-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 305-311
doi:10.5923/j.ajmms.20261601.65
Received: Dec. 26, 2025; Accepted: Jan. 12, 2026; Published: Jan. 26, 2026

Surgical Strategy for the Treatment of Atrial Fibrillation in Patients with Mitral Valve Disease
Khamiljanov J. N., Aliyev Sh. M., Muchiashvili O. R.
Department of Adult Cardiac Surgery of Republican Specialized Scientific and Practical Medical Center for Surgery named after Academician V. Vakhidov Tashkent, Uzbekistan
Correspondence to: Khamiljanov J. N., Department of Adult Cardiac Surgery of Republican Specialized Scientific and Practical Medical Center for Surgery named after Academician V. Vakhidov Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Atrial fibrillation (AF) is one of the most common and clinically significant complications of mitral valve disease, markedly worsening hemodynamics, prognosis, and patients’ quality of life. Despite advances in pharmacological therapy and interventional techniques, surgical treatment of AF in this category of patients remains the most effective method for restoring and maintaining sinus rhythm. The aim of this study was to evaluate the effectiveness of various surgical strategies for the treatment of atrial fibrillation in patients with mitral valve disease who underwent surgical correction of the mitral valve. The study included patients with mitral valve disease complicated by atrial fibrillation who underwent mitral valve correction combined with surgical ablation. Clinical, echocardiographic, and electrophysiological parameters were analyzed, as well as the rate of sinus rhythm restoration and early and long-term postoperative outcomes. The effectiveness of different surgical approaches was assessed taking into account the form of atrial fibrillation, the duration of the arrhythmia, and the degree of atrial remodeling. The results demonstrated that combined surgical treatment of mitral valve disease and atrial fibrillation leads to a statistically significant increase in the rate of sinus rhythm restoration and improvement in patients’ functional status. The most pronounced clinical effect was observed with the use of extended surgical ablation strategies in patients with long-standing persistent AF. The optimal choice of a surgical strategy for the treatment of atrial fibrillation in patients with mitral valve disease should be based on individual clinical and morphological characteristics and allows for a substantial improvement in long-term surgical outcomes.
Keywords: Atrial fibrillation, Mitral valve disease, Surgical treatment, Surgical ablation, Maze procedure, Mitral valve repair, Sinus rhythm restoration
Cite this paper: Khamiljanov J. N., Aliyev Sh. M., Muchiashvili O. R., Surgical Strategy for the Treatment of Atrial Fibrillation in Patients with Mitral Valve Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 305-311. doi: 10.5923/j.ajmms.20261601.65.
Article Outline
1. Introduction
- Atrial fibrillation (AF) is the most common sustained supraventricular arrhythmia and represents a serious clinical and socioeconomic problem in modern cardiology and cardiac surgery. The prevalence of AF steadily increases with age and the presence of structural heart disease and is associated with a significant rise in the risk of ischemic stroke, systemic thromboembolism, progression of heart failure, and overall mortality [1]. Mitral valve disease occupies a special place among the causes of AF, as chronic volume and pressure overload of the left atrium leads to pronounced electrical and structural remodeling of the atrial myocardium [2]. According to contemporary studies, AF is detected in 20–42% of patients referred for surgical correction of the mitral valve, with a markedly higher prevalence in cases of rheumatic involvement and long-standing valvular defects [3]. The presence of AF in this patient population is associated with a more severe clinical course, a reduction in functional class, an increased rate of hospitalizations, and an unfavorable long-term prognosis [1,4]. Isolated surgical correction of mitral valve disease, despite restoration of central hemodynamics, generally does not result in spontaneous recovery of sinus rhythm, especially in persistent and long-standing forms of AF. Therefore, over recent decades, the concept of combined surgical treatment has been developed, involving simultaneous mitral valve correction and surgical ablation of AF within a single operative procedure [2,3]. The most extensively studied and clinically validated surgical technique for the treatment of AF is the Cox–Maze procedure, which has evolved into the modern Cox–Maze IV modification based on the use of radiofrequency and/or cryoablation. This technique aims to create lines of electrical conduction block that prevent the circulation of reentry waves within the atria and has demonstrated high efficacy in restoring and maintaining sinus rhythm in both the early and long-term postoperative periods [3,5]. Recent meta-analyses and systematic reviews confirm that performing surgical AF ablation during mitral valve surgery is associated with a significant increase in arrhythmia-free survival, a reduction in stroke risk, and an improvement in patients’ quality of life, without a significant increase in operative mortality or the incidence of major complications [4,5]. Nevertheless, despite strong evidence, surgical AF ablation has not yet been widely implemented in routine clinical practice. This is largely due to the lack of unified standards for selecting the extent of ablation, concerns about prolonging operative time, and heterogeneity of clinical guidelines. In this context, a systematic analysis of contemporary surgical strategies for the treatment of atrial fibrillation in patients with mitral valve disease, as well as an evaluation of their effectiveness, safety, and impact on long-term clinical outcomes, remains highly relevant. The present study is devoted to a review and analysis of current approaches to the surgical treatment of AF in mitral valve pathology with the aim of optimizing management strategies for this patient population.Aim of the study. The aim of this study was to analyze and evaluate modern surgical strategies for the treatment of atrial fibrillation in patients with mitral valve disease, including combined mitral valve interventions and surgical ablation, in order to determine their effectiveness, safety, and impact on early and long-term clinical outcomes, as well as to substantiate the optimal surgical management strategy for this category of patients.
2. Materials and Methods
- Study design and patient population.The study had a retrospective–prospective clinical design and was conducted at the State Institution “Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov.” The analysis included 114 patients with mitral valve disease complicated by atrial fibrillation who underwent surgical treatment between 2018 and August 2022.Inclusion criteria were the presence of hemodynamically significant mitral valve disease (mitral regurgitation, mitral stenosis, or combined mitral valve disease), documented atrial fibrillation (paroxysmal, persistent, or long-standing persistent forms), and surgical correction of mitral valve pathology. Exclusion criteria included repeat mitral valve surgery, severe concomitant congenital heart defects, significant dysfunction of other cardiac valves requiring extended surgical intervention, and the absence of sufficient data for adequate follow-up.Surgical strategy and group allocation.Depending on the extent of surgical intervention performed, patients were divided into the following groups:• patients who underwent isolated mitral valve correction (repair or replacement);• patients who underwent mitral valve correction combined with surgical treatment of atrial fibrillation;• patients who underwent mitral valve replacement combined with radiofrequency ablation using the Cox–Maze IV technique.The choice of surgical approach was individualized, taking into account the clinical form of atrial fibrillation, the duration of the arrhythmia, the anatomical and functional status of the left atrium, and the overall operative risk.Methods of patient evaluation.To achieve the study objectives, a comprehensive set of general clinical, laboratory, instrumental, and specialized diagnostic methods was employed.General clinical evaluation included medical history assessment, evaluation of clinical status, determination of heart failure functional class according to the NYHA classification, and analysis of the postoperative course. Laboratory methods were used to assess the general condition of patients and to monitor metabolic parameters in the preoperative and postoperative periods.Instrumental diagnostic methods included electrocardiography, 24-hour Holter ECG monitoring, and echocardiography. Echocardiographic examinations were performed dynamically and were used to assess structural and functional cardiac parameters, including left ventricular ejection fraction, left ventricular end-diastolic dimension, and left atrial diameter. Particular attention was paid to the assessment of reverse cardiac remodeling in the long-term postoperative period.Restoration and maintenance of sinus rhythm were evaluated using standard ECG recordings and Holter monitoring data obtained in the early and long-term postoperative periods. Recurrences of atrial fibrillation were recorded when arrhythmia episodes were documented objectively.
3. Results of the Study
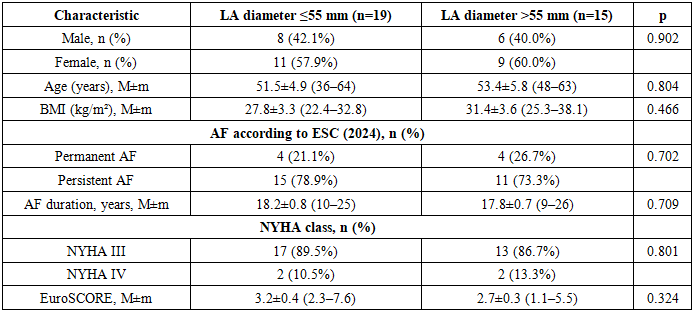
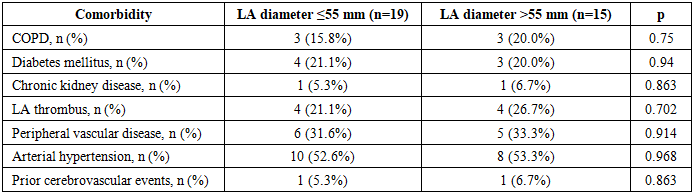
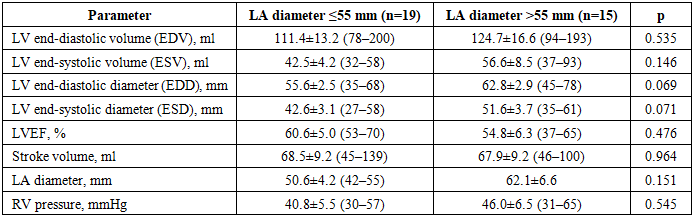
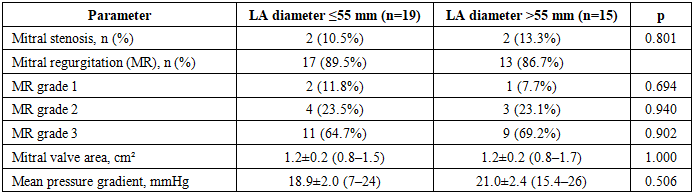
- Comparative characteristics of the groups depending on the baseline left atrial diameter.To assess the prognostic significance of left atrial (LA) size with respect to the risk of atrial fibrillation (AF) recurrence after surgical intervention, a comparative analysis was performed in patients divided into two groups according to baseline LA diameter: ≤55 mm (n = 19) and >55 mm (n = 15).Comparative analysis of clinical and demographic characteristics showed that the groups were comparable in terms of sex, age, body mass index, AF form and duration, NYHA functional class of heart failure, and integrated operative risk assessed by the EuroSCORE system (p > 0.05). This indicates the homogeneity of the study groups and allows the observed postoperative differences to be considered as predominantly related to baseline LA size.
|
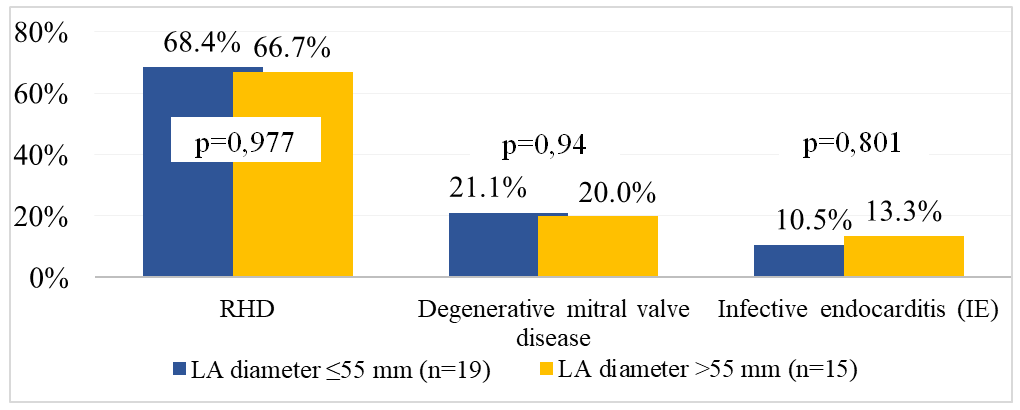 | Figure 1. Primary diagnosis in patients of the study groups depending on left atrial (LA) size |
|
|
|
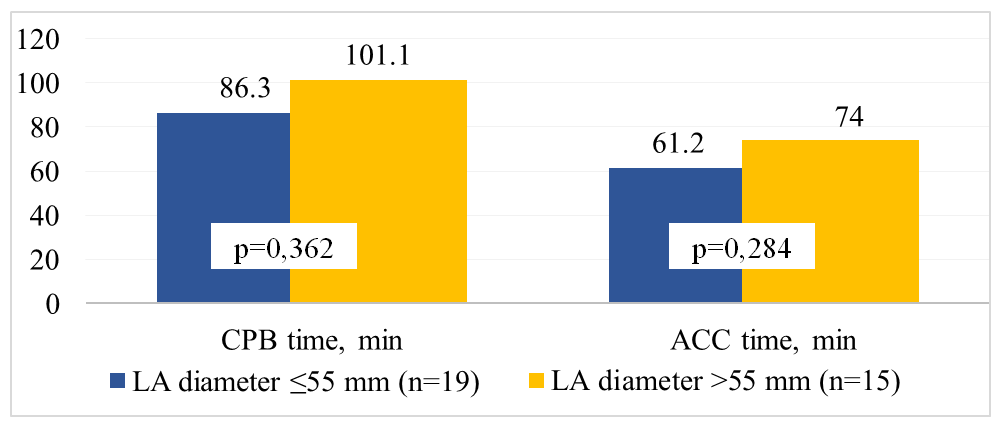 | Figure 2. Comparison of operative characteristics in patients of the study groups depending on left atrial (LA) size |
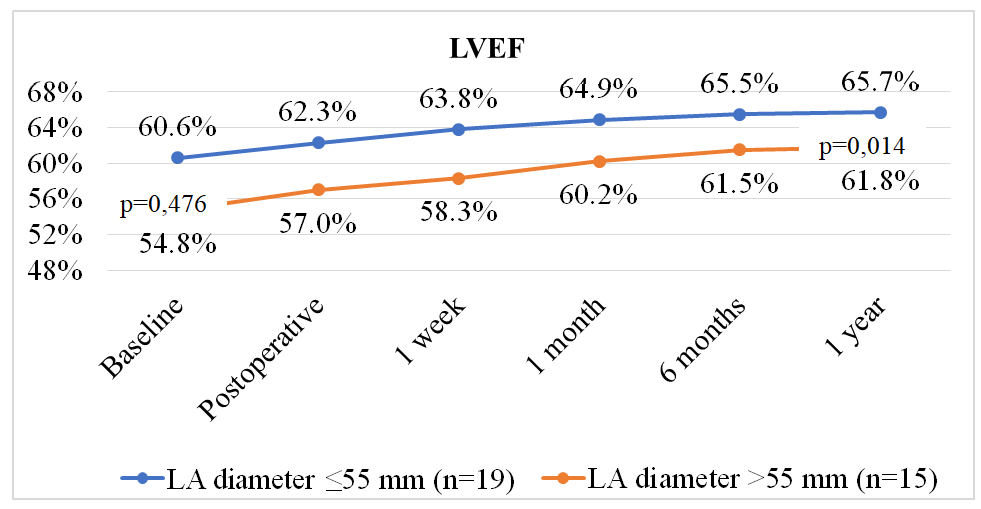 | Figure 3. Comparative analysis of changes in left ventricular ejection fraction (LVEF) dynamics depending on baseline left atrial (LA) size |
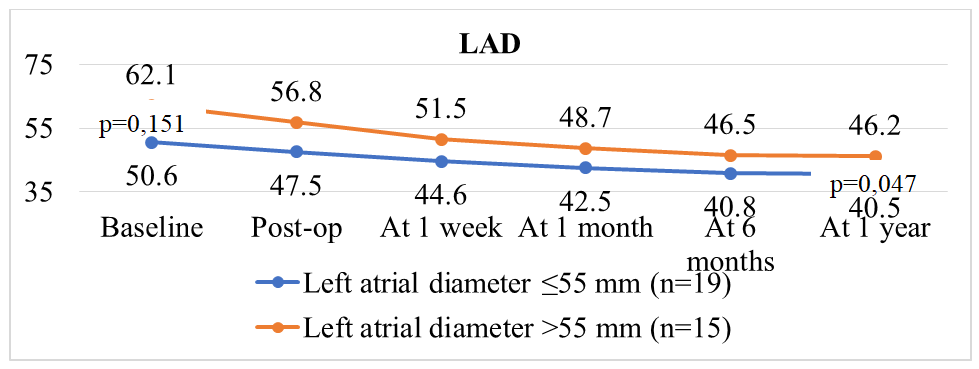 | Figure 4. Comparative analysis of changes in left atrial (LA) diameter dynamics depending on baseline LA size |
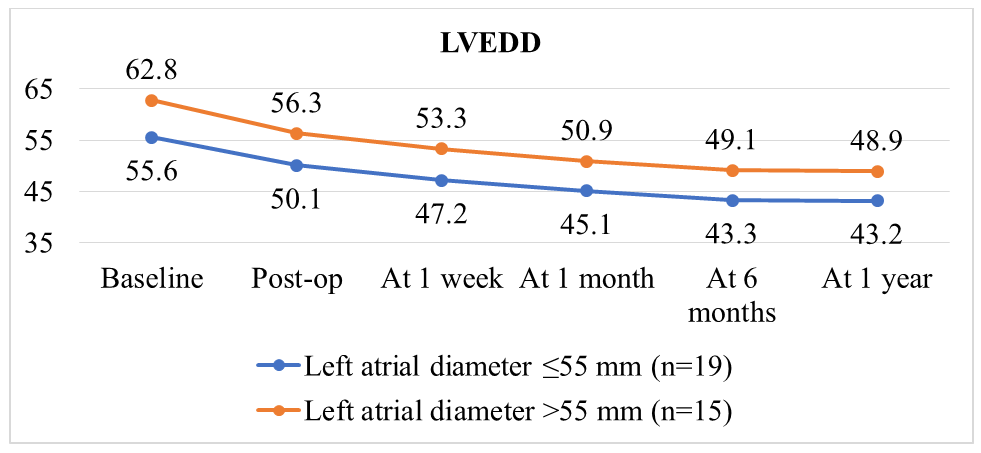 | Figure 5. Comparative analysis of left ventricular end-diastolic diameter (LVEDD) dynamics depending on baseline left atrial (LA) size |
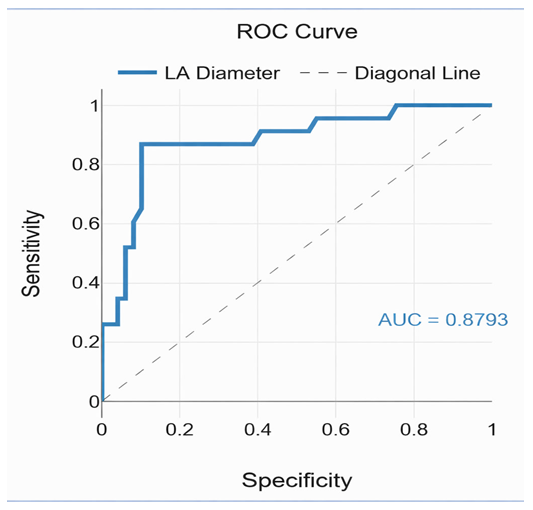 | Figure 6. ROC analysis: left atrial (LA) diameter as a prognostic factor for AF recurrence after surgical treatment in patients with mitral valve disease and atrial fibrillation |
4. Discussion
- Atrial fibrillation (AF) remains one of the most common arrhythmias in patients with mitral valve disease, significantly affecting prognosis and surgical outcomes. The results of this study confirm that baseline left atrial (LA) size is a key factor determining the effectiveness of sinus rhythm restoration and the dynamics of reverse cardiac remodeling after mitral valve surgery combined with radiofrequency ablation using the Cox-Maze IV technique [1,2,11].Our analysis showed that patients with a baseline LA diameter ≤55 mm demonstrated a more pronounced and sustained increase in left ventricular ejection fraction (LVEF), significant reduction in left ventricular end-diastolic diameter (LVEDD), and a decrease in LA diameter in both early and long-term postoperative periods compared with patients with LA >55 mm (p < 0.05). These findings are consistent with contemporary studies, where larger LA size was associated with lower likelihood of long-term sinus rhythm recovery and more pronounced structural atrial remodeling [3,4].The high accuracy of the ROC analysis (AUC = 0.879) and the sensitivity/specificity of the prognostic LA threshold >55 mm (86.7% and 89.8%, respectively) confirm the clinical significance of this parameter. Such metrics allow baseline LA size to be used as a criterion for risk stratification of AF recurrence and for individualizing the extent of surgical intervention [5,6]. Specifically, patients with LA >55 mm can be considered a high-risk group, requiring a more comprehensive surgical approach and intensive postoperative monitoring.The dynamics of LVEF and reverse LA remodeling in patients with smaller baseline sizes emphasize the importance of timely surgical intervention before significant atrial enlargement develops. Mechanistically, this is related to a lower degree of fibrotic changes in the atrial myocardium and a higher capacity for restoration of electrical conduction after creating conduction block lines during ablation [7]. Literature also emphasizes that combining mitral valve correction with surgical ablation (Cox-Maze IV) significantly reduces AF recurrence and improves clinical outcomes compared with isolated valve replacement [8,9].Intraoperative data showed that patients with LA >55 mm more frequently required inotropic and/or vasopressor support, reflecting greater hemodynamic instability. These findings highlight the need for careful preoperative planning and individualized anesthetic and cardiopulmonary management strategies for patients with enlarged LA.Furthermore, the results confirm the high efficacy of a combined surgical strategy: freedom from AF after mitral valve replacement combined with Cox-Maze IV ablation reached 91.2%, consistent with contemporary clinical studies [10]. Improvement in NYHA functional class and positive changes in patients’ quality of life further underscore the clinical value of this comprehensive approach.Thus, the results of this study strengthen the evidence base for using baseline LA diameter as a prognostic marker when planning surgical treatment in patients with mitral valve disease complicated by AF. Individualization of surgical intervention volume based on LA size and associated factors allows optimization of outcomes, increases the likelihood of maintaining sinus rhythm, and improves structural and functional cardiac recovery.
5. Conclusions
- Baseline left atrial (LA) size is a key prognostic factor for recurrence of atrial fibrillation (AF) after surgical correction of the mitral valve. An LA diameter >55 mm is associated with increased risk of AF recurrence and a reduced likelihood of long-term sinus rhythm restoration.A combined surgical strategy, including mitral valve repair or replacement with radiofrequency ablation using the Cox-Maze IV technique, is highly effective: 91.2% of patients remain in sinus rhythm during long-term follow-up.Reverse cardiac remodeling after surgery is more favorable in patients with baseline LA ≤55 mm, demonstrated by increased LVEF, and reduced LA and LVEDD.Baseline LA size can be used for preoperative risk stratification, planning the extent of surgical intervention, and individualizing surgical tactics, thereby improving sinus rhythm maintenance and optimizing postoperative recovery.The clinical and practical significance of these results lies in the ability to use baseline LA diameter as a marker to predict surgical outcomes, select a comprehensive surgical approach, and anticipate the potential need for intensive intraoperative hemodynamic support.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML