-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 448-452
doi:10.5923/j.ajmms.20261602.14
Received: Jan. 6, 2026; Accepted: Jan. 23, 2026; Published: Feb. 3, 2026

Pathomorphological Characteristics of Metastatic Breast Cancer in Women Living in the Lower Aral Sea Region
Adham A. Khodjaniyazov1, Baxrom S. Yuldashev2, Mekhriban Kh. Khadjimuratova1
1Department of Pathomorphology, Urgench State Medical Institute, Urgench, Uzbekistan
2Vice-Rectorate for International Relations, Mamun University, Khiva, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: The Lower Aral Sea region represents an ecologically compromised area with elevated cancer incidence rates. However, pathomorphological characteristics of metastatic breast cancer in this population remain insufficiently studied. Methods: This retrospective descriptive study analyzed 213 women with metastatic breast cancer from the Khorezm Regional Oncology Center (2020–2025). Histopathological examination was performed using H&E and special stains. Morphometric analysis was conducted using Fiji ImageJ software (version 1.54f). Statistical analysis employed chi-square test with significance at p < 0.05. Results: A total of 306 metastatic foci were identified. The highest metastatic burden was observed in women aged 45–59 years (n=101, 46.1%; χ² = 12.84, p = 0.005). Predominant metastatic sites included bones (32.4%), lungs (25.5%), lymph nodes (20.6%), and liver (15.0%). Invasive ductal carcinoma was the most common histological subtype (78.9%), followed by invasive lobular carcinoma (14.1%). Conclusion: Metastatic breast cancer in the Lower Aral Sea region predominantly affects perimenopausal women with multiple organ involvement. These findings emphasize the need for enhanced regional screening programs and individualized therapeutic strategies in this ecologically vulnerable population.
Keywords: Breast cancer, Metastasis, Histopathology, Lower Aral Sea region, Environmental oncology
Cite this paper: Adham A. Khodjaniyazov, Baxrom S. Yuldashev, Mekhriban Kh. Khadjimuratova, Pathomorphological Characteristics of Metastatic Breast Cancer in Women Living in the Lower Aral Sea Region, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 448-452. doi: 10.5923/j.ajmms.20261602.14.
Article Outline
1. Introduction
- Breast cancer remains the most prevalent malignancy among women globally and constitutes a significant public health burden. According to the International Agency for Research on Cancer (IARC), over 2.3 million new cases of breast cancer were diagnosed worldwide in 2020, establishing it as the most frequently occurring neoplasm in women [1]. Metastatic breast cancer represents a particularly challenging clinical entity, characterized by aggressive clinical behavior, limited therapeutic efficacy, and unfavorable prognosis [2].The Lower Aral Sea region warrants particular attention due to its unique ecological and geographical characteristics. The desiccation of the Aral Sea has precipitated an environmental catastrophe of unprecedented magnitude, manifesting as airborne dust-aerosol contamination laden with pesticide residues and heavy metals, soil salinization, and potable water scarcity. These environmental perturbations have substantially elevated the risk of chronic and oncological diseases among the regional population, particularly women [3,4]. Epidemiological surveillance has documented elevated cancer incidence rates in the Aral Sea basin compared to other regions, including a notably higher frequency of breast cancer [5].From a morphological perspective, invasive ductal carcinoma (IDC) accounts for the majority of breast cancers (70–80%), followed by invasive lobular carcinoma (ILC), which comprises approximately 10–15% of cases [6]. IDC is characterized by cellular pleomorphism, enlarged hyperchromatic nuclei, stromal fibrosis, and desmoplastic reaction. Conversely, ILC exhibits diminished intercellular adhesion, monomorphic nuclei, and a distinctive single-file growth pattern [7]. The pattern of metastatic spread varies according to histological subtype, with bones, lungs, liver, and brain representing the most common distant sites [8].Despite extensive global research on breast cancer metastasis, region-specific data from ecologically compromised areas such as the Lower Aral Sea region remain scarce. Understanding the pathomorphological characteristics of metastatic breast cancer in this population is essential for developing targeted screening programs and optimizing therapeutic approaches.The objective of this study was to investigate the pathomorphological characteristics of metastatic breast cancer among women residing in the Lower Aral Sea region, with particular emphasis on age distribution, metastatic site patterns, and histological variants.
2. Materials and Methods
2.1. Study Design and Population
- This retrospective descriptive study was conducted at the Khorezm Regional Branch of the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology during the period January 2020 to December 2025. The study population was derived from 652 female patients who received multimodal therapy for breast malignancies. From this cohort, 213 patients with histologically confirmed metastatic disease were selected for analysis.Inclusion criteria comprised: (1) histologically confirmed primary breast carcinoma; (2) morphologically verified metastatic foci using hematoxylin and eosin (H&E) or special stains; (3) complete clinical documentation; and (4) availability of adequate archival tissue specimens. Exclusion criteria included: (1) metastatic disease suspected but not morphologically confirmed; (2) insufficient quality archival materials; (3) synchronous primary malignancies of other anatomical sites; and (4) incomplete medical records.
2.2. Histopathological Examination
- Tissue specimens were fixed in 10% neutral buffered formalin and processed using standard paraffin embedding techniques. Serial sections of 4–5 μm thickness were prepared and stained with hematoxylin and eosin (H&E) for routine morphological assessment. Special staining techniques including Van Gieson (for connective tissue evaluation) and Alcian blue (for mucin detection) were employed when indicated. Histological typing was performed according to the World Health Organization (WHO) Classification of Breast Tumors (5th edition, 2019) [9].
2.3. Morphometric Analysis
- Morphometric analysis was performed using Fiji ImageJ software (version 1.54f, National Institutes of Health, USA). Digital images were captured using a light microscope equipped with a digital camera at standardized magnifications (×20, ×40, ×100). Cell and nuclear diameters were measured in micrometers (μm) across multiple representative fields. A minimum of 50 cells per specimen were measured to ensure statistical reliability.
2.4. Statistical Analysis
- Statistical analysis was performed using IBM SPSS Statistics (version 26.0, IBM Corp., Armonk, NY, USA). Categorical variables were expressed as frequencies and percentages. The chi-square (χ²) test was employed for comparison of categorical variables between groups. A p-value < 0.05 was considered statistically significant.
2.5. Ethical Considerations
- This study was approved by the Local Ethics Committee of Urgench State Medical Institute (Protocol No. ___, Date: ___). Due to the retrospective nature of the study and the use of anonymized archival data, the requirement for informed consent was waived. All procedures were conducted in accordance with the ethical standards of the institutional research committee and the Declaration of Helsinki (1964) and its later amendments.
3. Results
3.1. Patient Demographics and Age Distribution
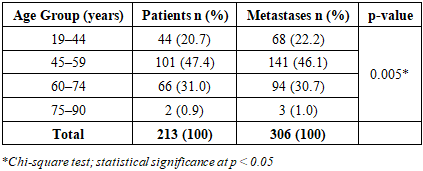
- The study cohort comprised 213 patients with metastatic breast cancer. A cumulative total of 306 metastatic foci were identified, indicating that multiple organ involvement was common (mean: 1.44 metastatic sites per patient). Age-stratified analysis revealed significant differences in metastatic distribution among age groups (χ² = 12.84, p = 0.005). The highest metastatic burden was observed in women aged 45–59 years, comprising 101 patients (47.4%) with 141 metastatic foci (46.1%). Detailed age distribution is presented in Table 1.
|
3.2. Metastatic Site Distribution
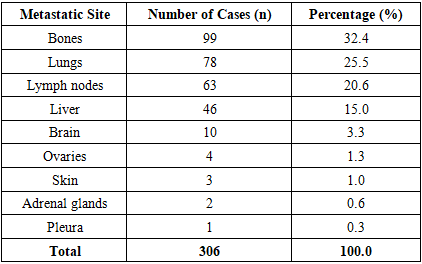
- Analysis of metastatic site distribution revealed predominant involvement of the skeletal system (n=99, 32.4%), followed by lungs (n=78, 25.5%), lymph nodes (n=63, 20.6%), and liver (n=46, 15.0%). Less frequent metastatic sites included the brain (n=10, 3.3%), ovaries (n=4, 1.3%), skin (n=3, 1.0%), adrenal glands (n=2, 0.6%), and pleura (n=1, 0.3%). Detailed distribution is presented in Table 2.
|
3.3. Geographic Distribution and Surgical Treatment
- Geographic distribution analysis revealed variable patient numbers across Khorezm Province and the Republic of Karakalpakstan. The highest patient numbers were recorded in Urgench city (n=37, 17.4%), Urgench district (n=27, 12.7%), Khonqa district (n=20, 9.4%), and Shovot district (n=20, 9.4%). Overall, 45 patients (21.1%) originated from urban areas, while 168 patients (78.9%) resided in rural districts. Regarding surgical intervention, 156 patients (73.2%) underwent surgical treatment, whereas 57 patients (26.8%) did not receive surgical intervention due to advanced disease stage or clinical contraindications.
3.4. Histopathological Findings
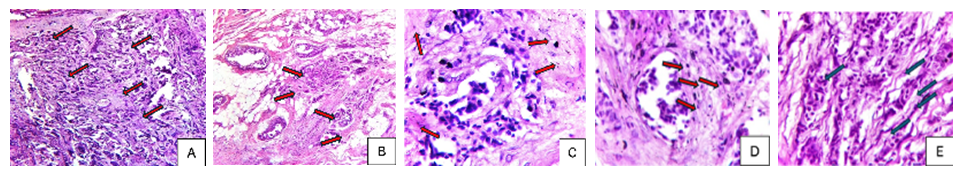
- Histopathological examination of metastatic foci revealed invasive ductal carcinoma as the predominant subtype (n=168, 78.9%), followed by invasive lobular carcinoma (n=30, 14.1%). Less common histological variants included mucinous carcinoma (n=8, 3.8%), tubular carcinoma (n=4, 1.9%), and micropapillary carcinoma (n=3, 1.4%).Invasive ductal carcinomas demonstrated characteristic morphological features including partial preservation of cribriform and ductal architectural elements, prominent desmoplastic stromal reaction, and variable lymphoid infiltration. Tumor cells exhibited enlarged hyperchromatic nuclei with irregular contours and increased nuclear-to-cytoplasmic ratio. Invasive lobular carcinomas displayed distinctive features including loss of intercellular adhesion (attributed to E-cadherin dysfunction), single-file (Indian file) growth pattern, and stromal fibrosis with collagenization (Figure 1).
3.5. Morphometric Analysis
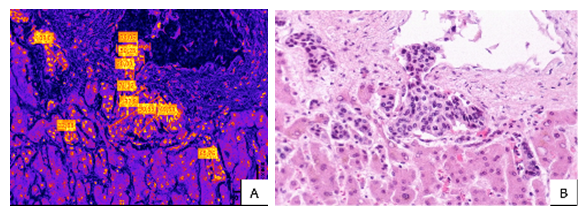
- Morphometric analysis using Fiji ImageJ software revealed significant variation in cell dimensions between metastatic sites. In lymph node metastases, cell and nuclear diameters ranged from 19.8 to 68.6 μm (mean: 42.3 ± 12.7 μm). Hepatic metastases demonstrated larger cellular dimensions, ranging from 20.0 to 132.8 μm (mean: 58.4 ± 28.9 μm), suggesting greater cellular pleomorphism in visceral metastases (Figure 2).
4. Discussion
- This study provides comprehensive pathomorphological characterization of metastatic breast cancer in women residing in the Lower Aral Sea region, an ecologically compromised area with unique environmental health challenges. Our findings demonstrate distinct patterns of metastatic distribution and histopathological features that may inform regional cancer control strategies.The predominance of multiple organ metastasis (306 metastatic foci in 213 patients; mean 1.44 sites per patient) underscores the aggressive nature of breast cancer in this population. This finding aligns with global data reported by Harbeck et al. [10] in their comprehensive review published in The Lancet, which documented that metastatic breast cancer frequently involves multiple organ systems simultaneously.Age-stratified analysis revealed that the highest metastatic burden occurred in women aged 45–59 years (47.4% of patients; p = 0.005), corresponding to the perimenopausal and early postmenopausal period. This observation is consistent with epidemiological data reported by Bray et al. [1] and may reflect the confluence of hormonal changes, accumulated environmental exposures, and delayed diagnosis in this age group. Notably, the peak incidence in our cohort coincides with the period of greatest cumulative exposure to environmental pollutants associated with the Aral Sea ecological disaster.The pattern of metastatic site distribution in our study—with bones (32.4%), lungs (25.5%), lymph nodes (20.6%), and liver (15.0%) as predominant sites—is consistent with published literature. A landmark study by Coleman et al. [11] in Nature Reviews Cancer reported that bone metastases occur in approximately 65–75% of patients with advanced breast cancer, followed by lung and liver involvement. Our findings confirm these global patterns while providing region-specific quantitative data.Histopathological analysis confirmed invasive ductal carcinoma as the predominant subtype (78.9%), which aligns with global epidemiological data indicating that IDC comprises 70–80% of all breast cancers [6]. The proportion of invasive lobular carcinoma (14.1%) in our cohort is consistent with international reports. Cardoso et al. [2] in their European Society for Medical Oncology (ESMO) guidelines emphasized the importance of histological subtyping for treatment planning, as different subtypes exhibit distinct patterns of metastatic spread and therapeutic response.Our study uniquely demonstrates region-specific metastatic patterns in an ecologically compromised population that has been exposed to environmental carcinogens associated with the Aral Sea disaster for several decades. The high proportion of poorly differentiated tumors with marked cellular pleomorphism and elevated mitotic activity observed in our cohort may reflect the influence of environmental factors on tumor biology, although this hypothesis requires further investigation through molecular studies.The geographic distribution analysis revealed that the majority of patients (78.9%) originated from rural areas, where access to early detection and screening programs may be limited. This finding emphasizes the urgent need for decentralized cancer screening initiatives in the Lower Aral Sea region. The disparity between urban and rural areas in diagnostic capability and oncological awareness represents a significant barrier to early detection and optimal treatment outcomes.From a clinical perspective, these findings have important implications for patient management. The identification of age-specific risk patterns may assist clinicians in risk stratification and surveillance planning for women in the perimenopausal age group. Furthermore, the characterization of predominant metastatic sites can guide imaging protocols and staging workup in this population.This study has several limitations. First, the retrospective design limits causal inference and may introduce selection bias. Second, molecular subtyping (ER, PR, HER2 status) was not uniformly available for all patients, precluding comprehensive biological characterization. Third, survival data were not analyzed in this study. Future prospective studies incorporating molecular profiling and survival analysis are warranted to further elucidate the clinical implications of our findings.
5. Conclusions
- This study characterized the pathomorphological features of metastatic breast cancer in 213 women residing in the Lower Aral Sea region. The key findings include: (1) multiple organ involvement was the predominant metastatic pattern, with a mean of 1.44 metastatic sites per patient; (2) women aged 45–59 years exhibited significantly higher metastatic burden (p = 0.005); (3) bones, lungs, lymph nodes, and liver were the most frequently affected metastatic sites; (4) invasive ductal carcinoma was the predominant histological subtype (78.9%); and (5) poorly differentiated tumors demonstrated aggressive morphological features including marked cellular pleomorphism and elevated mitotic activity.These findings underscore the necessity for enhanced regional cancer screening programs targeting perimenopausal women and the implementation of individualized therapeutic strategies in this ecologically vulnerable population. Future studies incorporating molecular profiling and survival analysis are recommended to further optimize cancer care in the Lower Aral Sea region.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML