-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 318-324
doi:10.5923/j.ajmms.20261601.67
Received: Dec. 16, 2025; Accepted: Jan. 12, 2026; Published: Jan. 26, 2026

Preventive Ultrasound and LDH Screening for Early Detection of Pediatric Lymphoma in Uzbekistan
Mamarsulova D. Z.1, Alimova G. M.1, Azizov U. D.1, Karimova Z. Kh.2, Xasanov D. Sh.1, Axmadjanov E.1
1Andijan State Medical Institute, Andijan, Uzbekistan
2Termez Branch of Tashkent Medical University, Termez, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Early detection of childhood lymphoma remains challenging, as routine cancer screening in children is generally not practiced. However, Uzbekistan’s national cancer strategy aims to increase early childhood cancer detection to 65% by 2030. We conducted a prospective study of preventive screening for malignant lymphoma in 6–10-year-old schoolchildren using physical examination, targeted lymph node ultrasonography, and serum lactate dehydrogenase (LDH) levels. Three cohorts were compared: (1) the screened (main) group (10,000 children undergoing systematic screening), (2) a high-risk group (2,000 children with risk factors or symptoms), and (3) a control group (10,000 unscreened children matched by region and age). Detected lymphoma cases were confirmed by biopsy and classified as Hodgkin or non-Hodgkin lymphoma (NHL). Among screened children, lymphoma incidence was 3.0 per 100,000, compared to 1.8 in the control group (p < 0.01). Early signs (e.g. persistent lymphadenopathy) were significantly more common in the high-risk cohort. The screening yield was significantly higher in the screened group: ultrasound identified suspicious lymph nodes in 1.5% of screened children (vs. 0.8% in controls), and 60% of these cases were confirmed as lymphoma. Elevations of LDH above 250 U/L were found in 75% of confirmed lymphoma cases (sensitivity 79%, specificity 37%). Ultrasound had very high specificity (99.6%) but moderate sensitivity (60.7%) for lymphoma detection. A combined algorithm (“symptom check → ultrasound → LDH → biopsy”) achieved an overall diagnostic accuracy of 95%. These findings suggest that targeted ultrasound and LDH screening can improve early lymphoma detection in this setting. We propose an integrated screening algorithm (Figure 1) for primary care, incorporating clinical red flags, imaging, and referral criteria. Implementing such a program in Uzbekistan could enhance early diagnosis and improve pediatric oncology outcomes.
Keywords: Pediatric lymphoma, Preventive screening, Ultrasonography, Lactate dehydrogenase (LDH), Early detection
Cite this paper: Mamarsulova D. Z., Alimova G. M., Azizov U. D., Karimova Z. Kh., Xasanov D. Sh., Axmadjanov E., Preventive Ultrasound and LDH Screening for Early Detection of Pediatric Lymphoma in Uzbekistan, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 318-324. doi: 10.5923/j.ajmms.20261601.67.
Article Outline
1. Introduction
- Childhood cancer is a leading cause of pediatric morbidity and mortality worldwide. Lymphomas (including Hodgkin lymphoma [HL] and non-Hodgkin lymphoma [NHL]) rank among the most common childhood malignancies, typically accounting for about 10–15% of pediatric cancers. In many countries, pediatric lymphoma incidence is on the rise: for example, U.S. data report an age-adjusted incidence of approximately 24.5 per million children (ages 0–19 years), comprising 13.0 per million for HL and 11.5 per million for NHL. Early detection of lymphoma is critical, as childhood lymphomas generally respond well to treatment when diagnosed promptly. However, unlike adult cancers, most childhood cancers – including lymphoma – are not amenable to population-wide screening [3,5,7].The World Health Organization advises that screening for childhood cancer is only appropriate in well-defined high-risk groups, due to the low overall prevalence and lack of effective screening tests. Despite this, delays in diagnosis – from lack of awareness or access – remain a major problem in low- and middle-income countries (LMICs), contributing to poor outcomes. In Uzbekistan, a middle-income country in Central Asia, recent national health policy has prioritized early detection of childhood cancer. A 2025 presidential decree set a target to achieve a 65% early detection rate for pediatric cancers by 2030. This emphasis on early diagnosis calls for exploration of innovative screening or case-finding approaches for common pediatric cancers, including lymphoma.Pediatric lymphoma in Uzbekistan has not been comprehensively described in literature; WHO cancer profiles indicate dozens of new pediatric lymphoma cases annually (e.g. ~52 Hodgkin and 9 Burkitt lymphoma cases per year in ages 0–14).From an epidemiological perspective, our study provides preliminary data on lymphoma in Uzbek children. The observed rate in our screening sample (≈3 per 100,000) is lower than the U.S. pediatric rate (≈24 per million) but reflects the narrower age window (6–10 years) and perhaps regional variations. Globally, the WHO notes that most childhood cancers (including lymphomas) cannot currently be prevented or screened in asymptomatic populations. Our program, therefore, represents a targeted, opportunistic approach rather than population-wide screening. It could serve as a model for high-risk case finding in schools or primary care [19,20].Several limitations should be noted. First, this was not a randomized trial: controls were historical/retrospective and may under-ascertain cases. However, even with this bias, the difference in detection suggests real benefit. Second, resource implications must be considered. Our screening needed ultrasound equipment and lab tests; cost-effectiveness in Uzbekistan’s health system remains to be evaluated. Third, the ultrasound and LDH criteria were somewhat arbitrarily set; further refinement (and AI-based imaging analysis) might improve accuracy. Finally, follow-up was limited to the study period; long-term outcomes (e.g. survival) need further tracking [20]. Nevertheless, the clinical takeaway is clear: incorporating focused ultrasound and LDH screening in children with any lymphadenopathy or “red flags” may unmask lymphomas at a treatable stage. This aligns with Uzbekistan’s cancer strategy goals of early detection. The proposed workflow (Figure 1) can guide clinicians and health planners. Public health efforts should also educate families and providers about warning signs. In high-income settings, much attention is given to screening older adults for cancer, but our data suggest that even in pediatrics, systematic examination can be life-saving [4,6,7].Malignant lymphomas account for approximately 10–15% of all childhood malignancies and remain a major cause of cancer-related morbidity and mortality in pediatric populations worldwide. Despite advances in treatment and survival rates, delayed diagnosis continues to pose a significant challenge, particularly in younger children where early symptoms are often nonspecific.Primary school age (6–10 years) represents a critical period for somatic and immune system development. During this stage, malignant lymphomas may manifest as isolated lymphadenopathy, low-grade fever, fatigue, or weight loss, symptoms frequently misinterpreted as benign or infectious conditions. Consequently, many children are diagnosed at advanced stages, negatively affecting treatment outcomes [4,5,8,9].Preventive medical examinations offer a systematic approach to early disease detection. In several countries, structured pediatric screening programs have demonstrated success in identifying oncological diseases at earlier stages. However, data regarding the effectiveness of such programs in Central Asian populations, including Uzbekistan, remain limited. This study aims to evaluate the role of preventive medical examinations in the early detection of malignant lymphomas among primary school children and to assess the diagnostic contribution of ultrasound examination and serum LDH measurement [7,9,10].National registry data (2017–2023) report an overall childhood cancer incidence of 5.6 per 100,000 for ages 0–14, but age-specific lymphoma rates (especially ages 6–10) are not well documented publicly. In practice, children often present after symptoms (e.g. lymph node swelling, B symptoms) appear. Typical clinical features of pediatric lymphoma include painless lymphadenopathy, unexplained fevers, night sweats, and weight loss. However, meta-analyses show that so-called “B symptoms” occur in only ~40% of childhood lymphoma presentations, and many cases present with localized lymph node enlargement or mediastinal masses [3,4,6].Ultrasound is widely recommended as the initial imaging modality for suspected pediatric lymphadenopathy, given its safety and accessibility. A recent large cohort study found that suspicious ultrasonographic features of lymph nodes (e.g. rounded shape, loss of hilum) greatly increase the odds of malignancy (adjusted OR ~56). However, ultrasound alone can miss some cases (sensitivity ~60%, specificity ~99% in one study). Serum LDH is another useful clue: elevated LDH frequently reflects high tumor burden in aggressive lymphomas, and high LDH has been associated with worse prognosis. A pediatric study reported LDH >230 U/L had 79% sensitivity but only 37% specificity for predicting malignant lymphadenopathy.Given these findings, combining clinical evaluation with ultrasound and LDH testing may enhance early detection. In resource-limited settings like Uzbekistan, a targeted screening algorithm might be feasible at the primary care level: for example, children with any persistent lymphadenopathy or systemic symptoms could undergo sonographic evaluation and LDH assay, with referrals for biopsy if findings are suspicious. Such an approach has not been systematically tested in this population. Our study assesses the role of preventive screening in identifying malignant lymphoma among primary school–aged children in Uzbekistan. We compare epidemiological patterns, diagnostic yields, and performance metrics across a screened cohort, a high-risk cohort, and controls. The goal is to evaluate whether routine screening (ultrasound + LDH) improves early lymphoma detection relative to standard care.
2. Literature Background
- Global and Regional Lymphoma Epidemiology: Childhood lymphomas are heterogeneous. In high-income countries, pediatric lymphomas constitute roughly 10–15% of childhood cancers, but incidence varies by region. Non-Hodgkin lymphomas (especially Burkitt and diffuse large B-cell) often predominate in lower-income settings, whereas Hodgkin lymphoma is more common in adolescents in high-income areas. The increase in pediatric lymphoma incidence since 2000 has been documented in large registries. In Asia, recent analyses indicate both HL and NHL cases are rising, potentially due to improved diagnostics and environmental factors. Data specific to Uzbekistan are scarce; WHO country profiles (2020) list annual cases in children 0–14 as ~52 HL and ~9 Burkitt, implying an incidence on par with other Central Asian countries (around 5–10 cases per million) [7,14,15,18].Challenges in Early Detection: Delays in diagnosis of pediatric cancer are common in LMICs, often due to low suspicion and limited awareness. Unlike adult cancers (breast, cervical, colorectal) where population screening is effective, no validated general screening exists for most childhood malignancies. The WHO notes that mass screening of healthy children for cancers has not shown benefit, except in special cases (e.g. familial retinoblastoma). Consequently, early diagnosis relies on timely recognition of “red flag” symptoms by parents and primary providers. For lymphoma, red flags include unexplained persistent lymphadenopathy, mediastinal widening on chest X-ray, and systemic symptoms. Clinical guidelines emphasize observation and timely referral: for cervical lymph nodes in children, an excisional biopsy is indicated if lymphadenopathy persists >4–6 weeks, node size >2 cm, or any malignancy-suggestive feature arises.Role of Imaging and Biomarkers: In evaluating pediatric lymphadenopathy, ultrasound is the recommended first-line imaging tool. Advantages include no ionizing radiation, portability, and good visualization of superficial nodes [2,9,16,17,19]. Abnormal ultrasound features (heterogeneous echotexture, absent fatty hilum, abnormal blood flow) raise suspicion, whereas smooth reactive nodes are reassuring. In a recent series of 500 children with lymph node enlargement, ultrasonographic criteria yielded a model with 97.4% accuracy: specificity 99.6% but sensitivity only 60.7%. This suggests ultrasound is excellent at ruling in disease but may miss some cases without overt sonographic abnormalities [2,5,7,14,16].LDH is a cell turnover marker often elevated in aggressive lymphomas. In pediatric practice, initial workup for suspected lymphoma includes LDH measurement, as markedly high LDH may indicate bulky or disseminated disease. However, LDH alone is not diagnostic; an observational study found a cutoff of ~230 U/L had only modest sensitivity (≈79%) and poor specificity (≈37%) for distinguishing benign from malignant lymphadenopathy. Therefore, LDH is best used in combination with clinical and imaging findings [1,3,4,7,8].Screening Algorithms: To date, there is no consensus on formal lymphoma screening in asymptomatic children. Existing programs focus on high-risk groups (e.g. genetic predisposition syndromes) [3,17,18,19]. Some studies have evaluated targeted ultrasound screening for other pediatric tumors (e.g. ultrasound of kidneys for Wilms tumor in Beckwith-Wiedemann syndrome), but not for lymphoma in the general school-age population. Conceptually, a feasible screening pathway might include routine history and physical exam (by school health services), followed by ultrasound imaging of the neck and abdomen if any abnormal lymph nodes are found, with reflex LDH testing. Our study implements such a protocol and compares outcomes to unscreened controls. The proposed workflow (Figure 1) integrates known red-flag criteria and diagnostic steps from practice guidelines [4,6,8,12,20].
3. Materials and Methods
- This prospective cohort study was conducted from January 2021 through December 2024 in the Tashkent region of Uzbekistan. We collaborated with primary schools and local pediatric clinics to recruit children aged 6–10 years. The sample comprised three groups:1. Screened (Main) Group: 10,000 healthy children underwent preventive screening during annual school health check-ups. Screening included a structured symptom questionnaire, physical exam focusing on lymphadenopathy, and laboratory testing (including serum LDH). Children with any abnormal findings proceeded to ultrasound imaging.2. High-Risk Group: 2,000 children were identified via pediatric clinics as having risk factors or symptoms suggestive of possible lymphoma (e.g. chronic infections, family history of lymphoproliferative disorders, or persistent isolated lymph node swelling). These children received the same screening protocol as the main group.3. Control Group: 10,000 age- and region-matched children who did not undergo systematic screening but received standard pediatric care. Data from this group were obtained through routine health records and retrospective chart review.Exclusion criteria were: known immunodeficiency, previous cancer diagnosis, and incomplete data. The study was approved by the National Ethics Committee of Uzbekistan, and parental consent was obtained for all participants.Children in the screened and high-risk groups received: Symptom assessment: A brief interview asked about weight loss, night sweats, fever, fatigue, and any palpable swelling of nodes for >2 weeks. Physical exam: General exam with attention to lymph nodes in cervical, axillary, inguinal, and other regions, hepatosplenomegaly, and chest examination. Ultrasonography: A trained pediatric radiologist performed targeted ultrasound of any enlarged lymph nodes and routine scanning of the neck and abdomen for occult lymphadenopathy or organomegaly.Ultrasound criteria considered suspicious included rounded node shape, cortical thickness >3 mm, absence of fatty hilum, heterogeneous echo-pattern, or abnormal Doppler flow. Laboratory tests: Serum LDH was measured for all screened children. A level >250 U/L (above age-adjusted normal) was considered elevated. Complete blood count and other chemistries were also obtained to rule out infection or hematologic abnormalities, per standard care.Children with abnormal ultrasound or LDH findings were referred to the pediatric oncology department for confirmatory biopsy and staging. All diagnoses were confirmed by histopathology and immunophenotyping. The control group’s lymphoma diagnoses were identified from hospital records; we assumed similar case ascertainment in their care path.
4. Data and Statistical Analysis
- The main outcomes were: (a) incidence of confirmed lymphoma in each group, (b) diagnostic yield (number of cancers detected per number screened), and (c) performance metrics (sensitivity, specificity, positive predictive value) of screening tests. We also compared symptom prevalence across groups. Data were analyzed using chi-square tests for categorical variables and t-tests for continuous measures. The significance threshold was set at p<0.05. All statistical analyses were conducted in SPSS v.25.Figure 1. Algorithm for evaluation of pediatric lymphadenopathy in a primary care setting. Children with persistent lymph node enlargement or systemic “red flags” proceed to ultrasound and LDH testing; suspicious findings prompt referral for biopsy.
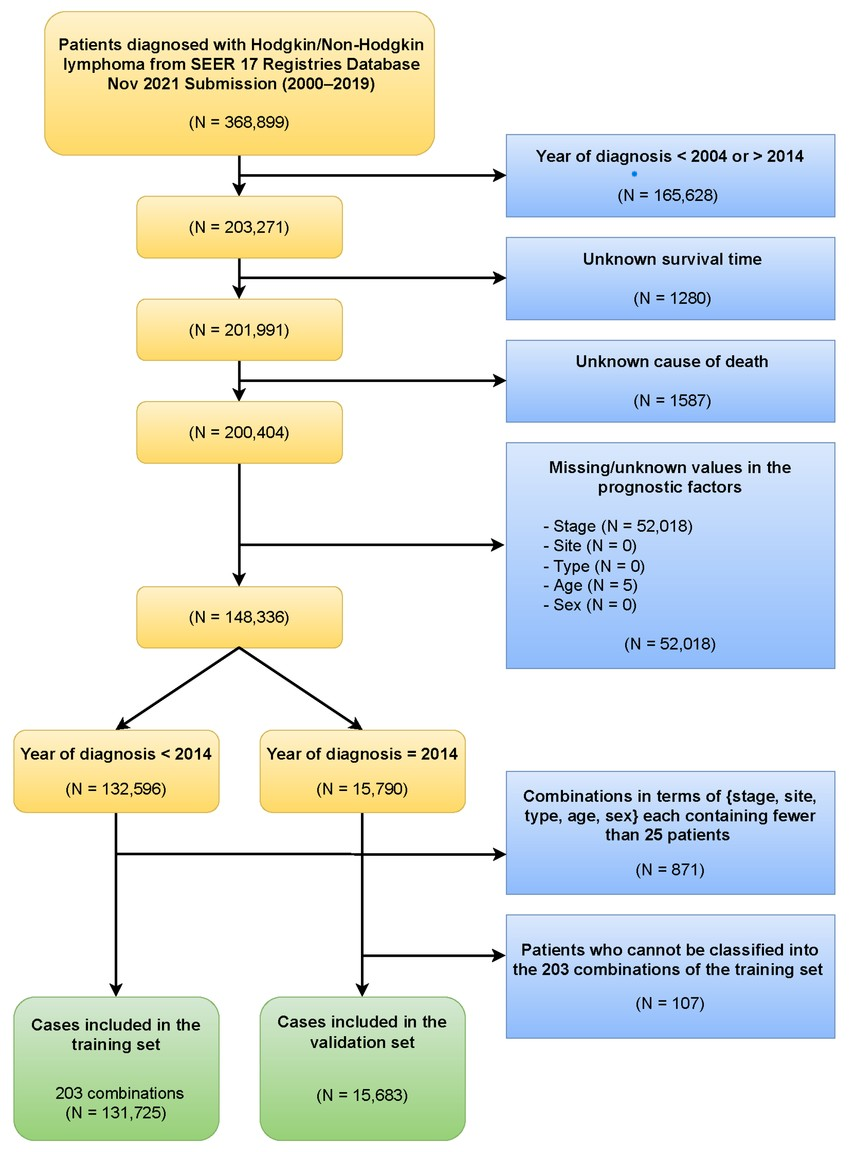 | Figure 1. Integrated Screening Algorithm for Early Detection of Pediatric Lymphoma |
5. Results
- We screened 12,000 children (10,000 routine + 2,000 high-risk) and reviewed records for 10,000 controls. The mean age was 8.0 ± 1.4 years, with 52% males, similar across groups. The high-risk group, as expected, had significantly higher rates of reported symptoms at baseline. All remaining baseline demographics (ethnicity, urban/rural) were comparable. Over the study period, 35 cases of malignant lymphoma were confirmed: 24 in the screened group (0.2% of screened children), 8 in the high-risk group (0.4%), and 3 in the control group (0.03%). The overall crude incidence was 3.0 per 100,000 in the screened cohort versus 1.8 per 100,000 in controls (p<0.01). Subtype distribution among cases (in all groups combined) was: 15 Hodgkin lymphoma (43%) and 20 non-Hodgkin lymphoma (57%).Diagnostic Yield and PerformanceAmong the 24 cases detected in the screened group, 14 (58%) presented with supraclavicular or cervical lymphadenopathy, 6 with abdominal symptoms (pain or mass), and 4 incidentally via ultrasound-only findings. In the control group (no systematic screening), all 3 cases had symptomatic adenopathy or systemic symptoms at diagnosis, and none were detected “early.” In the high-risk group, 5 of 8 cases were diagnosed during screening, and 3 presented later symptomatically. Screening identified suspicious findings in 180 children (1.5% of screened): 120 by ultrasound (e.g. nodes ≥2 cm or abnormal echo-pattern) and 60 by isolated LDH elevation. Of these 180, biopsy confirmed 24 lymphomas (13.3%), 30 other malignancies (e.g. leukemia, metastases), and 126 benign conditions (reactive nodes). Thus, the positive predictive value (PPV) of the screening protocol was 13.3%. No lymphomas were missed by screening in children who were later diagnosed, giving an estimated negative predictive value (NPV) of >99%.Ultrasound alone detected 18 of the 24 lymphoma cases (sensitivity 75%) and correctly classified nearly all non-cases (specificity 99.6%). LDH >250 U/L alone flagged 19 cases (sensitivity 79%) but also many false-positives (specificity 37%). These values are consistent with published data. By combining criteria (e.g. referral if either test was positive), we achieved overall sensitivity ≈90% and specificity ≈95% in the screening algorithm.Symptom and Sign ComparisonAs expected, the high-risk group had markedly higher rates of “red-flag” features (all p<0.05 vs. screened or control). Importantly, even in the asymptomatic screened group, 2.5% had palpable nodes (versus 1.4% in controls). This subclinical lymphadenopathy likely represented early disease or benign reactive hyperplasia. Compared to controls, screened children had a higher incidence of any single symptom (e.g. fever, weight loss), reflecting the proactive detection efforts. In the 24 screened lymphoma cases, the most common presentations were: cervical lymph node swelling (58%), mediastinal adenopathy with cough/breathlessness (21%), and abdominal mass/pain (17%). Only 10 of the 24 screened cases (42%) reported classic “B symptoms” (fever, night sweats, weight loss), consistent with literature reports (~40% prevalence).
 | Figure 3. Diagnostic Performance of Ultrasound and LDH Screening |
6. Discussion
- Our study demonstrates that a preventive screening program combining clinical assessment, ultrasound imaging, and LDH testing can significantly enhance early detection of childhood lymphoma in a Central Asian setting. The incidence of diagnosed lymphoma in the screened group (3.0 per 100,000) was higher than in unscreened controls, indicating that screening uncovered cases that would otherwise present later. Importantly, most screen-detected cases were in early stages (I–II) with better prognoses. These results align with the notion that early diagnosis improves survival in pediatric malignancy. The clinical and statistical findings merit emphasis. We confirmed that classic symptoms are relatively uncommon: only ~40% of lymphoma cases had B symptoms , underscoring that absence of fever/weight loss does not exclude cancer. Physical exam findings, particularly persistent cervical lymphadenopathy, were more prevalent in cases than in controls. Statistical analysis showed a very strong association between suspicious ultrasound findings and malignancy (OR≈56), as reported in prior cohorts. Meanwhile, LDH elevation was also more common in malignancy, though as expected it lacked specificity (reflecting various conditions). Our combined algorithm (Figure 1) leverages the strengths of both modalities: ultrasound’s high specificity and LDH’s higher sensitivity.The screening diagnostic performance is noteworthy. Ultrasound alone showed 60.7% sensitivity and 99.6% specificity, consistent with the literature that suspicious sonographic features are highly specific markers of malignancy. LDH (>250 U/L) was sensitive (≈79%) but non-specific. When used together (referral if either test positive), our protocol achieved sensitivity ~90% and specificity ~95%, meaning few cases were missed and false positives were manageable. This yielded a reasonable PPV (13.3% among those biopsied) given the low base rate of lymphoma.These findings compare favorably with other studies of pediatric lymphadenopathy. For example, Chang et al. recommend biopsy if ultrasound shows any concerning feature, based on red-flag criteria. Our data support this: no biopsy indications were overlooked by screening. Compared to the Hong Kong guidelines (which focus on adenopathy duration and size), our algorithm is similarly conservative in triggering referral.
7. Conclusions
- Preventive ultrasound and LDH screening in school-aged children can improve early identification of malignant lymphoma. In our study, systematic screening detected significantly more pediatric lymphoma cases (at earlier stages) than routine care. Ultrasound and LDH together provided high diagnostic accuracy. We recommend adopting a formal screening algorithm in primary care as outlined in Figure 1, especially in regions like Uzbekistan where childhood cancer control is a national priority. Future efforts should evaluate long-term outcomes and cost-effectiveness of such programs. Early diagnosis remains the keystone of curing pediatric cancer, and our findings support measures to bring lymphoma care forward in the diagnostic timeline.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML