-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 288-295
doi:10.5923/j.ajmms.20261601.63
Received: Dec. 29, 2025; Accepted: Jan. 21, 2026; Published: Jan. 26, 2026

Clinical and Hormonal Characteristics of Children with Precocious Puberty Living in the Republic of Uzbekistan
Khalimova Zamira Yusufovna1, Uralova Dilafruz Ulugbek kizi2, Kholova Dilorom Sharipovna3, Abdullayeva Aziza Uktam kizi2
1DSc, Professor, Deputy Director for Scientific Affairs of RSSPMCE named after Academician Y.Kh. Turakulov, Tashkent, Uzbekistan
2PhD Student of RSSPMCE named after Academician Y.Kh. Turakulov, Tashkent, Uzbekistan
3DSc, Professor, Head of the Scientific Department of Neuroendocrinology with Pituitary Surgery RSSPMCE named after Academician Y.Kh. Turakulov, Tashkent, Uzbekistan
Correspondence to: Khalimova Zamira Yusufovna, DSc, Professor, Deputy Director for Scientific Affairs of RSSPMCE named after Academician Y.Kh. Turakulov, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: This study aimed to investigate the etiological structure, incidence of different forms, and clinical and hormonal characteristics of children with precocious puberty (PP) in the Republic of Uzbekistan from 2016 to 2024, with analysis by sex. Materials and methods: A retrospective analysis was conducted in 508 children diagnosed with PP using medical records from the Republican Specialized Scientific and Practical Medical Center of Endocrinology named after academician Y.Kh. Turakulov. Clinical, hormonal, and instrumental data were evaluated. Results: Among 508 children with PP, 471 (92.7%) were girls and 37 (7.3%) were boys. The mean age was 4.23 ± 2.57 years in girls and 6.1 ± 2.6 years in boys. In girls, the most common etiological factors were isolated premature thelarche (75.2%) and central precocious puberty (17.4%), whereas in boys congenital adrenal hyperplasia predominated (59.5%). More than half of patients with central (50.6%) and peripheral (53.7%) PP showed bone age advancement of ≥2 years. Elevated basal luteinizing hormone levels (>0.3 IU/L) were detected in 92.4% of children with central PP. Conclusion: In girls with early pubertal signs, isolated thelarche and central PP should be primarily considered, while congenital adrenal hyperplasia and organic central PP should be excluded first in boys. Despite the diagnostic value of basal LH, the GnRH stimulation test remains the gold standard for confirming central PP.
Keywords: Precocious puberty, Central precocious puberty, Peripheral precocious puberty, Gonadotropin-dependent, Gonadotropin-independent, Congenital adrenal hyperplasia, Mc-Cune Albright syndrome, Tanner staging, Hamartoma, Hydrocephalus, Premature telarche, Premature adrenarche
Cite this paper: Khalimova Zamira Yusufovna, Uralova Dilafruz Ulugbek kizi, Kholova Dilorom Sharipovna, Abdullayeva Aziza Uktam kizi, Clinical and Hormonal Characteristics of Children with Precocious Puberty Living in the Republic of Uzbekistan, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 288-295. doi: 10.5923/j.ajmms.20261601.63.
Article Outline
1. Introduction
- Precocious puberty (PP) is the appearance of secondary sexual characteristics under the age of 8 years in girls and up to 9 years in boys. The overall incidence of sexual precocity is estimated to be 1:5000 to 1:10000 children. In girls, the incidence exceeds boys in a ratio of 10: 1 [1]. However, several countries of the world have described the growth of incidence and prevalence, including the USA, Spain, France, Denmark, Korea and China since the 1990s [2]. Historical data from Europe show a sharp decrease in the middle age of menarche from about 17 years in the early nineteenth century to about 13 years by the middle of the twentieth century, with a slight decrease over the past 25-30 years. Understanding changes in sexual development is important, because more earlier puberty can be associated with psychosocial difficulties, is a risk factor for younger age during the first sexual intercourse and carries negative consequences for health in the long term, including an increased risk of type 2 diabetes mellitus [3], obesity, cardiovascular disease, depression and premature death. Large -scale genetic data indicates that early puberty is associated with an increased risk of breast cancer in women, while, in men, sexual development can increase the risk of prostate cancer [2]. Precocious puberty classified to gonadotropin-dependent (central, true), gonadotropin-independent (peripheral) and partial forms [4].
2. Materials and Methods
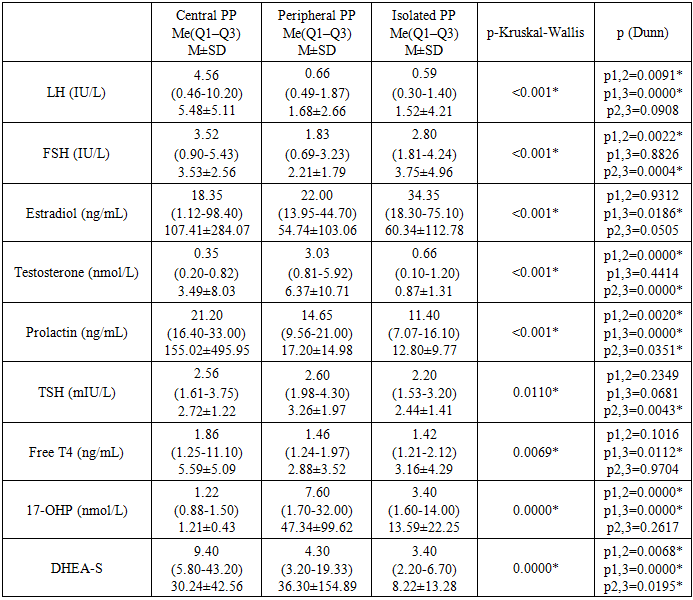
- We conducted a retrospective chart review of 508 patients with precocious puberty from 2016 to 2024 seen in the Republican Specialized Scientific and Practical Medical Center of Endocrinology named after academician Y.Kh. Turakulov. Precocious puberty was defined as the onset of secondary sexual characteristics in boys aged < 9 years or girls < 8 years or menstruation in girls < 10 years old. Tanner staging was used to record the development of breast and pubic hair in girls and testicular volume and pubic hair in boys. We collected clinical data (height, weight, age at PP diagnosis, personal and family history of major diseases, family history for PP) and hormonal data. In addition, a pelvic ultrasonography, a bone age (BA – assessed according to Greulich and Pyle) assessment using radiographs of the left hand and wrist, brain MRI imaging (if necessary), CT of adrenal glands (if necessary) were performed. In boys CPP was defined as a testicular volume ≥ 4mL, and/or a baseline luteinizing hormone (LH) > 0.3 IU/L and/or a bone age greater than 2 years from the chronological age as established by hand X-ray. In girls CPP was diagnosed as Tanner breast stage ≥ B2 and a baseline luteinizing hormone (LH) > 0.3IU/L and/or a bone age greater than 2 years from the chronological age as established by hand X-ray and/or menstruation < 10 years old. In our study chemiluminescense immunoassay (CLIA) method with the “Cobas e311” analyzer was used to determine the levels of following hormones: LH-luteinizing hormone, FSH-follicle stimulating hormone, estradiol, testosterone, prolactin, TSH-thyroid stimulating hormone, f-T4-free thyroxin, DHEA-S-dehydroepiandrosterone sulfate, 17-OHP - 17-hydroxyprogesterone. Ultrasound was used to assess the morphological and functional state of the internal genital organs (small pelvis in girls, scrotal organs in boys) using “Philips”, “Mindray” and “Hain” ultrasound machines with sensors from 2 to 12 MHz The following parameters were measured: a) uterine length, transverse diameter (width) and fundal anteroposterior diameter and b) ovarian height, width and length. All children with CPP underwent cranial MRI. Any cranial pathology (e.g. hamartomas, adenomas or hydrocephalus) that could be a potential cause of CPP was recorded. The shape, size and any heterogeneity of the pituitary gland was noted.
3. Statistical Analysis
- All univariate analyses were performed in SPSS for Windows, version 24 (IBM). A two sided P value < 0.05 was defined as statistically significant. The importance of difference between the groups in terms of the mean values was analyzed using the Student t test when there were 2 independent groups. P values <0.05 were considered statistically significant.
4. Results
- Among 508 children with PP 471 of the children (92,72%) were girls, 37 (7,28%) were boys. Mean age for girls and boys under the study was 4,23±2,57 and 6,1±2,6 respectively. In Figure 1 the distribution of patients by gender is given. According to clinical, hormonal and instrumental findings patients in terms of precocious puberty were classified in three categories: central precocious puberty (CPP), peripheral precocious puberty (PPP) and isolated forms of precocious puberty (premature telarche, premature adrenarche).Of the patients with central PP, 68 (86%) were girls, 11 (14%) were boys. Among patients with peripheral PP, 15 (36.6%) were girls, 26 (63.4%) were boys. When analyzing the isolated form, all 388 (100%) patients were girls. From this we concluded that the incidence of central and isolated PP is significantly higher in girls compared to boys. On the contrary, in the peripheral form of PP, a predominance of boys was revealed.
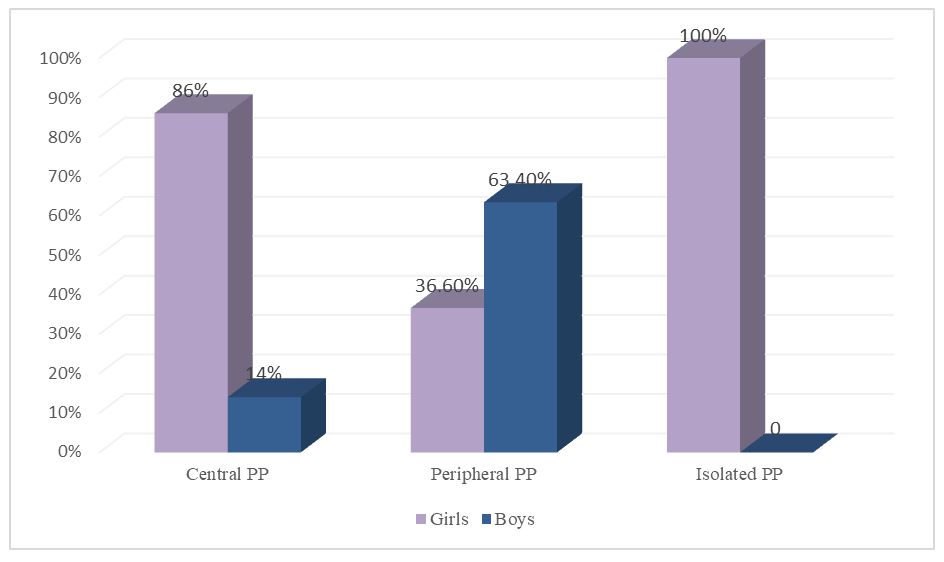 | Figure 1. Distribution of patients with PP by gender |
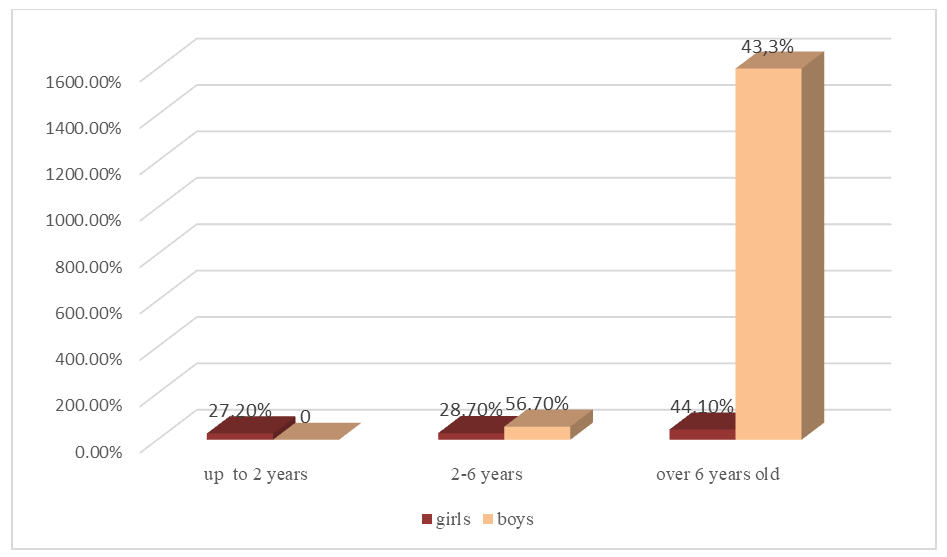 | Figure 2. The distribution of children with PP by age |
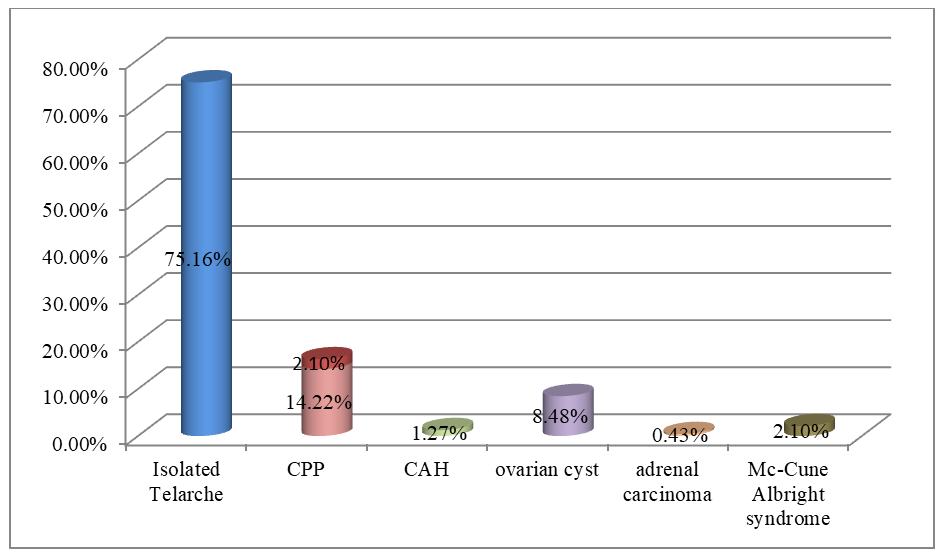 | Figure 3. Frequency of etiology of precocious puberty in Uzbek girls |
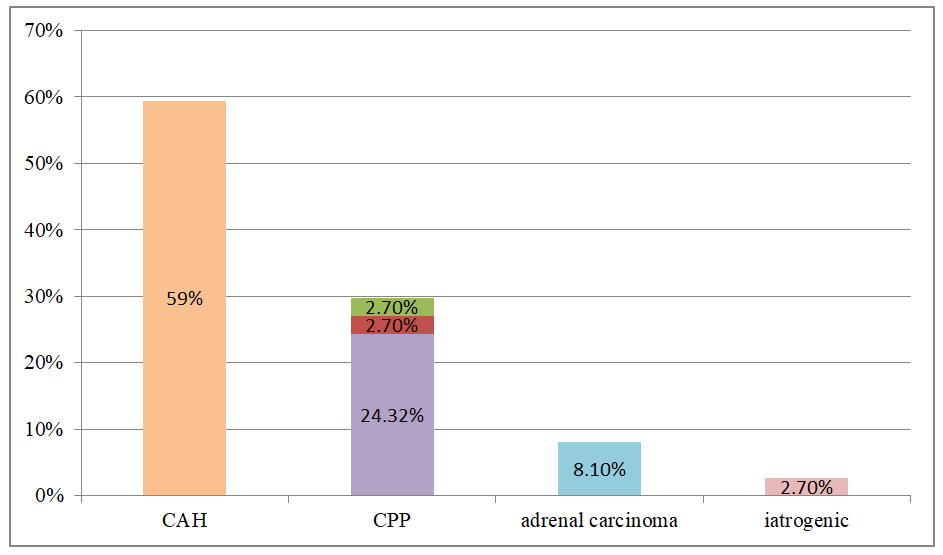 | Figure 4. Frequency of etiology of precocious puberty in Uzbek boys |
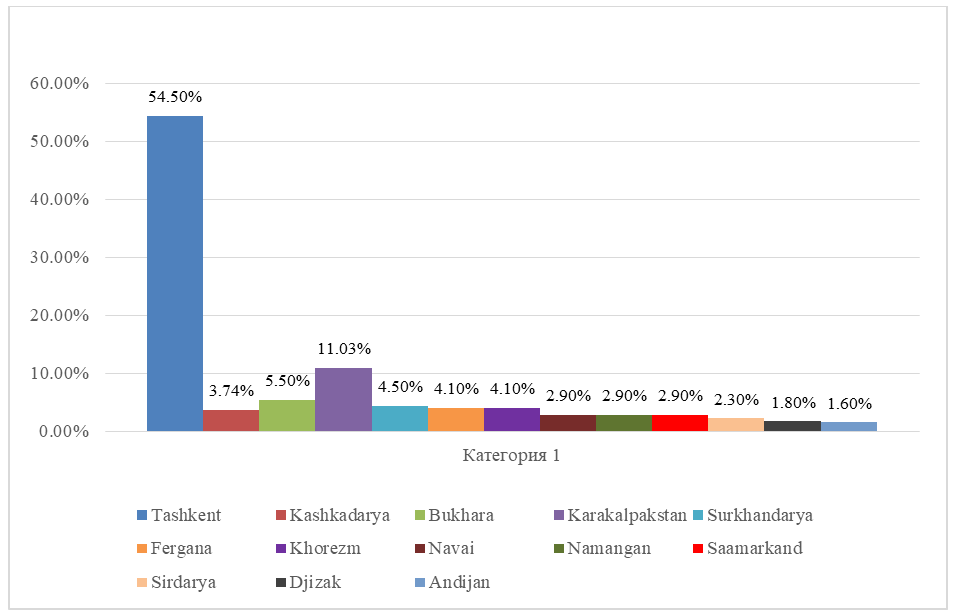 | Figure 5. Distribution of patients with PP in Uzbekistan and Karakalpakstan |
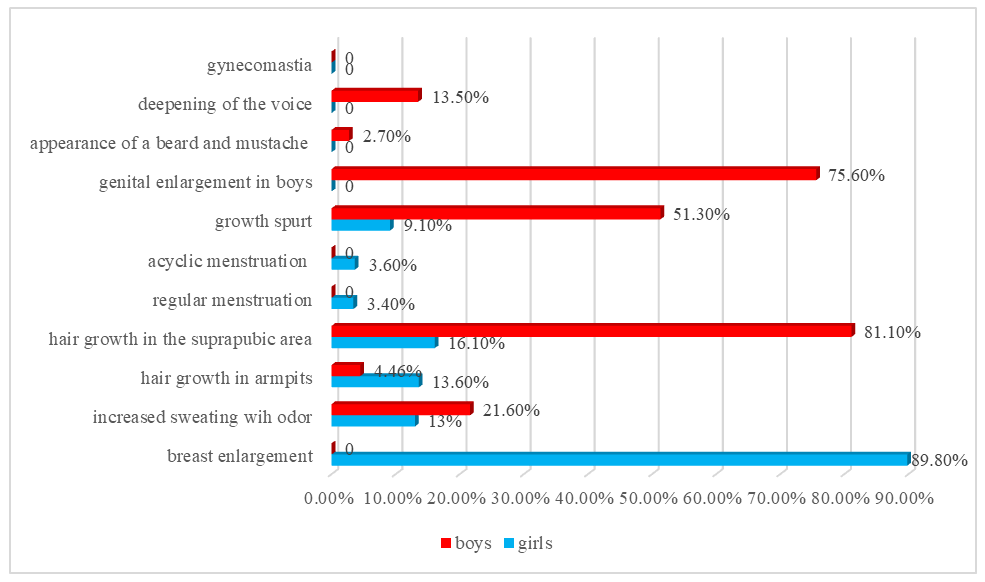 | Figure 6. Analysis of complaints of patients with PP |
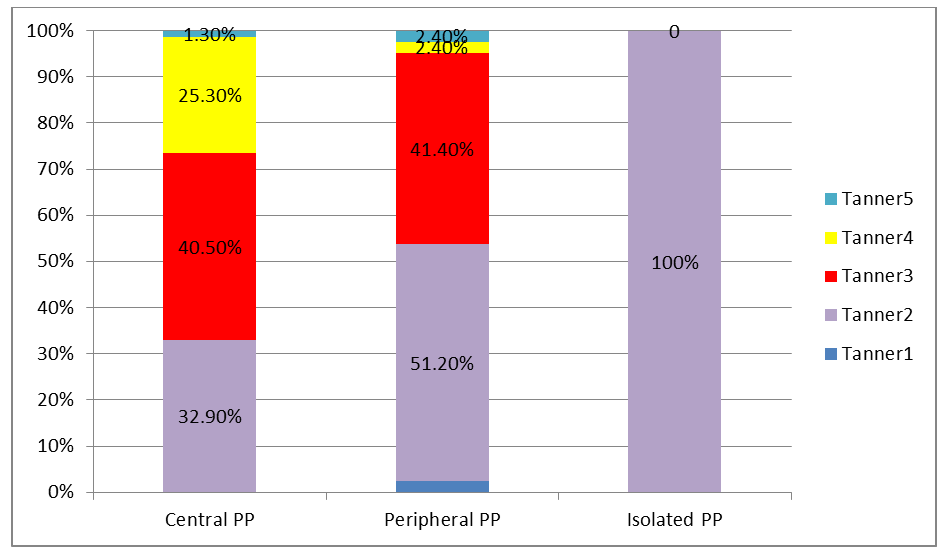 | Figure 7. Tanner staging of various PPs |
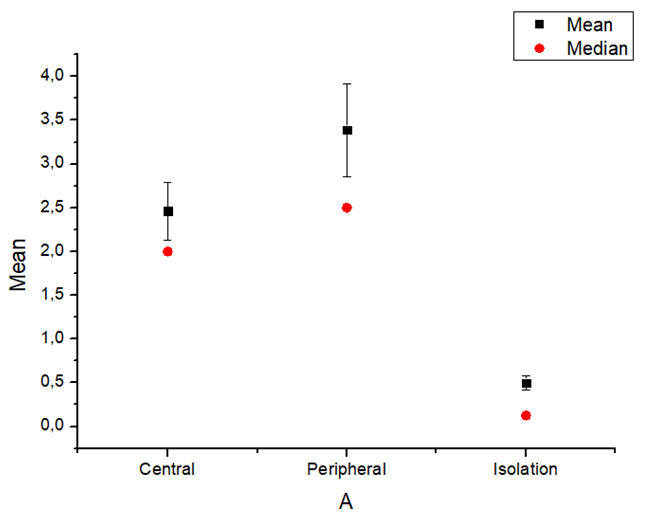 | Figure 8. Mean and median value of bone age and chronological age difference by groups |
|
5. Discussion
- According to number of authors, precocious puberty is more common in girls than in boys, female to male ratio 5-10:1 [1,5,6]. In our study, among analyzed children with precocious puberty 92,72% were girls, 7,28% were boys, and the ratio of girls to boys was 12,7:1. In Farzaneh R et al. study the most common etiology of precocious puberty in girls was idiopathic CPP (47.7%) and premature thelarche (34.1%) [7]. The most common etiology of precocious puberty in boys were neurogenic CPP (25%) and CAH (37.5%). Our research work had shown that in Uzbek and Karakalpak population the main causes of precocious puberty in girls were premature telarche (75,16%) and central precocious puberty (14,44%), whereas in boys the most common etiological factors of PP were congenital adrenal hyperplasia (59,46%) and central precocious puberty (29,72%). In Moayeri et al study, 40% of female patients had CPP, 90% of which were idiopathic CPP. Furthermore, 38.8% of boys with CPP had neurogenic cause while 61% of them had idiopathic CPP [6]. Analysis of the central form of precocious puberty in Uzbek girls showed that 98,53% had idiopathic, 1,47% had organic cause. In contrast, among boys with central precocious puberty 81,83% had idiopathic and 18,17% had organic cause.In the central and peripheral form, the frequency of advancing bone age was high, which means a big auxiliary diagnostic value of this index.The analysis of the distribution frequency on the territory of Uzbekistan showed that 54,5% of patients lived in Tashkent. This can be explained by the population density in the capital city, the high number of appeals of the patients and the process of urbanization, that data from number of researchers confirm this findings [8]. Accordingly, ultrasound findings among girls with central PP in approximately 76,4% of girls average uterine length and width were above normal reference range by age, when it comes to ovarian length and width, it was above normal in 47,8% girls with CPP. Hence, although we cannot use the size of the uterus and ovaries as a diagnostic criterion of the central precocious puberty in girls, it was confirmed that it has a great diagnostic value in CPP in girls. 92,4% patients with the central form of PP had LH level above 0,3 IU/l. In children with peripheral and isolated forms, 85,36% and 73,45% in turn had LH level above 0,3 IU/l. Based on this data, we suggest that LH>0,3 IU/l high probability indicates the presence of a central form of PP. However, in girls with telarche under the age of 2, LH level may correspond to pubertal values in accordance with the age-specific “mini-puberty”. In addition, levels of LH, FSH, testosterone and estradiol were higher in CPP patients, than other two groups. According to the level of the FSH in our study, we can’t diagnose the central or peripheral form of precocious puberty. Therefore, the gold standard for diagnosing central PP is the GnRH-stimulation test.
6. Conclusions
- Among girls with PP, the majority were older than 6 years, and among boys the highest rate was at the age of 2-6 years. The analysis of the stages of sexual development according to Tanner in different forms of PP showed that majority of patients were at 2-4 stages in the central form, 2 stage in isolated form, and 2-3 stages in the peripheral form. The main causes of precocious puberty in girls were premature telarche (75,16%) and central precocious puberty (17,35%), whereas in boys, main etiologic factor of precocious puberty was congenital adrenal hyperplasia (59,46%). Therefore, in girls presenting with symptoms of early puberty, it is necessary to first look for isolated telarche and ICPP, in boys we should firstly include congenital adrenal hyperplasia and organic CPP. In the central and peripheral form, the frequency of advancing bone age was high, which means a big auxiliary diagnostic value of this index. Although we cannot use the size of the uterus and ovaries as a diagnostic criterion of the central precocious puberty in girls, it was confirmed that it has a great diagnostic value in CPP in girls. The gold standard for diagnosing central PP is the GnRH-stimulation test.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML