-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 457-461
doi:10.5923/j.ajmms.20261602.16
Received: Jan. 3, 2026; Accepted: Jan. 20, 2026; Published: Feb. 3, 2026

Effectiveness of Treatment Strategies for the Adverse Course of Chronic Heart Failure in Patients with Metabolic Syndrome
Nabieva Dilnoza Azamatovna, Kodirova Shakhlo Salomovna
Bukhara State Medical Institute, Bukhara, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Chronic heart failure (CHF) is frequently complicated by metabolic syndrome (MS), which significantly worsens prognosis and treatment outcomes. Objective: To evaluate the effectiveness of comprehensive treatment strategies in patients with CHF and metabolic syndrome. Methods: A prospective study included 162 patients with CHF (NYHA II–IV), including 96 patients with metabolic syndrome. Clinical evaluation, echocardiography, NT-proBNP measurement, heart rate variability (HRV) analysis, and quality-of-life assessment (SF-36) were performed. Results: Patients with CHF and MS demonstrated higher NT-proBNP levels, reduced HRV, and lower quality-of-life indices compared to patients without MS (p < 0.05). Comprehensive treatment significantly improved functional class, reduced NT-proBNP, and enhanced quality of life. Conclusion: Integrated cardiovascular and metabolic therapy significantly improves outcomes in CHF patients with metabolic syndrome.
Keywords: Chronic heart failure, Metabolic syndrome, Treatment effectiveness, NT-proBNP, Quality of life
Cite this paper: Nabieva Dilnoza Azamatovna, Kodirova Shakhlo Salomovna, Effectiveness of Treatment Strategies for the Adverse Course of Chronic Heart Failure in Patients with Metabolic Syndrome, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 457-461. doi: 10.5923/j.ajmms.20261602.16.
1. Introduction
- Chronic heart failure (CHF) represents one of the most significant and complex challenges in contemporary cardiovascular medicine. Despite remarkable advances in pharmacological therapy, interventional cardiology, and preventive strategies, CHF continues to be associated with high rates of morbidity, mortality, and rehospitalization worldwide [1,2]. According to global epidemiological data, more than 64 million people are currently living with heart failure, and this number is steadily increasing due to population aging and the rising prevalence of cardiometabolic disorders [2,3].Metabolic syndrome (MS) is one of the most clinically relevant comorbidities in patients with CHF. It comprises a cluster of interrelated metabolic abnormalities, including central obesity, insulin resistance, arterial hypertension, and dyslipidemia, all of which independently and synergistically contribute to cardiovascular risk [6,29]. The prevalence of metabolic syndrome among patients with CHF ranges from 40% to 60%, depending on diagnostic criteria and heart failure phenotype [5,8].From a pathophysiological standpoint, the interaction between CHF and metabolic syndrome is bidirectional. Insulin resistance alters myocardial energy metabolism and reduces metabolic efficiency, while abdominal obesity promotes chronic inflammation, oxidative stress, endothelial dysfunction, and myocardial fibrosis [4,8]. Dyslipidemia accelerates atherosclerosis and impairs coronary microcirculation, whereas arterial hypertension increases left ventricular afterload and promotes adverse remodeling [6,9]. Conversely, chronic heart failure exacerbates metabolic dysfunction through neurohumoral activation and reduced tissue perfusion, creating a vicious cycle of disease progression [1,17].Clinically, patients with CHF and metabolic syndrome exhibit more severe symptoms, higher NYHA functional class, impaired autonomic regulation, and poorer quality of life compared to patients without metabolic abnormalities [8,11]. Elevated natriuretic peptide levels and reduced heart rate variability have been consistently associated with worse prognosis in this population [18–21].Recent studies have highlighted the heterogeneity of heart failure phenotypes in the context of metabolic syndrome. Heart failure with preserved ejection fraction (HFpEF) is particularly strongly associated with obesity, insulin resistance, and systemic inflammation, leading to the concept of a metabolically driven HFpEF phenotype [4,9,10]. However, metabolic syndrome also adversely affects outcomes in patients with heart failure with reduced ejection fraction (HFrEF), attenuating reverse remodeling and therapeutic response [8,14].Large international cohort studies and registries, including the Framingham Heart Study, ARIC, ESC-HF Long-Term Registry, and GWTG-HF program, have demonstrated that metabolic syndrome is an independent predictor of heart failure development, hospitalization, and mortality [7,11,12]. Randomized controlled trials such as PARADIGM-HF, DAPA-HF, and EMPEROR-Reduced further suggest that modern therapies, particularly sodium–glucose cotransporter-2 inhibitors, may partially mitigate the adverse cardiometabolic profile of these patients [13–16].Despite these advances, real-world data indicate that standard heart failure therapy alone is often insufficient to fully address residual cardiometabolic risk [8,30]. Therefore, a comprehensive and integrated treatment approach is required. In this context, the present study aims to evaluate the effectiveness of combined cardiovascular and metabolic management strategies in patients with chronic heart failure and metabolic syndrome, with particular emphasis on biomarkers, autonomic function, and quality-of-life outcomes.
2. Materials and Methods
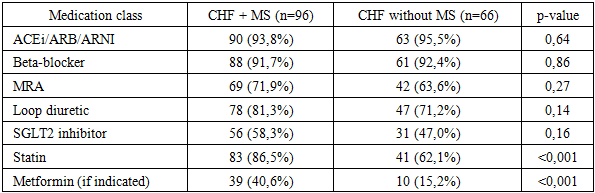
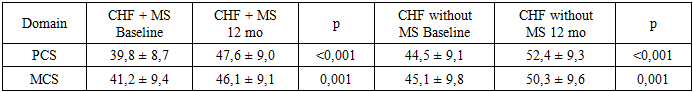
- This prospective observational study was conducted in specialized cardiology departments of tertiary medical institutions. The study aimed to evaluate the effectiveness of comprehensive treatment strategies in patients with chronic heart failure (CHF) with and without metabolic syndrome (MS) under routine clinical conditions. The follow-up period lasted 12 months, allowing assessment of both short-term and intermediate therapeutic outcomes. A total of 162 consecutive patients with clinically stable chronic heart failure were enrolled. Inclusion criteria were: age ≥18 years, established diagnosis of CHF according to the European Society of Cardiology (ESC) guidelines, and NYHA functional class II–IV. Metabolic syndrome was diagnosed based on the International Diabetes Federation (IDF) criteria, requiring central obesity plus at least two additional metabolic abnormalities. Patients were divided into two groups: Group 1: CHF patients with metabolic syndrome (n = 96), Group 2: CHF patients without metabolic syndrome (n = 66) Exclusion criteria included acute decompensated heart failure, myocardial infarction or stroke within the previous three months, severe valvular heart disease requiring surgery, malignant neoplasms, chronic inflammatory diseases, and advanced hepatic or renal failure. All patients underwent comprehensive clinical evaluation, including medical history, assessment of cardiovascular risk factors, physical examination, anthropometric measurements, and blood pressure monitoring. Functional status was assessed using the New York Heart Association (NYHA) classification. Transthoracic echocardiography was performed using standardized protocols. Left ventricular ejection fraction (LVEF) was calculated by the biplane Simpson method. Left ventricular volumes, left atrial diameter, and diastolic function parameters were also assessed. Examinations were performed by experienced cardiologists blinded to metabolic status. Fasting blood samples were collected in all patients. NT-proBNP levels were measured using standardized immunoassay techniques. Additional laboratory parameters included fasting plasma glucose, lipid profile, renal function markers, and liver enzymes. Heart rate variability (HRV) was assessed by 24-hour Holter monitoring. Time-domain indices, particularly SDNN, were analyzed to evaluate autonomic nervous system function. Quality of life was evaluated using the validated SF-36 questionnaire. Physical and mental component summary scores were calculated. Lower scores reflected poorer quality of life. All patients received guideline-directed medical therapy for CHF, including ACE inhibitors or ARNI, beta-blockers, mineralocorticoid receptor antagonists, diuretics, and SGLT2 inhibitors when indicated. Patients with metabolic syndrome additionally received dietary counseling, weight reduction programs, optimization of glycemic control, lipid-lowering therapy, and structured lifestyle modification. Statistical analysis was performed using standard statistical software. Continuous variables were expressed as mean ± standard deviation, and categorical variables as percentages. Group comparisons were conducted using Student’s t-test or Mann–Whitney U test. Correlation analysis was performed using Pearson or Spearman coefficients. A p-value < 0.05 was considered statistically significant.
3. Results
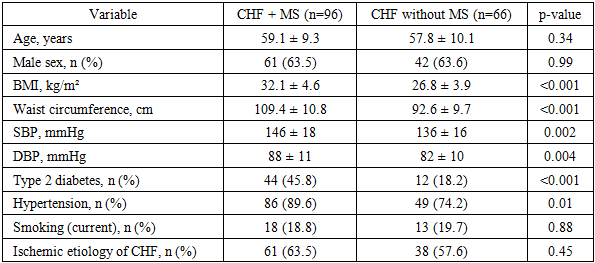
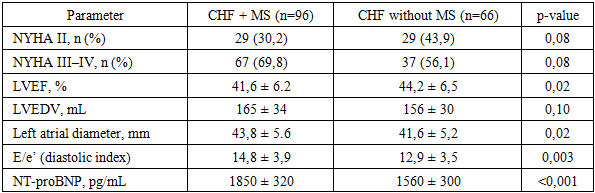
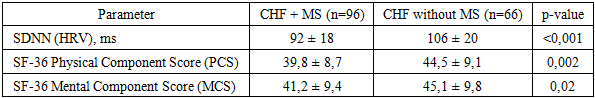
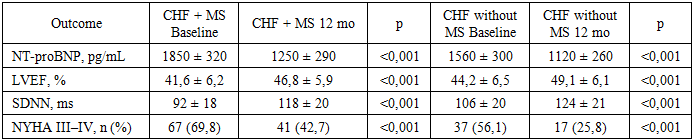
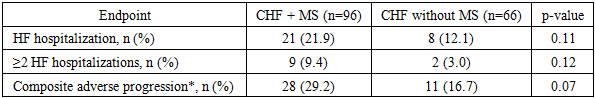
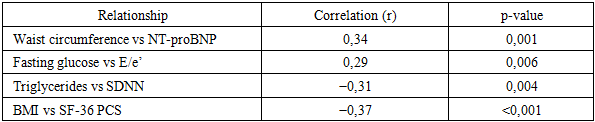
- A total of 162 patients with stable chronic heart failure (CHF) were included and stratified into CHF with metabolic syndrome (MS) (n = 96) and CHF without MS (n = 66). During the 12-month follow-up, complete clinical reassessment and laboratory testing were available for 155 (95.7%) participants. Loss to follow-up occurred due to relocation or non-attendance of scheduled visits.At baseline, patients with MS demonstrated a more adverse cardiometabolic profile, including higher waist circumference, higher fasting glucose, elevated triglycerides, and lower HDL-C. The MS group also had higher systolic blood pressure and a higher prevalence of type 2 diabetes mellitus.
|
|
|
|
|
|
|
|
|
4. Conclusions
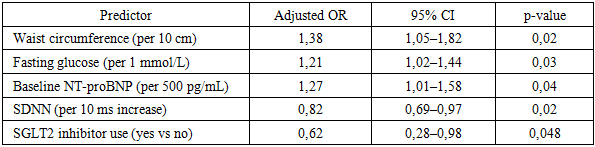
- The findings of this study demonstrate that metabolic syndrome significantly aggravates the clinical course of chronic heart failure and adversely affects treatment response. Patients with concomitant metabolic syndrome exhibited more severe baseline disease characteristics, including higher NYHA functional class, elevated NT-proBNP levels, impaired autonomic regulation, and reduced quality of life, which is consistent with previous international reports [8,11,18].Comprehensive guideline-directed medical therapy combined with targeted metabolic interventions resulted in significant clinical and functional improvement over the 12-month follow-up period. Reductions in NT-proBNP levels, improvements in left ventricular ejection fraction, partial restoration of autonomic balance, and meaningful gains in quality-of-life indices were observed, confirming the effectiveness of integrated treatment strategies in this high-risk population [13–16,21].Nevertheless, patients with metabolic syndrome demonstrated less pronounced improvement and retained a higher residual risk compared to patients without metabolic abnormalities. Persistent central obesity, impaired glucose metabolism, and dyslipidemia appeared to limit the extent of functional recovery, supporting the concept that metabolic syndrome represents a major driver of residual risk in chronic heart failure [5,8,29].These results emphasize the necessity of a multidisciplinary approach to heart failure management that extends beyond conventional neurohumoral modulation. Early identification of metabolic syndrome, aggressive metabolic risk factor control, and incorporation of therapies with combined cardiovascular and metabolic benefits should be considered essential components of personalized heart failure care [1,14,30].In conclusion, metabolic syndrome is a powerful modifier of chronic heart failure progression and therapeutic response. Integrated cardiometabolic treatment strategies significantly improve clinical outcomes and quality of life but do not completely eliminate residual risk. Future research should focus on refining personalized, long-term management models aimed at reducing cardiometabolic burden and improving prognosis in this growing patient population [3,9,10].In conclusion, metabolic syndrome significantly aggravates the adverse course of chronic heart failure and attenuates the response to therapy. Comprehensive, multidisciplinary treatment strategies targeting both cardiac dysfunction and metabolic derangements lead to significant clinical improvement but do not fully eliminate residual risk. Future research should focus on long-term, large-scale studies to refine integrated treatment models and develop personalized approaches aimed at improving prognosis and quality of life in this growing and particularly vulnerable patient population.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML