-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 462-469
doi:10.5923/j.ajmms.20261602.17
Received: Jan. 7, 2026; Accepted: Feb. 1, 2026; Published: Feb. 3, 2026

The Expression of the Systemic Inflammatory Response in Patients with Anterior Abdominal Wall Hernias Following the Use of Endoprostheses
Kuldashev G. N., Abdurakhmanov M. M.
Bukhara State Medical Institute named after Abu Ali ibn Sina, Bukhara, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In the presented study, a comprehensive analysis of the systemic inflammatory response in patients with postoperative ventral hernias is conducted, aiming to assess the bioinert properties and the impact on the cytokine profile of two different types of endoprostheses: polypropylene Esfil and titanium Titan Mesh. Based on their physicochemical characteristics and interactions with biological tissues, the study extensively describes the mechanisms of implant interaction with the immune system, with a particular focus on the roles of key cytokines, including TNF-α, IL-6, IL-8, and IL-4, in the processes of healing and scar formation following surgery. The results revealed that the use of titanium Mesh is associated with reduced inflammatory activity and a lower frequency of complications compared to polypropylene prostheses, indicating its superior bioinert properties and potential to improve clinical outcomes. The importance of the micro-porous structure of titanium Mesh and its promoting effect on cell adhesion and regeneration is also discussed as a key factor in accelerating the healing processes. The study's concluding recommendations emphasize the need for further research into the influence of different endoprostheses on the inflammatory response and regenerative processes, as well as the development of new materials with optimized biocompatible characteristics to enhance the treatment of postoperative ventral hernias and improve the quality of life for patients.
Keywords: Inflammation, Ventral Hernia, Biocompatibility, Prosthetic Implants, Cytokine Profile, TNF-α, IL-6, IL-8, IL-4, Scar Formation
Cite this paper: Kuldashev G. N., Abdurakhmanov M. M., The Expression of the Systemic Inflammatory Response in Patients with Anterior Abdominal Wall Hernias Following the Use of Endoprostheses, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 462-469. doi: 10.5923/j.ajmms.20261602.17.
Article Outline
1. Introduction
- Since the inception of abdominal surgery, the issue of ventral hernias occurring after surgeries has remained significant and is still not completely resolved. Studies indicate that anywhere from 5% to 40% of surgical interventions on abdominal organs lead to the formation of such hernias [1,2]. Recurrent hernias occurring on the anterior wall of the abdomen account for up to 30% of all surgical procedures in this area [3]. Modern medicine recognizes that the best treatment method for these hernias involves the use of endoprosthetics with various types of synthetic implants. Sublay or inlay techniques are common methods for these surgeries. However, in cases where postoperative hernias reach significant sizes, causing distortions in the structure of the anterior abdominal wall and subcutaneous tissue, the 'onlay' method is often required to reduce the operation time [4,5]. These circumstances emphasize the potential and, above all, the necessary need to study the conditions under which the implantation of endoprostheses occurs, including the investigation of changes in the cytokine profile over time.The variety of available synthetic endoprostheses and herniorrhaphy techniques necessitates a critical approach when selecting a specific type of implant, taking into account the expected size of the hernia, potential complications, and other relevant factors [6]. The issue of immunoreactivity, which depends on the structural and chemical components of endoprostheses, as well as their physical properties such as thickness and rigidity, remains at the forefront of modern herniology. Underestimating the immunological aspects of endoprosthetics can significantly complicate the interpretation of surgical outcomes in the context of wound dynamics [7,8].This need for a selective approach is compounded by the complexity arising from the diversity of hernioplasty outcomes, including postoperative exudative complications and optimal timing and methods of wound drainage [9].The study of immunological reactivity, especially considering the chemical composition of endoprostheses, is critically important for assessing treatment effectiveness [10]. Ignoring the immunological properties of the endoprosthesis can be a barrier to achieving reliable treatment outcomes, as the body's immune response to the prosthesis directly affects the wound healing process.The fundamental principle that certain cytokines promote inflammation while others modulate it is of critical importance for both cytokine biology and practical clinical practice [11,12]. It provides the basis for predicting the body's response to the introduction of artificial materials. The outcome of the inflammatory response depends on the balance of activity between cytokines with pro-inflammatory and anti-inflammatory properties [13]. In the initial phase of inflammation, pro-inflammatory cytokines activate the inflammatory process, causing vasodilation, increased local blood flow, and vascular wall permeability, leading to the formation of exudate in the inflamed area.At the initial stage of inflammation, pro-inflammatory cytokines serve a protective function by recruiting effector cells, such as neutrophils and macrophages, to the site of inflammation, enhancing their phagocytosis and bactericidal activity, and stimulating the onset of a specific immune response [14]. Cytokines-chemokines, especially IL-8, produced by activated macrophages, endothelial cells, and fibroblasts, regulate the migration of leukocytes to the site of inflammation, acting as chemotactic agents and recruiting them to the site of injury.It is important to emphasize that the protective effect of pro-inflammatory cytokines is manifested only when they act locally in the inflammatory zone. Excessive and systemic production of these mediators, on the other hand, can lead to the development of hyperthermia and deterioration of the overall condition of the organism [15].Tumor Necrosis Factor-alpha (TNF-α) is classified as a cytokine with pro-inflammatory properties. Its main function is through the activation of the endothelium and subsequent induction of intercellular adhesion molecule-1 (ICAM-1), which promotes transendothelial migration of granulocytes to the site of inflammation. TNF-α can be secreted not only by neutrophils but also by activated lymphocytes, and it acts as an endogenous pyrogen, affecting the thermoregulatory centers of the hypothalamus and causing hyperthermia. TNF-α also initiates the early phase of the immune response, catalyzing the release of IL-8 in response to the transplantation of foreign materials [16,17]. Interleukin-8 (IL-8) is synthesized by activated macrophages, as well as fibroblasts, epithelial cells, and hepatocytes in response to stimulation by IL-1 or TNF-α [18]. It facilitates the degranulation of neutrophils and can contribute to tissue damage. As a pro-inflammatory agent, IL-8 is involved in the process of leukocyte extravasation from the bloodstream into tissues and belongs to the family of chemokines with the ability to induce directed leukocyte migration to the inflammatory focus.To prevent excessive systemic inflammatory responses in the body, regulatory mechanisms are activated, including the synthesis of anti-inflammatory cytokines such as IL-4 and IL-10, which exert negative control over the inflammation process.Interleukin-4 (IL-4) plays a key role in regulating the body's anti-inflammatory responses, acting as an immunosuppressive agent and promoting the enhanced proliferation of B-lymphocytes. This cytokine facilitates the growth and differentiation of B-cells in cooperation with interleukins 5 and 6, thereby enhancing their functional activity [19]. Additionally, IL-4 inhibits the activation of macrophages and effectively blocks the actions of other cytokines, including IL-1, as well as the synthesis of nitric oxide and prostaglandins, thus limiting the development and progression of inflammatory reactions. The outcome of the inflammatory process, therefore, is a direct result of the balance between the activity of pro-inflammatory and anti-inflammatory cytokines, where excessive pro-inflammatory responses are controlled through the production of anti-inflammatory cytokines, particularly IL-4 and IL-10 [20]. These dynamic underscores the importance of further research in the field of cytokine profiles at the implantation site, as well as the analysis of morphological changes in this area.Modern surgical practices involve the use of numerous implants made from various materials. Titanium, characterized by its low molecular weight and high biocompatibility, has long established itself as one of the preferred materials in medicine due to its neutral interaction with biological tissues [21]. In Russia, a unique titanium mesh implant known as 'Titan Mesh' (NPV 'Temp') has been developed for the reconstruction of the aponeurosis in cases of anterior abdominal wall hernias.This study aimed to conduct a comparative analysis of clinical outcomes using two different types of synthetic endoprostheses: polypropylene 'Esfil' from the company 'Lintex' (Saint Petersburg) and 'Titan Mesh' (NPV 'Temp,' RUSSIA).
2. Materials and Methods
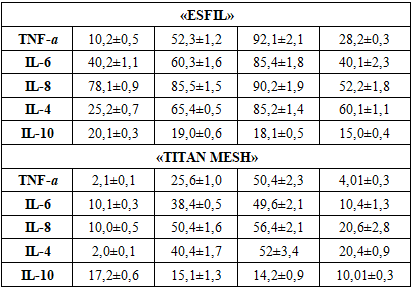
- The scientific study conducted in the surgical departments of the Bukhara branch of the National Research Center for Emergency Medicine from 2019 to 2022 aimed to compare the effectiveness of two different types of synthetic endoprostheses: polypropylene 'Esfil' from the 'Lintex' company (Saint Petersburg) and 'Titan Mesh'. During the study, which involved 112 patients divided into two groups, the outcomes of surgeries performed using the 'onlay' technique were evaluated. The patients were divided into two groups: in the first group (58 patients), a polypropylene mesh implant 'Esfil' was used as the endoprosthesis, while in the second group (54 patients), a mesh implant made from 'Titan Mesh' was used. This analysis provided valuable data on the clinical effectiveness and safety of these endoprostheses, contributing to a more informed choice of implant for the treatment of anterior abdominal wall hernias.Within the scope of this study, an analysis of the cytokine profile was conducted in two groups of patients using polypropylene and titanium Mesh, each group consisting of 20 individuals. To ensure the accuracy of the comparison, demographic and clinical characteristics, including gender, age, body mass index, and assessment of operative-anesthesiological risk, were strictly standardized between both groups. The study only included patients who presented with giant and extensive hernia defects, excluding small and medium-sized hernias due to their low statistical representativeness and short duration of patient stay in the hospital.The collection of wound exudate samples began on the third day after surgical intervention when the material was no longer saturated with erythrocytes. The extended use of drains was dictated by the necessity to perform cytokine studies on the 3rd, 5th, 7th, and 10th days after the surgeries. However, when exudation ceased, the drains were removed before the previously indicated 10-day period.The first phase of the study included the analysis and comparison of clinical data and cytokine indicators in aspirates from the area of endoprosthesis. Subsequently, key clinical parameters, including temperature reaction, leukocyte count, volume, and organoleptic characteristics of the fluid drained through the drains, were correlated with the cytokine profile.Active ultrasound monitoring of the postoperative wound was conducted for all study participants. In case of seroma or hematoma detection, ultrasound-guided puncture drainage was performed.The analytical process of studying cytokines involved the analysis of exudate collected through drainage systems and punctures of exudative formations detected using ultrasound diagnostics. The study focused on determining the characteristics of the fluid, such as color and volume, as well as on the levels of cytokines, including IL-8, IL-6, TNF-α, IL-10, and IL-4, the selection of which was based on their role in the development of the immune response.
3. Research Results and Their Discussion
- We found that postoperative formation of exudative formations in the implantation area is typical. Data on the frequency of exudative complications depending on the type of implant used were systematized and presented in Table 1.
|
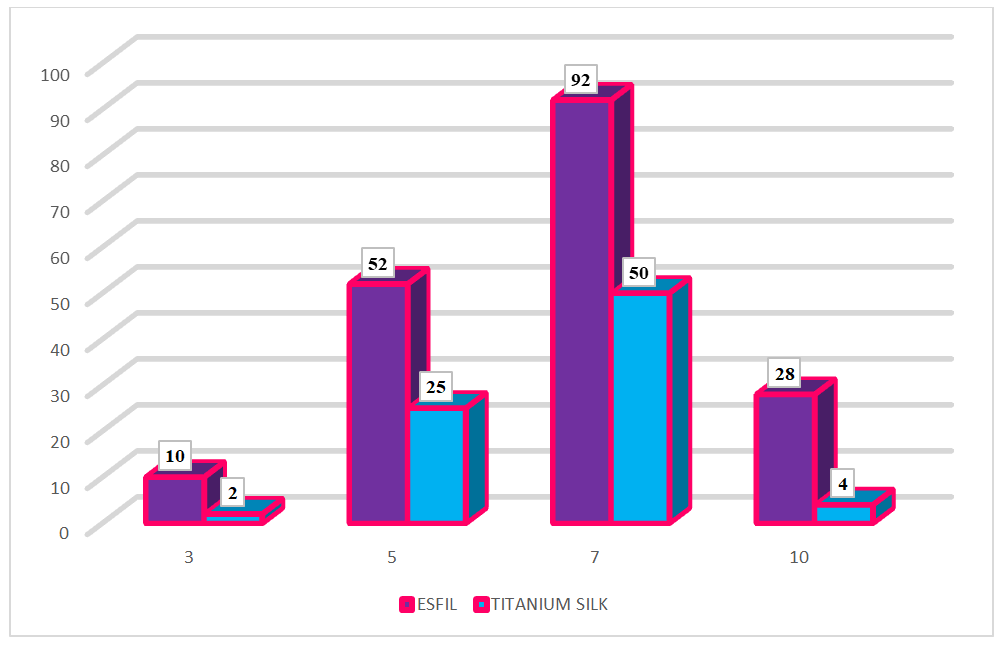 | Figure 1. The dynamics of changes in TNF-α concentration from day 3 to day 10 (pg/ml) |
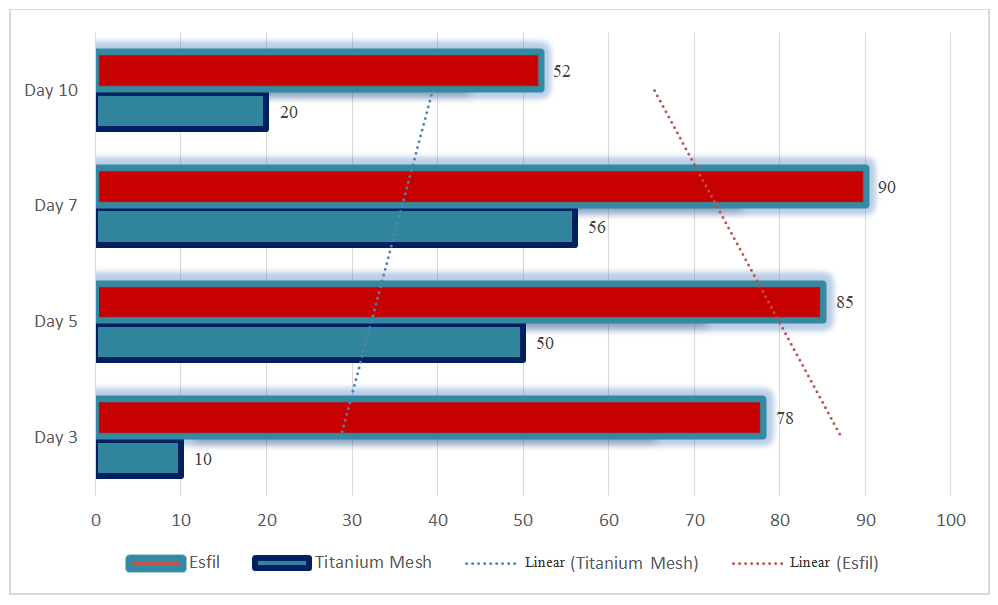 | Figure 2. Dynamics of changes in IL-8 concentration from day 3 to day 10 (pg/ml) |
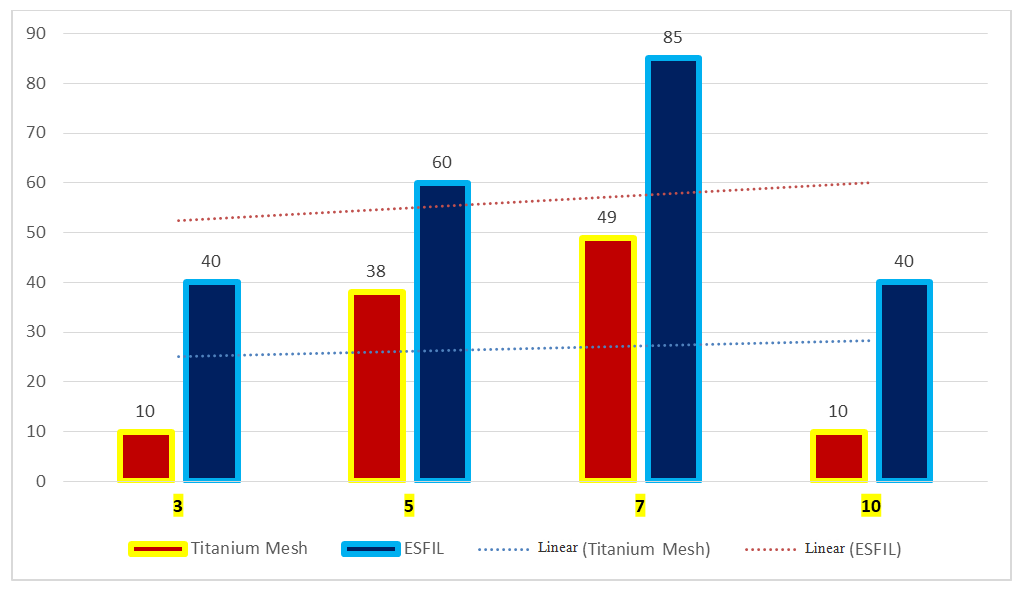 | Figure 3. Dynamics of changes in IL-6 concentrations from day 3 to day 10 (pg/ml) |
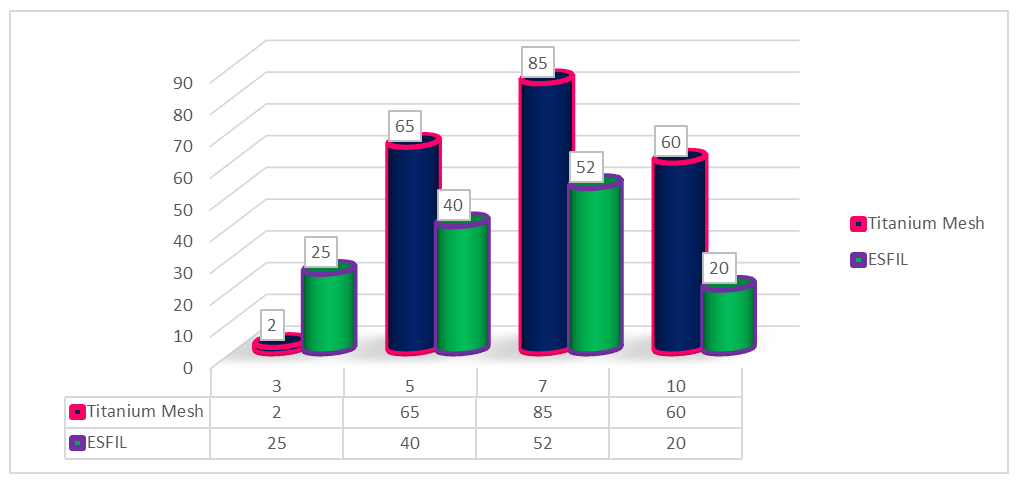 | Figure 4. Dynamics of changes in IL-4 concentrations from day 3 to day 10 (pg/ml) |
|
4. Conclusions
- The impact of the material on healing: The research demonstrates a noticeable difference in the healing process and inflammatory response between polypropylene "Esfil" and titanium "Titanium Mesh" implants. Titanium implants, thanks to their biologically inert properties, induce a milder inflammatory response, as evidenced by lower levels of inflammatory cytokines such as TNF-α, IL-6, IL-8, IL-4, and IL-10 compared to polypropylene implants.Dynamics of cytokine profiles: The dynamics of cytokine profiles, especially TNF-α, IL-6, IL-8, IL-4, suggest a more aggressive inflammatory response when polypropylene implants are used. The study implies that the chemical composition of the implant plays a significant role in modulating the local inflammatory environment.Complications with the use of different implants: It has been observed that postoperative complications, such as seroma and hematoma, occur more frequently when polypropylene implants are used compared to titanium implants. This indicates that the physical and chemical properties of "Titanium Mesh" contribute to a reduced risk of certain postoperative complications.The role of cytokines in wound healing: The study underscores the critical role of cytokines in the wound healing process. Balanced interaction between pro- and anti-inflammatory cytokines is crucial for effective healing, and the choice of implant material significantly influences this balance.The importance of implant selection: The results highlight the need for a careful approach when selecting the appropriate implant. Factors such as the biological inertness of the material, mechanical properties, and interaction with the immune system should be considered to minimize complications and optimize outcomes for the patient.Prospects for future research: The study points to the necessity of further investigating the long-term consequences of these findings. It also emphasizes the importance of developing targeted therapeutic strategies for inflammation management and tissue regeneration tailored to the type of implant used.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML