-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 470-474
doi:10.5923/j.ajmms.20261602.18
Received: Jan. 2, 2026; Accepted: Jan. 23, 2026; Published: Feb. 3, 2026

Premalignant Pathomorphology of the Liver in Chronic Hepatitis C: Clinicopathological and Immunohistochemical Correlations
Abdullaev O. R.
Department of Pathomorphology, Urgench State Medical Institute, Urgench, Uzbekistan
Correspondence to: Abdullaev O. R., Department of Pathomorphology, Urgench State Medical Institute, Urgench, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Chronic hepatitis C (CHC) remains an important etiological factor for liver cirrhosis and hepatocellular carcinoma (HCC). Premalignant morphological changes in the liver are mediated by progressive fibrosis, necroinflammatory activity, immune-cell infiltration, and activation of Kupffer cells and hepatic stellate cells [1–4]. Aim: To evaluate premalignant pathomorphological changes in the liver of patients with chronic hepatitis C and to determine relationships between histological activity, fibrosis stage, and immunohistochemical markers of inflammation and fibrogenesis. Methods: A retrospective cohort analysis of liver biopsy specimens from 60 adult patients with serologically confirmed CHC was performed. Histological grading and staging were assessed using the Desmet/METAVIR-based approaches (fibrosis F0–F4) and the IGA activity score. In 20 cases, immunohistochemistry (IHC) for Ki‑67 and smooth muscle actin (SMA) was conducted; staining intensity and percentage of positive cells were assessed semi‑quantitatively. Morphometric analysis employed a 100‑point grid in 20 high‑power fields per case. Statistical testing used Student’s t‑test and Pearson correlation (p<0.05). Results: Fibrosis stages were distributed as follows: F0–F1 (16.6%), F2 (26.6%), F3 (33.3%), and F4/cirrhosis (23.3%). Higher inflammatory activity was associated with interface hepatitis, lobular necrosis, and dense portal infiltration. A moderate positive correlation was observed between fibrosis stage and IGA score (r=0.61, p<0.05). Ki‑67 expression was predominantly moderate (85.22%), with expansion of Ki‑67‑positive stromal/mesenchymal cells in advanced fibrosis. SMA expression increased in periportal and septal zones, reflecting stellate cell activation and fibrogenesis [5–8]. Conclusion: CHC is characterized by a spectrum of premalignant liver changes including progressive fibrosis, increasing necroinflammatory activity, immune-cell infiltration, and activation of mesenchymal components. Integrating routine histology with IHC markers (Ki‑67, SMA) can improve HCC risk stratification and support individualized surveillance strategies [2,3,7,9,19].
Keywords: Chronic hepatitis C, Liver fibrosis, Hepatocellular carcinoma, Premalignant pathology, Ki‑67, SMA, Kupffer cells, Immunohistochemistry
Cite this paper: Abdullaev O. R., Premalignant Pathomorphology of the Liver in Chronic Hepatitis C: Clinicopathological and Immunohistochemical Correlations, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 470-474. doi: 10.5923/j.ajmms.20261602.18.
1. Introduction
- Chronic hepatitis C (CHC) is one of the leading causes of progressive liver fibrosis, cirrhosis, and hepatocellular carcinoma (HCC) worldwide [1,2,19]. The transition from chronic inflammation to malignant transformation is driven by a long-term interplay of necroinflammatory activity, immune-cell infiltration, stellate cell activation, and extracellular matrix remodeling [3,4,22]. Histopathological evaluation of CHC using standardized scoring systems (including METAVIR/Desmet-based approaches) remains an essential tool for estimating fibrosis stage, inflammatory activity, and associated parenchymal alterations such as steatosis and cholestasis [4,6,20]. In addition, immunohistochemical (IHC) methods expand diagnostic and prognostic capacity by assessing proliferative activity (Ki‑67), myofibroblast/stellate cell activation (SMA), and immune-cell populations (e.g., CD8+ lymphocytes) [5,7,8,18]. However, region-specific clinicopathological datasets remain limited in some populations, including Central Asia. This study provides a combined histological and immunohistochemical characterization of premalignant changes in CHC liver biopsies in a regional cohort.
2. Materials and Methods
- Study design: Retrospective cohort analysis of liver biopsy specimens. Patients: Sixty adult patients (age ≥18 years) with serologically confirmed hepatitis C (anti‑HCV positive and HCV RNA positive) were included. Exclusion criteria were other etiologies of hepatitis (HBV/HDV, autoimmune hepatitis), alcohol-related or drug-induced liver disease.Clinical data: Age, sex, duration of disease, biochemical parameters (ALT, AST, bilirubin), and scoring systems (Child–Pugh, MELD when available) were extracted from medical records.Histology: Biopsy specimens were fixed in formalin, paraffin-embedded, sectioned, and stained with hematoxylin–eosin; additional stains were used as required. Fibrosis was scored as F0–F4, and inflammatory activity was evaluated with an IGA score considering portal, periportal, and lobular infiltration [4,6,24]. Steatosis (microvesicular/macrovesicular; focal/diffuse) and cholestasis were assessed semi‑quantitatively.Immunohistochemistry: IHC was performed in 20 cases using Ki‑67 (proliferation marker) and SMA (stellate cell/myofibroblast activation). Staining intensity and proportion of positive cells were graded (0–3 points). Kupffer cell activity was additionally evaluated morphometrically.Morphometry: A 100‑point ocular grid at ×600 magnification was used; 20 high‑power fields were analyzed per specimen.Statistics: Student’s t‑test and Pearson correlation were applied; p<0.05 was considered significant.
3. Results
|
|
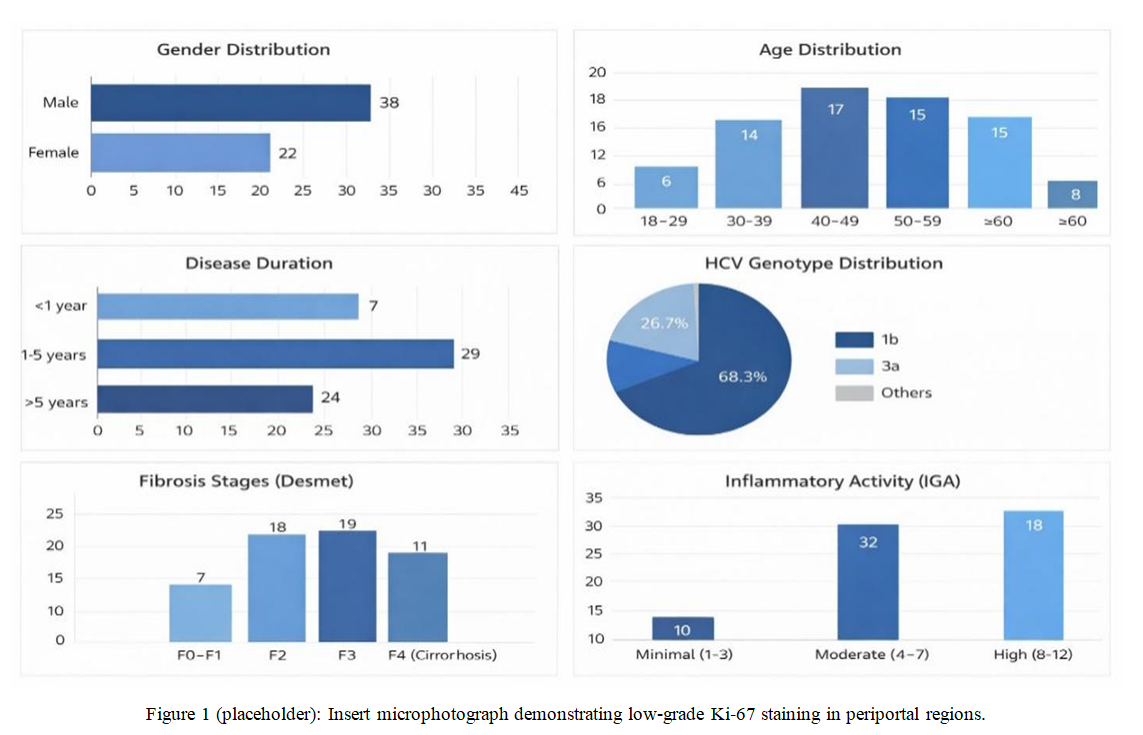 | Figure 1. Low-grade Ki‑67 positive expression pattern in liver tissue of chronic hepatitis C (representative image) |
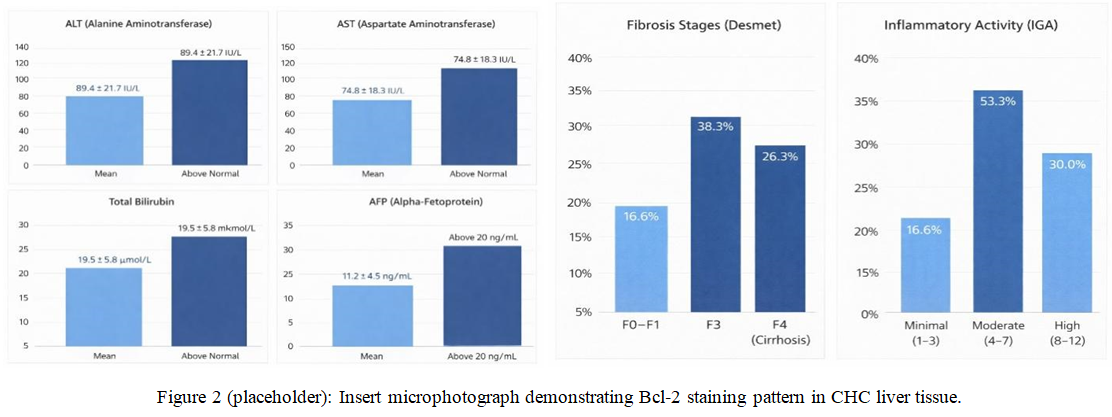 | Figure 2. Moderate Bcl‑2 positive expression in perilobular regions and low-grade positivity in centrilobular hepatocytes (representative image) |
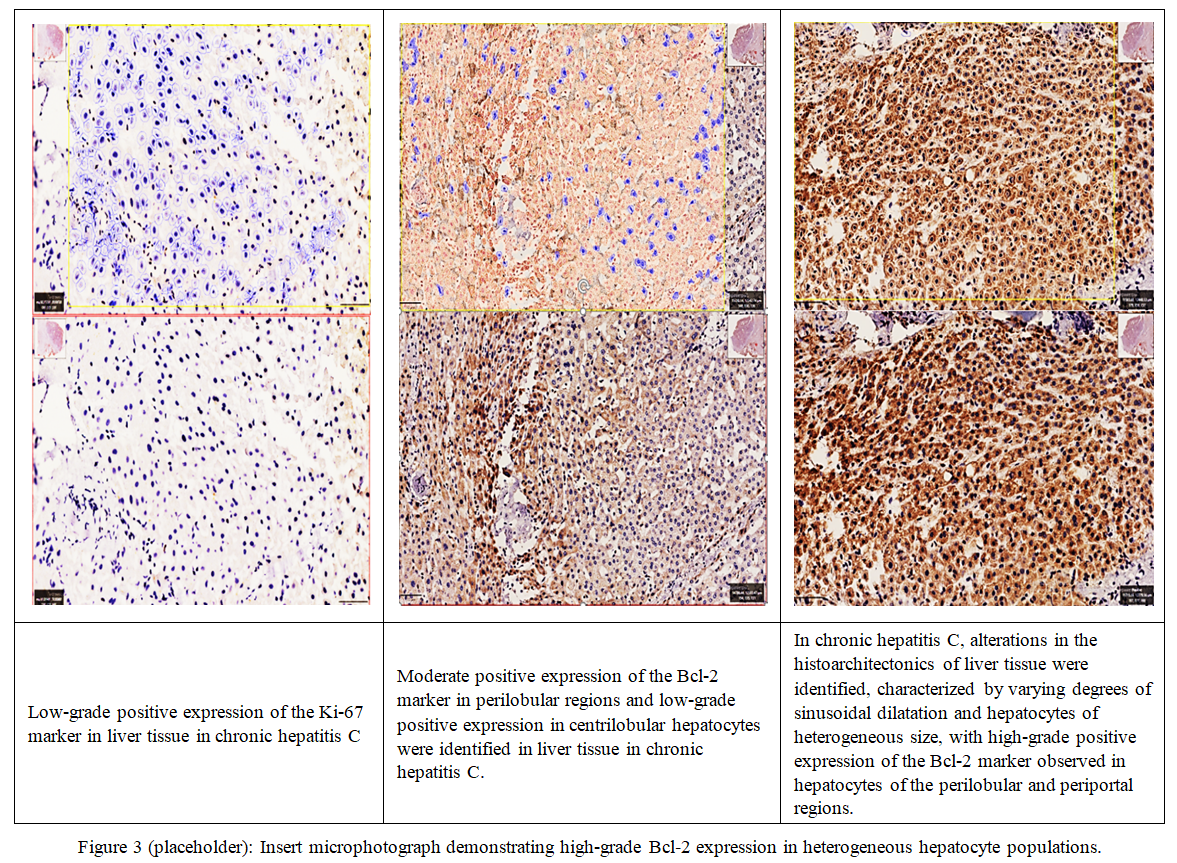 | Figure 3. High-grade Bcl‑2 positive expression in periportal/perilobular hepatocytes with sinusoidal dilatation (representative image) |
4. Discussion
- This study demonstrates a complex spectrum of premalignant pathomorphological changes in CHC, characterized by progressive fibrosis, increasing necroinflammatory activity, immune-cell infiltration, and stromal remodeling. The positive association between fibrosis stage and IGA activity is consistent with the concept that chronic inflammation promotes fibrogenesis and sets a biological background for malignant transformation [3,4,20]. The observed expansion of Ki‑67‑positive stromal/mesenchymal cells in advanced fibrosis supports the role of proliferative remodeling in septal formation and regenerative nodule development. Increased SMA expression in periportal/septal zones indicates stellate cell activation and myofibroblast differentiation, which are core mechanisms in extracellular matrix deposition and scar formation [5,7,8,18]. Kupffer cell hyperplasia and macrophage clustering in advanced fibrosis reflect persistent antigenic stimulation and cytokine-driven modulation of fibrogenesis (e.g., TGF‑β, TNF‑α pathways) described in prior studies [7,9,19]. From a practical perspective, combined histological grading/staging together with IHC markers may refine risk stratification for HCC and help tailor surveillance intensity, including AFP monitoring and follow-up biopsy intervals when clinically appropriate [1,2,9,17].
5. Conclusions
- 1. Premalignant liver changes in CHC are frequently represented by fibrosis stages F2–F4, moderate to high inflammatory activity, steatosis, and Kupffer cell hyperplasia.2. A positive correlation exists between fibrosis stage and necroinflammatory activity (IGA), supporting a close linkage between inflammation and fibrogenesis.3. Increased Ki‑67 proliferative activity and elevated SMA expression with advancing fibrosis suggest active stromal remodeling and stellate cell activation.4. Integrated histological and IHC assessment (Ki‑67, SMA and selected immune/macrophage parameters) can improve identification of high-risk patients for HCC development.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML