek Xasanovich, Morphometric Characteristics and Morphological Types of Pterygium, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 475-478. doi: 10.5923/j.ajmms.20261602.19.
Article Outline
1. Introduction
- Pterygium (from the Greek “pterygion”) is a disease characterized by the growth of a fibrovascular membrane from the conjunctiva onto the surface of the cornea along the horizontal meridian. Its prevalence varies worldwide among different populations, ranging from 0.7% to 31%. This condition is more commonly observed in peri-equatorial regions located between 37° north and south of the equator, where high temperatures and dry climatic conditions predominate [1,2].Pterygium represents squamous metaplasia of the epithelium and is characterized by fibroblast proliferation, infiltration of inflammatory cells, neovascularization, and remodeling of the extracellular matrix. The disease has degenerative features, including degradation of Bowman’s membrane, thinning of the basement membrane, and elastoid and hyaline degeneration. It is also distinguished by epithelial hyperplasia and a high rate of recurrence after surgical excision [3,4].At present, the main method of pterygium treatment is surgical intervention; according to the literature, the average postoperative recurrence rate is approximately 40% [5]. The high frequency of recurrences highlights the necessity for an in-depth study of the morphological characteristics of pterygium.The aim of this study is to investigate the morphometric characteristics of the fibrovascular tissue structure in different morphological variants of pterygium (proliferative, fibromatous, and atrophic-sclerotic) and to assess their clinical significance.
2. Literature Review
- Pterygium is a common ocular surface disorder with a complex and multifactorial pathogenesis. Epidemiological studies have consistently demonstrated a strong association between pterygium development and chronic exposure to ultraviolet (UV) radiation, high ambient temperatures, dry climates, wind, and airborne irritants [1,2]. These environmental factors are particularly prevalent in peri-equatorial regions, where the incidence of pterygium reaches its highest levels.Traditionally, pterygium was regarded as a degenerative condition; however, accumulating evidence suggests that it represents a degenerative–proliferative disorder with tumor-like biological behavior [3,4]. Histologically, pterygium is characterized by squamous metaplasia of the conjunctival epithelium, epithelial hyperplasia, fibroblast proliferation, neovascularization, inflammatory cell infiltration, and extensive remodeling of the extracellular matrix [6,9]. Degenerative changes such as Bowman’s membrane fragmentation, elastoid degeneration, and hyaline transformation further support the complexity of this pathology [9].Fibroblasts play a central role in the progression of pterygium. Activated fibroblasts demonstrate increased proliferative activity, enhanced synthesis of extracellular matrix components, and resistance to apoptosis [5]. Dushku and Reid reported abnormal expression of the tumor suppressor protein p53 in pterygium tissues, suggesting dysregulation of cell cycle control mechanisms and contributing to uncontrolled fibroblast proliferation and recurrence [10]. These findings reinforce the concept that pterygium shares biological features with premalignant lesions.Morphological heterogeneity of pterygium has been widely documented in the literature. Several authors classify pterygium into proliferative, fibromatous, and atrophic-sclerotic variants based on histopathological features and clinical behavior [8]. The proliferative variant is characterized by high cellular density, active fibroblast proliferation, pronounced vascularization, and inflammatory infiltration, which clinically corresponds to rapid growth and a high risk of postoperative recurrence [3,7]. In contrast, the fibromatous variant exhibits dense, coarse collagen bundles with reduced cellular activity, representing a relatively stable or quiescent form of the disease [6]. The atrophic-sclerotic variant demonstrates minimal fibroblast activity, predominant sclerotic changes, and low metabolic activity [9].Despite extensive descriptive histological studies, quantitative morphometric investigations of pterygium tissue remain limited. Conventional qualitative assessments are insufficient for objectively comparing tissue components among different morphological variants [4]. Recent advances in digital pathology, including whole-slide imaging and image analysis software, have enabled precise morphometric evaluation of fibrovascular tissue, allowing objective measurement of fibroblasts, collagen fibers, vascular structures, and inflammatory components [7].Postoperative recurrence remains a major challenge in pterygium management, with reported rates ranging from 10% to 40% depending on surgical technique and adjuvant therapy [5]. Increasing evidence suggests that recurrence is strongly influenced by the intrinsic morphological and cellular characteristics of the excised tissue rather than surgical factors alone [8]. High fibroblast density, active neovascularization, and poor collagen organization have been identified as key predictors of recurrence, highlighting the importance of preoperative and postoperative risk stratification.Therefore, morphometric analysis represents a valuable approach for understanding the biological behavior of pterygium. Quantitative assessment of fibrovascular tissue components provides objective criteria for differentiating morphological variants, predicting recurrence risk, and selecting individualized treatment strategies, including the potential use of adjuvant anti-VEGF or antifibrotic therapies [5,10]. Comprehensive morphometric studies are essential for improving long-term surgical outcomes and optimizing patient management in pterygium.
3. Materials and Methods
- Study Material.The study examined biopsy specimens from 78 patients diagnosed and treated for pterygium at the Khorezm Regional Center of Eye Microsurgery between 2022 and 2024. Among the patients, 35 (44.9%) were male and 43 (55.1%) were female, with ages ranging from 21 to 69 years.Histological Examination.Biopsy specimens were fixed in a 10% neutral formalin solution and embedded in paraffin blocks. Sections 4–5 µm thick were prepared using a microtome and stained with hematoxylin and eosin. The stained histological slides were examined using a ZEISS Primo Star light microscope (Germany) at magnifications of ×40, ×100, and ×400.Morphometric Analysis.For morphometric analysis, 10–15 sections were prepared from each specimen. Histological slides were digitized using a NanoZoomer scanner (REF C13140-21, Hamamatsu Photonics, Japan). The obtained images were processed and analyzed using QuPath-0.5.0 software.The following parameters were evaluated as morphometric indices: the area occupied by fibroblasts, the area of collagen fibers, the area of blood vessels, the area of inflammatory infiltrate, and the area of interstitial tissue.Statistical Analysis.The obtained data were analyzed using methods of variation statistics. The arithmetic mean (M) and standard error (±m) were calculated. Statistical significance was assessed at a level of P < 0.05.
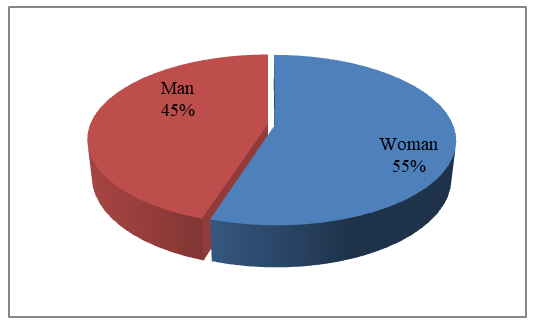 | Figure 1. Distribution of pterygium according to gender |
 (male) = 35/78 = 0.448795% confidence interval (Wilson): 0.3343 – 0.5589Distribution of pterygium prevalence by age groups (percentages). A detailed analysis of our study showed that the highest prevalence was observed in the 51–60 age group, with 23 cases (30%). This age period is characterized by high occupational activity, during which initial changes in the visual system begin to appear.Other age groups with a relatively high prevalence included individuals aged 41–50 years, with 18 cases (23%), and those aged over 60 years, with 22 cases (28%). The 30–40 age group accounted for 14 cases (18%), while the 20–30 age group included only 1 case (1%). These groups also represent periods of active lifestyle and sustained work activity.The overall prevalence in the 40–70 age range amounted to 63 cases (81%). During this period, aging-related changes in the visual system begin to develop; however, functional and occupational activity generally remains high.The lowest prevalence was observed in the 21–30 age group, with 1 case (1%), and in the 15–20 age group, where no cases (0%) were recorded. This finding suggests that in these age groups, either physical activity is relatively low (in older individuals) or protective behaviors and caution are more pronounced (in younger individuals).
(male) = 35/78 = 0.448795% confidence interval (Wilson): 0.3343 – 0.5589Distribution of pterygium prevalence by age groups (percentages). A detailed analysis of our study showed that the highest prevalence was observed in the 51–60 age group, with 23 cases (30%). This age period is characterized by high occupational activity, during which initial changes in the visual system begin to appear.Other age groups with a relatively high prevalence included individuals aged 41–50 years, with 18 cases (23%), and those aged over 60 years, with 22 cases (28%). The 30–40 age group accounted for 14 cases (18%), while the 20–30 age group included only 1 case (1%). These groups also represent periods of active lifestyle and sustained work activity.The overall prevalence in the 40–70 age range amounted to 63 cases (81%). During this period, aging-related changes in the visual system begin to develop; however, functional and occupational activity generally remains high.The lowest prevalence was observed in the 21–30 age group, with 1 case (1%), and in the 15–20 age group, where no cases (0%) were recorded. This finding suggests that in these age groups, either physical activity is relatively low (in older individuals) or protective behaviors and caution are more pronounced (in younger individuals).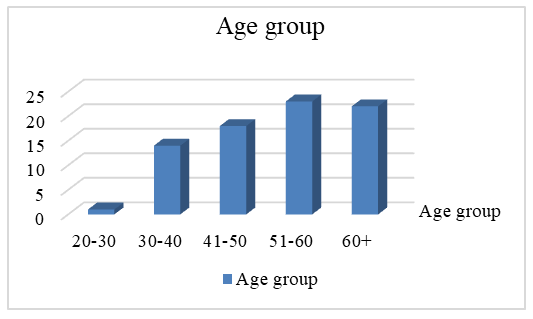 | Figure 2. Distribution by age groups |
4. Results
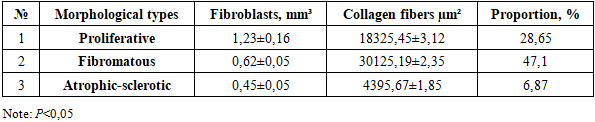
- Among the 89 examined cases, the morphological variants of pterygium were distributed as follows: proliferative type – 32 cases (36.0%), fibromatous type – 35 cases (39.3%), and atrophic-sclerotic type – 22 cases (24.7%).The results of the multiplex confocal morphometric analysis are presented in Table 1.
|
5. Discussion
- The obtained results demonstrated significant differences in the fibrovascular tissue composition among the various morphological variants of pterygium. In the proliferative variant, a high proliferation of fibroblasts and a relatively low amount of collagen fibers were observed, which explains the clinical aggressiveness of this variant.In the fibromatous variant, a high content of collagen fibers (47.1%) and relatively low fibroblast activity were identified. These findings confirm that this variant represents the most “stable” or “quiescent” form of pterygium. Morphometric analysis showed that in the fibromatous type, collagen fibers occupy 47.1% of the measured area, which in clinical practice indicates a lower likelihood of recurrence after treatment.In the atrophic-sclerotic variant, the overall metabolic activity of the tissue was low, and sclerotic changes predominated. However, fragmentation of collagen fibers within the loose connective tissue contributed to the formation of mononuclear infiltrates in foci of chronic inflammation.In the proliferative variant, the area of fibroblast proliferation accounted for 28.65%, which indicates the potential need for post-treatment anti-VEGFR-1 targeted therapy. This approach may improve patients’ quality of life.Thus, morphometric analysis allows for quantitative assessment of tissue composition in different morphological variants of pterygium. These data are important for selecting treatment strategies, determining the necessity of additional therapy in the postoperative period, and predicting the risk of recurrence.
6. Conclusions
- 1. Morphometric analysis revealed distinct quantitative differences in fibrovascular tissue composition among morphological variants of pterygium. These findings provide objective criteria for assessing disease activity, may assist in selecting individualized treatment strategies, and are valuable for predicting postoperative recurrence risk. 2. Pterygium predominantly occurs in individuals aged 41–70 years, accounting for the majority of cases. The highest prevalence was observed in the 41–50, 51–60, and 61–70 age groups. In younger adults (21–30 years), the condition was relatively uncommon. The absence of data for the 15–20 and 70–80 age groups limits the completeness of the analysis.3. Among the morphological variants of pterygium, fibroblast activity in the fibrovascular tissue was highest in the proliferative type (1.23 ± 0.16 mm³) and lowest in the atrophic-sclerotic type (0.45 ± 0.05 mm³).4. In the fibromatous type, the area occupied by dense fibrous connective tissue measured 30,125.19 ± 2.35 µm², accounting for 47.1% of the measured surface, which was the highest value compared to other variants.5. Morphometric data serve as an important criterion for selecting treatment strategies and predicting the risk of recurrence in different morphological variants of pterygium.6. In the proliferative variant, post-treatment anti-VEGFR therapy is recommended, whereas conservative monitoring is advised for the fibromatous variant.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML