-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 436-439
doi:10.5923/j.ajmms.20261602.11
Received: Jan. 2, 2026; Accepted: Jan. 22, 2026; Published: Feb. 3, 2026

Morphological Aspects of Early Carcinogenesis of the Stomach and the Role of Helicobacter Pylori Strains
Ismailova J. A. , Rakhimova Sh. Sh. , Pulatov S. S.
Republican Specialized Scientific Practical Medical Center of Therapy and Medical Rehabilitation, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

With the aim of early diagnosis of stomach cancer, the authors studied the significance of morphological studies and pathogenic strains of H. pylori in 100 patients with gastric diseases infected with H. pylori and in a control group of 20 individuals. The results of the study showed that a mild degree of gastric mucosal damage by H. pylori in chronic gastritis is associated with low intensity of inflammatory changes, and the CagA gene can be considered a marker gene indicating the pathogenicity status of H. pylori.
Keywords: H. pylori, Chronic gastritis, Endoscopic studies, Gastric mucosa, ureC and CagA genes, Biopsy, Molecular genetic studies, OLGA and OLGIM systems, Atrophy, Intestinal metaplasia
Cite this paper: Ismailova J. A. , Rakhimova Sh. Sh. , Pulatov S. S. , Morphological Aspects of Early Carcinogenesis of the Stomach and the Role of Helicobacter Pylori Strains, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 436-439. doi: 10.5923/j.ajmms.20261602.11.
1. Introduction
- In 2022, 20 million new cancer cases were registered worldwide, and 9.7 million deaths were recorded. 53.3 million cancer patients survived 5 years after diagnosis [3,14,16]. Approximately 1 in 5 people will develop cancer during their lifetime. According to IARC, it is 12 times more common in underdeveloped and developing countries than in developed countries [17]. Several factors contribute to the development of cancer, one of the main factors of which is Helicobacter pylori (H. pylori). Studies have shown that aggressive strains of H. pylori are known to cause severe forms of the disease [1,8,15]. In addition, molecular genetic factors of a person have been confirmed to play an important role. Gastric cancer ranks 2nd in the structure of oncological diseases in our country, with an incidence of 6.8 per 100,000 population (2022). In our country, gastric cancer, regardless of etiopathogenetic factors, is treated mainly on the basis of oncological principles, and unlike other types of cancer, the 5-year survival rate does not exceed 50% even in early stages [7]. The high aggressiveness, moderate sensitivity to radiation and chemotherapy indicate the need to focus on the molecular genetic factors of this disease and develop personalized treatment methods in the future [9,12]. To date, gastric cancer and precancerous lesions in our country have not been sequenced or genotyped for exome, labeled, expression-based marker genes, marker polymorphisms, region-specific H. pylori strains, and disease-associated polymorphisms [2,13]. Therefore, the percentage of patients diagnosed at an early stage of cancer remains quite low. Of course, morphological examinations play a key role in the early diagnosis of gastric cancer and help to identify precancerous changes in the gastric mucosa due to H. pylori infection [5,10]. Numerous literature reviews have shown that the risk of gastric cancer is higher in patients with advanced age, gender (more men than women), atrophic gastritis, intestinal metaplasia, and gastric dysplasia than in patients with other precancerous diseases (for example, gastric polyposis), and aggressive strains of H. pylori are involved as their “conductors” [4,6,11]. This, in turn, requires a serious and individual approach to precancerous diseases.The aim of the study is to study the importance of morphological examinations and pathogenic strains of H. pylori in the early diagnosis of stomach cancer.The object of the study was 100 patients with gastric diseases infected with H. pylori, who underwent molecular genetic testing and morphological examination of targeted biopsy samples to identify pathological changes in the gastric mucosa.
2. Methods of the Study
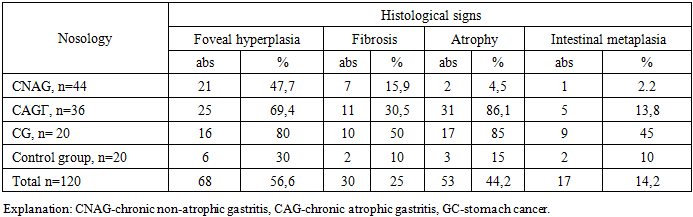
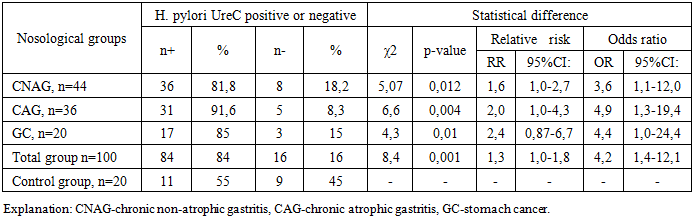
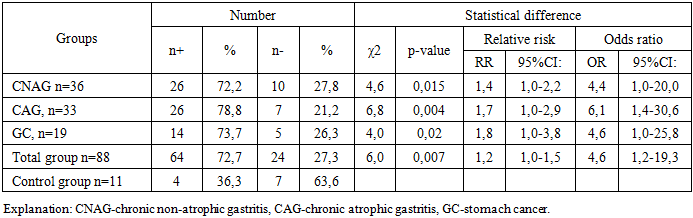
- All patients were divided into three study groups according to the nosology of the pathological process, and their results were studied comparatively: 80 patients with chronic gastritis (CG), and 20 patients with gastric cancer (GC) and a control group of 20 healthy people.All patients underwent gastroscopy and a five-point gastric biopsy. Based on the studied material, a morphological study of biopsies was carried out to identify preneoplastic changes (fibrosis, atrophy, intestinal metaplasia (IM), dysplasia) in patients with gastric diseases infected with H. pylori (Table 1).
|
|
|
3. Conclusions
- 1) In chronic gastritis, the weak level of infection of the gastric mucosa with H. pylori is associated with a low intensity of inflammatory changes.2) The results of the study showed that the CagA gene is a risk factor for the development of gastrointestinal diseases and that this gene can be used as a marker gene for the pathogenic status of H. pylori.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML