-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 330-335
doi:10.5923/j.ajmms.20261601.69
Received: Jan. 3, 2026; Accepted: Jan. 23, 2026; Published: Jan. 26, 2026

Morphological Changes in the Spleen Tissue After Biocorrection Against the Background of Chemotherapy-Induced Renal Failure in an Experimental Model
Khodjiyeva Nozima Shukhratovna, Dustov Sherali Hayatovich
Bukhara State Medical Institute, Uzbekistan
Correspondence to: Khodjiyeva Nozima Shukhratovna, Bukhara State Medical Institute, Uzbekistan.
| Email: | 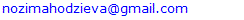 |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this study, morphological changes in the spleen tissue were investigated in the context of chemotherapy-induced renal failure, and the effects of soapwort (Saponaria officinalis) root infusion were evaluated as a method of biocorrection. In the experimental model, pathological alterations were observed in the parenchymal and stromal components of the spleen under the influence of chemotherapy, including edema and a decrease in capillary and lymphocyte activity. Treatment with soapwort root infusion resulted in a reduction of inflammatory signs, restoration of lymphocyte populations, and normalization of stromal structure in the spleen tissue. The findings highlight the scientific and practical significance of biocorrection in preserving spleen morphology and supporting immunological stability.
Keywords: Experimental chemotherapy, Renal failure, Spleen, Morphological changes, Biocorrection, Soapwort root infusion, Lymphocytes, Stromal structure
Cite this paper: Khodjiyeva Nozima Shukhratovna, Dustov Sherali Hayatovich, Morphological Changes in the Spleen Tissue After Biocorrection Against the Background of Chemotherapy-Induced Renal Failure in an Experimental Model, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 330-335. doi: 10.5923/j.ajmms.20261601.69.
Article Outline
1. Relevance of the Study
- In recent years, the prevalence of oncological diseases and their treatment methods has been steadily increasing [1]. Despite the effectiveness of chemotherapy, it is often associated with numerous adverse effects, including pathological changes in the liver, kidneys, and immune system organs [2]. Among these, renal failure is one of the most severe and frequently encountered complications of chemotherapy [3,4]. Impaired kidney function negatively affects the activity of various organs and systems, particularly leading to morphological and immunological alterations in spleen tissue [5].The spleen is the body’s primary immune organ, playing a critical role in lymphocyte production and the filtration of immunological components of the blood [6]. In experimental settings, renal failure and chemotherapy-induced changes in spleen tissue—such as a decrease in lymphocyte populations, damage to stromal and parenchymal components, and activation of inflammatory processes—lead to reduced immune function and overall weakening of the body’s resistance [7,8].From this perspective, restoring the morphological state of spleen tissue and normalizing its functions is of significant relevance [9]. Biocorrection agents, including natural plant extracts and infusions, offer the potential to support immune and parenchymal systems while minimizing adverse effects on the body. Soapwort (Saponaria officinalis) root infusion, traditionally known for its antioxidant and immunomodulatory properties, has not been scientifically studied for its effectiveness in restoring spleen tissue [10,11]. Therefore, studying the morphological changes in spleen tissue under chemotherapy-induced renal failure and evaluating the therapeutic potential of soapwort root infusion as a biocorrection agent is of great scientific and practical importance [12,14]. The findings of this research may serve as a foundation for preventing immune and parenchymal disorders associated with chemotherapy, guiding the selection of biocorrection agents, and developing recommendations for clinical practice [13].Purpose of the study: To investigate the morphological changes in spleen tissue under chemotherapy-induced renal failure and to evaluate the effectiveness of soapwort root infusion as a biocorrection agent in restoring spleen tissue structure and normalizing immune functions.
2. Materials and Methods
- The experimental study was conducted at the Experimental and Research Laboratory of Bukhara State Medical Institute. Healthy non-pedigreed rats aged 18 months and weighing 180–250 g were used in the study. The animals were kept under standard laboratory conditions (temperature 22–25 °C, 12-hour light/dark cycle, ad libitum access to water and a balanced diet) and were allowed an adaptation period prior to the start of the experiment.A model of renal failure was experimentally induced using nephrotoxic chemotherapeutic agents. For this purpose, chemotherapy drugs (e.g., cisplatin or similar nephrotoxic substances) were administered parenterally in defined doses and intervals. Throughout the study, the animals’ general condition, body weight, water and food intake, and clinical signs were regularly monitored. At the end of the experimental period, the animals were euthanized in accordance with ethical guidelines (under brief ether anesthesia), and their spleens were harvested for subsequent histological examination.Within the study design, the laboratory rats were divided into four groups:Group I (Control group): Healthy animals, no treatment administered.Group II (Chemotherapy group): Animals treated with experimental chemotherapy.Group III (Chemotherapy + Renal Failure): Animals with chemotherapy-induced renal failure.Group IV (Biocorrection group): Animals treated with soapwort (Saponaria officinalis) root infusion.Renal failure model: Experimental renal dysfunction was induced after chemotherapy exposure using nephrotoxic agents and/or disruption of water-electrolyte balance.Biocorrection: Soapwort root infusion was administered orally to Group IV animals daily in a dose adjusted to body weight (mg/kg) for 14 days.Throughout the experiment, general health status, body weight, feeding activity, and behavioral patterns of the animals were monitored. The development of renal failure was confirmed by biochemical analysis, including measurement of serum creatinine and urea levels. At the end of the study period, the animals were euthanized under anesthesia in accordance with bioethical standards, and spleen tissues were collected for morphological analysis.The spleen samples were fixed in 10% formalin solution, embedded in paraffin blocks, and sectioned at a thickness of 4–5 μm. Histological examination was performed using hematoxylin and eosin (H&E), Van Gieson, and other special staining methods. Microscopic observations were carried out with modern light microscopes, and morphometric measurements were performed using digital image analysis software.The ratio of white to red pulp, architecture of lymphoid follicles, condition of the stroma, vascular changes, and distribution of immunocompetent cells in the spleen were evaluated. Statistical analysis of each parameter was conducted, and results were expressed as arithmetic mean (M), standard error of the mean (m), and significance levels (p).Statistical analysis: Data were processed using Microsoft Excel and SPSS software. Intergroup differences were determined using Student’s t-test or the Mann–Whitney U test. Results were considered statistically significant at p < 0.05.
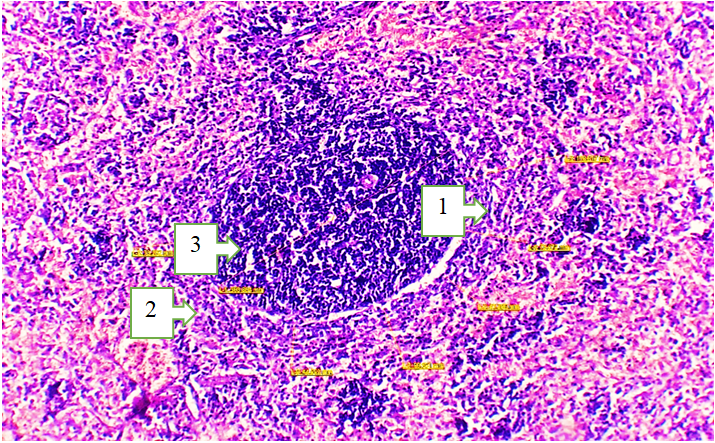
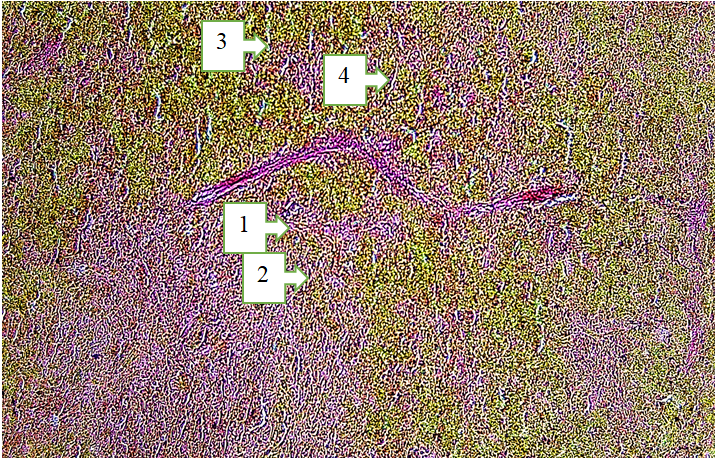
3. Results and Discussion
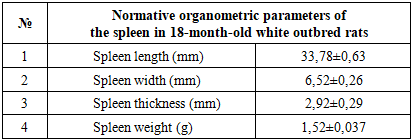
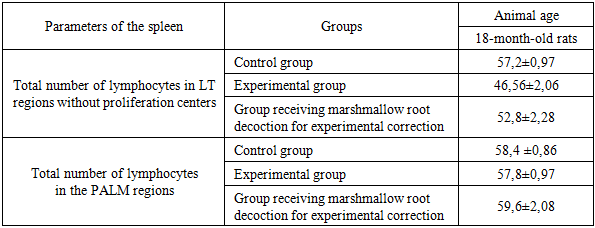
- The spleens of 18-month-old healthy non-pedigreed rats were examined, and the following data were obtained:The body weight of 18-month-old animals ranged from 290 g to 320 g, with a mean of 312.26 ± 3.68 g. The absolute spleen weight ranged from 1.3 g to 1.7 g, with a mean of 1.52 ± 0.037 g. The spleen weight index varied between 0.333% and 0.552%, averaging 0.455 ± 0.024%.The spleen length ranged from 29.4 mm to 35.2 mm, with an average of 33.78 ± 0.63 mm, showing a growth rate of −22.0%. The spleen width ranged from 4.6 mm to 7.4 mm, averaging 6.52 ± 0.26 mm. The spleen thickness ranged from 2.8 mm to 3.9 mm, with a mean of 2.92 ± 0.29 mm, showing a growth rate of 243.3%.
|
|
|
4. Conclusions
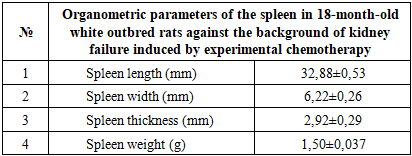
- Analysis of the anatomical and morphological parameters of the spleen in 18-month-old healthy white outbred rats showed an average spleen length of 33.78 ± 0.63 mm, width 6.52 ± 0.26 mm, and thickness 2.92 ± 0.29 mm, with a spleen weight of 1.52 ± 0.037 g. The relative area of white pulp was 20.54 ± 0.69%, and connective tissue elements 6.22 ± 0.22%. The number of lymphocytes and germinal center volumes were within the normal range, indicating preserved immune function of the spleen.Experimental chemotherapy-induced renal failure caused significant changes in the anatomical and morphological parameters of the spleen. Spleen length and width slightly decreased, thickness was maintained, the relative area of white pulp decreased, and the number of lymphocytes in LF and PALS decreased. In the red pulp, erythrocyte accumulation and sinusoid expansion, thickening of trabeculae and capsule, and hyaline and fibrinoid changes in the vascular walls were observed. These changes indicate impaired immune and parenchymal function of the spleen.In the biocorrection group treated with soapwort root infusion, the anatomical and morphological parameters of spleen tissue approached normal. Spleen length and thickness increased, lymphocyte numbers in white pulp increased, and LF and PALS lymphocyte populations were restored. Edema and vascular congestion in the red pulp decreased, and macrophage phagocytic activity partially recovered. These results confirm the positive effect of soapwort root infusion in biocorrecting spleen tissue and supporting immune function.Chemotherapy-induced renal failure significantly disrupts the morphological and immune parameters of the spleen. As a biocorrection agent, soapwort root infusion helps normalize spleen tissue, restore lymphocyte populations, and support immune function, confirming the scientific and practical significance of the study.
References
| [1] | Abbas, A. K., Lichtman, A. H., & Pillai, S. (2018). Cellular and Molecular Immunology (9th ed.). Elsevier. |
| [2] | Almasry Y. et al. Kidney injury: the spleno-renal connection and splenic tyrosine kinase // Journal of Nephrology. – 2024. – С. 1-10. |
| [3] | Ahrorovna K. D. Evaluation of the effect of a genetically modified product on the morphological parameters of the spleen of experimental animals // Academicia: an international multidisciplinary research journal. – 2021. – Т. 11. – №. 1. – С. 885-888. |
| [4] | Banach M., Juranek J. K., Zygulska A. L. Chemotherapy‐induced neuropathies—a growing problem for patients and health care providers // Brain and behavior. – 2017. – Т. 7. – №. 1. – С. e00558. |
| [5] | Chabner, B. A., & Longo, D. L. (Eds.). (2011). Cancer Chemotherapy and Biotherapy: Principles and Practice (5th ed.). Lippincott Williams & Wilkins. |
| [6] | Chiruvella V., Annamaraju P., Guddati A. K. Management of nephrotoxicity of chemotherapy and targeted agents: 2020 // American journal of cancer research. – 2020. – Т. 10. – №. 12. – С. 4151. |
| [7] | Cesta, M. F. (2006). Normal structure, function, and histology of the spleen. Toxicologic Pathology, 34(5), 455-465. |
| [8] | D'Amico, G., & Bover, J. (Eds.). (2015). Kidney Pathology in the Context of Experimental Models. Springer. |
| [9] | Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. (2012). KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney International Supplements, 2(1), 1-138. |
| [10] | Kashyap, S., et al. (2019). Nephrotoxic chemotherapy induces alterations in splenic immune cells in a murine model. Frontiers in Immunology, 10, 192. |
| [11] | Perazella, M. A. (2014). Oncologic drugs and the kidney: what the oncologist needs to know. American Journal of Kidney Diseases, 64(4), 633-644. |
| [12] | Ramos-Vara, J. A. (2014). Technical aspects of immunohistochemistry. Veterinary Pathology, 51(1), 42-53. |
| [13] | Savage, K. J., & Rimsza, L. M. (2017). Immunohistochemistry in Lymphoma Diagnosis. Seminars in Diagnostic Pathology, 34(3), 200-217. |
| [14] | Zraik I.M., Heß-Busch Y. Management of chemotherapy side effects and their long-term sequelae // Urologe A. 2021; 60(7): 862-871. DOI: 10.1007/s00120021-01569-7. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML