-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 453-456
doi:10.5923/j.ajmms.20261602.15
Received: Jan. 8, 2026; Accepted: Feb. 1, 2026; Published: Feb. 3, 2026

Rheumatoid Arthritis After COVID-19: Clinical and Immunological Changes and the Role of Interleukin-6
Sharapov Zafar Abdunayemovich1, Abduazizova Nargiza Khakimjanovna2
1Assistant, Department of Internal Medicine and Endocrinology, Tashkent State Medical University, Tashkent, Uzbekistan
2Associate Professor, Department of Faculty and Hospital Therapy No. 1 and Occupational Diseases, Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Rheumatoid arthritis (RA) is a chronic autoimmune inflammatory disease characterized by persistent synovitis, systemic inflammation, and progressive joint damage. Interleukin-6 (IL-6) plays a pivotal role in RA pathogenesis by amplifying immune activation and enhancing the acute-phase response. Coronavirus disease 2019 (COVID-19) may induce post-infectious immune dysregulation and inflammatory activation, potentially leading to increased RA activity. Objective: To compare serum IL-6 levels in RA patients who had confirmed COVID-19 within the last 6 months and RA patients without a history of COVID-19, and to evaluate the associations between IL-6 and C-reactive protein (CRP), disease activity (DAS28), and pain intensity (VAS). Methods: This study included 70 patients diagnosed with RA according to the 2010 ACR/EULAR classification criteria. Participants were divided into two groups: Group I—RA patients with confirmed COVID-19 within the preceding 6 months (n=35), and Group II—RA patients without prior COVID-19 (n=35). Disease activity was assessed using the DAS28 index. Pain intensity was evaluated using the Visual Analog Scale (VAS), functional status by the Health Assessment Questionnaire (HAQ), and health-related quality of life using the SF-36 questionnaire. CRP was measured as a systemic inflammatory marker. Serum IL-6 levels were determined by enzyme-linked immunosorbent assay (ELISA). Non-parametric statistical analyses were performed; results are presented as median (Q1; Q3). Between-group comparisons were conducted using the Mann–Whitney U test, and correlations were assessed using Spearman’s rank correlation coefficient. Statistical significance was defined as p<0.05, and high statistical significance as p<0.01. Results: Patients in the post-COVID-19 group demonstrated significantly higher inflammatory markers. Median CRP levels were greater in Group I compared with Group II: 28.0 (23.0; 35.0) vs 23.0 (19.0; 28.4) mg/L (p<0.01). Serum IL-6 levels were also higher in Group I: 41.4 (37.0; 47.9) vs 35.6 (30.4; 41.2) pg/mL (p<0.01). Moreover, clinical indices were significantly worse in Group I, including DAS28, VAS, HAQ, and the Ritchie Articular Index (all p<0.01). SF-36 scores were significantly lower among patients after COVID-19 (p<0.01), indicating poorer quality of life. Conclusion: Within 6 months following COVID-19, RA patients exhibited significantly elevated IL-6 and CRP levels, accompanied by higher disease activity, increased pain intensity, greater functional impairment, and reduced quality of life. Monitoring IL-6 and CRP may be clinically valuable for identifying and assessing RA activity exacerbation during the post-COVID period.
Keywords: Rheumatoid arthritis, COVID-19, Interleukin-6, C-reactive protein, DAS28, Post-COVID inflammation
Cite this paper: Sharapov Zafar Abdunayemovich, Abduazizova Nargiza Khakimjanovna, Rheumatoid Arthritis After COVID-19: Clinical and Immunological Changes and the Role of Interleukin-6, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 453-456. doi: 10.5923/j.ajmms.20261602.15.
1. Introduction
- Rheumatoid arthritis (RA) is a chronic autoimmune inflammatory disorder characterized by persistent synovial inflammation, disruption of immune–cytokine homeostasis, and progressive destructive joint damage. The disease is a major cause of pain, functional limitation, reduced work capacity, and impaired quality of life [1,2]. The clinical course of RA is largely determined by the magnitude of systemic inflammation and the activity of key cytokines regulating immune responses [2,3].Interleukin-6 (IL-6) is considered one of the most important cytokines in RA pathogenesis. It promotes T- and B-lymphocyte activation, enhances autoantibody production, and stimulates hepatic synthesis of acute-phase proteins—particularly C-reactive protein (CRP) [3,5]. Numerous studies have confirmed that elevated IL-6 levels in RA are associated with higher DAS28 scores, increased pain intensity, and worsening functional status [6–9]. Therefore, IL-6 is regarded as a clinically meaningful biomarker of RA activity and a therapeutic target that influences treatment strategies.The COVID-19 pandemic has further highlighted the complex interplay between autoimmune rheumatic diseases and viral infections. COVID-19 can trigger a pronounced inflammatory response, and immune dysregulation may persist for an extended period during the post-infectious phase [10,12]. In COVID-19, IL-6 is a central mediator of inflammation and is closely associated with increased CRP levels and clinical severity [11,12]. Thus, sustained elevation of IL-6 after COVID-19 may represent one of the mechanisms contributing to increased RA activity in affected patients.Based on these considerations, the aim of the present study was to compare IL-6 levels in RA patients who had COVID-19 within the previous 6 months and those without a history of COVID-19, and to evaluate the relationship between IL-6, CRP, and clinical disease activity indices.Literature ReviewThe pathogenesis of RA is closely related to immune cell activation within the synovial membrane and the production of multiple inflammatory cytokines. According to contemporary concepts, RA is not merely a localized joint disease but rather a systemic inflammatory condition affecting the entire organism [1,2]. In this context, biomarkers reflecting systemic inflammation play a crucial role in assessing disease activity and prognosis.IL-6 represents a key component of the “tissue injury—immune response—acute-phase reaction” axis in RA. Under IL-6 stimulation, B cells are driven toward autoantibody production, T helper cells are activated, and the synthesis of inflammatory mediators increases [3,4]. In addition, IL-6 enhances hepatic CRP production, further strengthening its clinical and laboratory relevance in RA [5]. Several studies have demonstrated that patients with high IL-6 levels tend to exhibit higher DAS28 scores, more intense pain, and greater functional impairment [6–9].Biologic agents targeting the IL-6 receptor—most notably tocilizumab—have been proven effective in reducing clinical disease activity in RA [9]. This provides strong evidence for the central role of IL-6 in RA pathogenesis and suggests that IL-6 monitoring may be useful in guiding therapeutic decision-making.With regard to COVID-19, the infection may induce not only acute-phase changes but also persistent immunological alterations after recovery. IL-6 has been identified as one of the leading mediators in the “cytokine storm” phenomenon and is associated with increased CRP levels and severe clinical outcomes [11,12]. Therefore, prolonged elevation of IL-6 after COVID-19 may be considered a potential factor contributing to disease exacerbation in autoimmune disorders, including RA.There is growing evidence that autoimmune and inflammatory diseases may worsen during the post-COVID period. Some authors have reported activation of autoimmune responses, the emergence of new autoimmune syndromes, or exacerbation of pre-existing autoimmune diseases following COVID-19 infection [10]. The mechanisms underlying these observations may vary and include virus-driven immune activation, disruption of immune equilibrium, and sustained persistence of inflammatory mediators [10,12].Consequently, evaluating markers such as IL-6 and CRP in RA patients during the post-COVID period is important not only from a scientific standpoint but also in clinical practice. By comparing RA patients with and without a history of COVID-19, the present study sought to clarify the clinical significance of post-infectious inflammatory activation and its impact on disease activity.
2. Materials and Methods
- This comparative study included 70 patients diagnosed with rheumatoid arthritis (RA) according to the 2010 ACR/EULAR classification criteria. All participants underwent clinical examination and follow-up at (institution name, city, country to be inserted). The study was approved by the Local Ethics Committee, and written informed consent was obtained from all participants prior to enrollment.Patients were stratified into two groups based on their COVID-19 history: Group I (COVID+) consisted of RA patients with confirmed COVID-19 within the preceding 6 months (n=35), whereas Group II (COVID−) included RA patients without a history of COVID-19 (n=35). The groups were comparable with respect to age, sex, disease duration, and clinical-radiological stage.Inclusion and Exclusion CriteriaInclusion criteria were: (1) diagnosis of RA according to the 2010 ACR/EULAR criteria; and (2) age ≥18 years. At the time of assessment, participants were required to have clinically active RA manifestations. For Group I, confirmation of COVID-19 within the past 6 months was required and was based on at least one of the following: PCR or rapid antigen testing, chest computed tomography findings, medical documentation, or physician-confirmed diagnosis.Exclusion criteria included: presence of other systemic autoimmune diseases, acute infections at the time of evaluation, malignancy, severe decompensated comorbid conditions, or refusal to participate in the study.Clinical AssessmentRA disease activity was assessed using the Disease Activity Score in 28 joints (DAS28). Pain intensity was evaluated using a 100-mm Visual Analog Scale (VAS; 0–100 mm). Functional status was assessed with the Health Assessment Questionnaire (HAQ; score range 0–3), and health-related quality of life was evaluated using the Short Form-36 (SF-36) questionnaire. In addition, the Ritchie Articular Index was calculated as a clinical measure of joint tenderness.Laboratory MeasurementsSerum C-reactive protein (CRP) was measured as a marker of systemic inflammation. Serum IL-6 concentration was determined using enzyme-linked immunosorbent assay (ELISA). Blood samples were collected in the morning after an overnight fast, centrifuged, and the separated serum was stored under standard conditions until analysis.Statistical AnalysisStatistical analyses were performed using (Statistica/SPSS/Microsoft Excel; specify software and version if available). Non-normally distributed variables are presented as median (Me) and interquartile range (Q1; Q3). Between-group comparisons were conducted using the Mann–Whitney U test. Associations between IL-6 and CRP, DAS28, and VAS were evaluated using Spearman’s rank correlation coefficient. A p-value <0.05 was considered statistically significant, while p<0.01 indicated high statistical significance.
3. Results
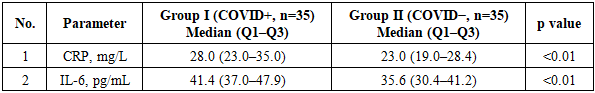
- RA patients with confirmed COVID-19 within the previous 6 months exhibited higher systemic inflammatory activity and greater clinical disease burden than those without COVID-19 history. As summarized in Table 1, median CRP levels were significantly higher in Group I (COVID+) compared with Group II (COVID−) (28.0 [23.0–35.0] vs 23.0 [19.0–28.4] mg/L; p<0.01). Similarly, serum IL-6 concentrations were elevated in Group I (41.4 [37.0–47.9] vs 35.6 [30.4–41.2] pg/mL; p<0.01), indicating increased inflammatory activation in the post-COVID cohort.
|
4. Discussion
- The findings of the present study indicate that RA patients evaluated within 6 months after COVID-19 exhibited significantly higher inflammatory activity compared with RA patients without a history of COVID-19. This observation is supported by elevated CRP levels in the post-COVID group. CRP is synthesized in the liver under IL-6 stimulation and reflects the magnitude of systemic inflammation. Therefore, increased CRP after COVID-19 may suggest persistent inflammatory activation during the post-infectious period.In addition, serum IL-6 concentrations were significantly higher in Group I, further supporting the pivotal role of this cytokine in RA pathogenesis. Increased IL-6 may contribute to enhanced autoimmune activity and amplification of the acute-phase response. Moreover, prolonged immune dysregulation after COVID-19 may further intensify IL-6–mediated inflammatory pathways, potentially leading to sustained disease activation.Clinical outcomes were also markedly worse among post-COVID RA patients, including higher DAS28 scores, greater pain intensity, increased functional impairment, and reduced quality of life. These findings are consistent with RA exacerbation during the post-infectious period and may reflect aggravation of clinical symptoms following COVID-19.Overall, the results suggest that COVID-19 may adversely affect the clinical course of RA, with IL-6 serving as a clinically relevant biomarker in this process. Consequently, monitoring of IL-6 and CRP during the post-COVID period may be valuable for patient follow-up and timely adjustment of therapeutic strategies.
5. Conclusions
- Within 6 months after COVID-19, RA patients demonstrated significantly higher serum IL-6 and CRP levels, accompanied by increased disease activity, greater pain intensity, and more severe functional limitations. In addition, health-related quality of life assessed by the SF-36 questionnaire was lower in the post-COVID group. Monitoring IL-6 and CRP during the post-COVID period is clinically important for assessing RA activity escalation and optimizing patient management.Key Findings1. RA patients after COVID-19 had significantly elevated CRP and IL-6 levels (p<0.01).2. The post-COVID group demonstrated higher DAS28, VAS, HAQ, and Ritchie Articular Index scores (p<0.01).3. Post-COVID patients exhibited lower SF-36 scores, indicating poorer quality of life (p<0.01).4. IL-6 and CRP represent clinically relevant biomarkers for assessing RA activity during the post-COVID period.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML