Eshbaev Erkin Abdukhalimovich1, Boltaboev Tulkinboy Tursunalievich2
1DSc, Professor, Department of Pathological Anatomy, Tashkent State Medical University, Tashkent, Uzbekistan
2Independent Researcher, Fergana Medical Institute of Public Health, Fergana, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Peritonitis in infants in the neonatal period, mainly primary hematogenous, lymphogenous foci, necrotic enterocolitis, is a process that occurs in the perforation of the intestines, and as a result of sepsis, septicopyemia, purulent inflammation of all layers of the peritoneum, in addition to massive atrophic, sclerotic changes in the regional lymph nodes, acute insufficiency of all morphofunctional layers, leading to T and B cell collapse, delymphatization in the lymph node and lymph node reticulocytosis. It was found that in the subcapsular and medullary layers of the lymph node, coarsely dispersed proteinaceous deposits consisting of various damaged tissue and cell components were accumulated.
Keywords:
Lymph node, Peritonitis, Lymphoid follicle, Immunodeficiency, Infant
Cite this paper: Eshbaev Erkin Abdukhalimovich, Boltaboev Tulkinboy Tursunalievich, Pathomorphology of Mesenteric Lymph Nodes in Infants Who Died from Peritonitis in the Neonatal Period, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 283-287. doi: 10.5923/j.ajmms.20261601.62.
1. Introduction
Perinatal and neonatal mortality associated with generalized inflammatory processes, including peritonitis, continues to occupy a significant place in the structure of causes of death in newborns. Neonatal peritonitis is characterized by a fulminant course, a high incidence of septic complications, and an unfavorable prognosis, especially in premature infants and those with morphofunctional immaturity [1,2,11]. In the world today, the lethality of purulent peritonitis is very high, reaching 20-60%. When this disease occurs, it is accompanied by the development of rapidly developing endogenous intoxication and multi-organ failure. In the USA and Europe, the incidence rate is 10-15 per 100 thousand, while in Turkey and Korea it is 12-17 per 100 thousand [3,4,6]. In the Russian Federation and the CIS countries, it is 15-20 per 100 thousand, while in Central Asian countries, Azerbaijan, and Armenia this figure is 50-71 per 100, with a lethality rate of 60-85% [3,4,5].In our country, on average, 700-1300 infants under the age of 1 die from various types of peritonitis per year. In peritonitis, the main organs actively involved in the drainage of the abdominal cavity are only the regional lymph nodes, and at 1-3 months of age, this drainage is dominated by the occurrence of physiological paralysis and slow blood circulation, while at 3-6 months, the weakening of intestinal peristalsis and drainage in the regional lymph nodes, insufficiency of the barrier function, lead to the stagnation of local infection in the intestinal lymph nodes and the development of lymphostasis [7,8]. This requires the development of specific recommendations, taking into account the specific morphofunctional aspects of each period [9,10,12]. The insufficient development of morphological criteria for mesenteric lymph node involvement complicates an objective forensic and clinicopathological assessment of the causes of death, the degree of generalization of the inflammatory process, and the severity of the immune response in infants who died during the neonatal period. In addition, the lack of systematized pathomorphological data limits the possibilities for correlating clinical, laboratory, and autopsy findings. Objective Study and improvement of morphological, morphometric, immunohistochemical aspects of regional lymph nodes in peritonitis developed in infants under one year of age.
2. Materials and Methods
A total of 71 patients, including 12 control group and deceased children, who were brought to the Republican Center for Pathological Anatomy in 2018-2023 were used as material. The samples were stained with hematoxylin and eosin and studied.
3. Results of the Study and Discussion
Microscopic examination of regional lymph nodes in infants who died of peritonitis at 28 days of age revealed that the immature medullary and medullary layers were not fully formed, and changes that occurred as a result of severe large-scale purulent and non-purulent inflammation, such as necrotic enterocolitis, before the development of peritonitis led to changes in the lymphatic drainage system in the form of blockage or paralysis.This, in turn, was manifested by a sharp tightening of the capsule in the mesenteric lymph nodes of 28-day-old infants, a sharp expansion of the subcapsular spaces, which were significantly enlarged. In particular, in the development of peritonitis, a sharp decrease in the function of lymphatic drainage mainly leads to the accumulation of exudate fluid in the abdominal cavity. Hyperproduction of mesothelial cells and the formation of multifocal desquamation foci, many erosive foci were also detected in the visceral sheets.In the mesenteric lymphatic vessels, a sharp slowdown in the drainage function of lymphostasis is observed, which is explained by the expansion of the vessels and the increase in size of the fluid accumulated due to plasmarrhagia in the regional lymph nodes. As a result, the capsules of the lymph nodes are stretched, the subcapsular spaces expand, and lymphostasis develops. Eosinophilic inclusions of varying degrees of light and dark staining in the cortex and medulla of the lymph node: cellular components, various tissue parts with destructive changes were detected. The lymph nodes have a swollen appearance, the capsule is tense, and the subcapsular spaces are filled with lymphatic fluid (see Figure 1). Secondary lymphoid follicles are not detected, the germinal center is quiet, the location of lymphocytes in the cortical and paracortical areas of the active areas of the lymph nodes is reduced compared to normal, and in the active areas of the cortex, foci of lymphocytes migrating towards the postcapillary venules are detected. In the direction of the cerebral bands of the medulla, it is determined that a swelling appearance in the areas of the lymphatic gate and a variable mixture of lymphocytes and other types of cells (macrophages, dendritic cells, interdigitating cells, fibroblasts, histiocytes, reticulocytes, etc.) are relatively preserved in the areas of the reticulum and trabeculae of the brain bands.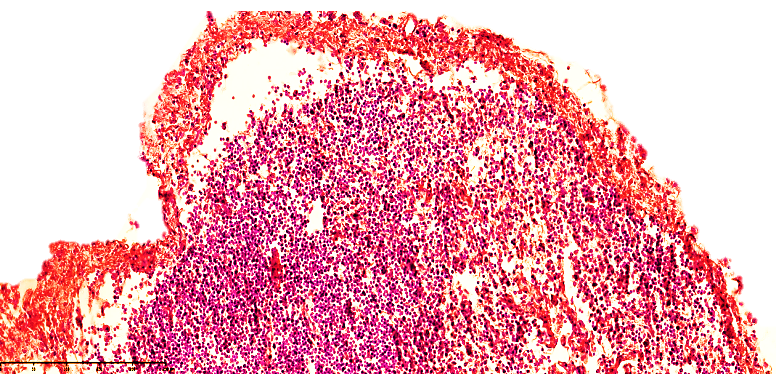 | Figure 1. Mesenteric lymph node of a 28-day-old infant with peritonitis. Treated for 10 days. The lymph node capsule is of variable width, the subcapsular space is dilated, and lymphoid follicles are not identified. Small vessels are fully visible. Stained with GE. Size 10x10 |
It was found that the main part of the regional lymph nodes is located in the ileocecal mandible and that massive enlargement of the lymph nodes occurs precisely in peritonitis. The lymph node capsule is tense, of uniform thickness, and reticulocytosis and macrophages are retained in the subcapsular space. As a result of the sharp increase in the level of fluid drainage, massively denuded stroma is detected in the lymphoid follicles in the active areas of the lymph node (cortical and paracortical areas) (see Figure 2). This indicates a connection with the massive recruitment of lymphocytes to the foci of inflammation. As a result, the lymph node acquires a swollen appearance, and loose lymph nodes are detected. The appearance of lymphoid follicles is sharply blurred against the general background, the follicle texture is lost, and neutrophils are detected around the postcapillary venules in the paracortical area. (see Figure 3). The abundance of mast cells confirms the acute course of the inflammatory process.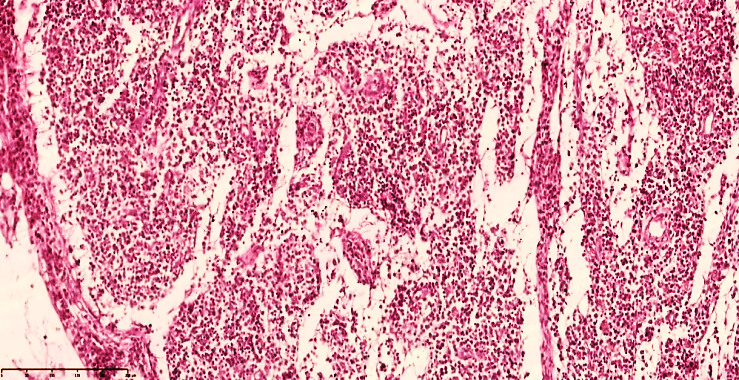 | Figure 2. Mesenteric lymph node of a 28-day-old infant with peritonitis. The capsule is thickened, the subcapsular space is markedly enlarged, the peritrabecular areas are bare, formed in the form of a cavity, and the lymphoid follicles do not have a clear border. Staining G.E. Size 10x10 |
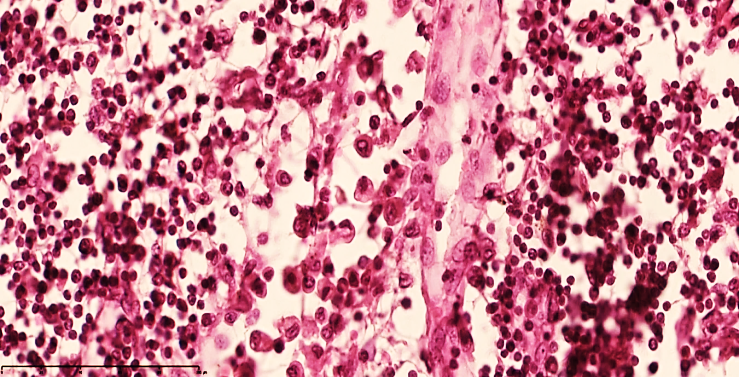 | Figure 3. Mesenteric lymph node of a 28-day-old infant with peritonitis. Macrophages are increased in the peritrabecular area, lymphocytes are decreased, and oligonucleated lymphoblasts are increased, and stroma is disrupted. Staining G.E. Size 40x10 |
Lymphostasis and dilatation of lymphatic capillaries are preserved in the lymph nodes. In the subcapsular area of the lymph node, a small number of macrophages and lymphocytes are detected in the spaces where the cyst has expanded. Areas of regenerating V-lymphocytes in the cortical area are defined, although the lymphoid follicular boundaries and texture are uneven (see Fig. 4).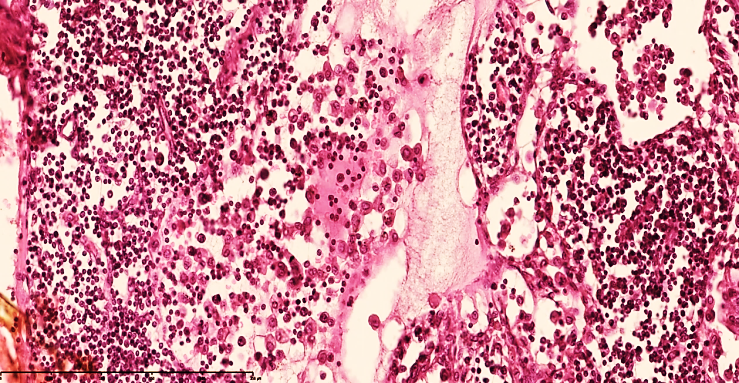 | Figure 4. Mesenteric lymph node of a 28-day-old infant with peritonitis. Treated on the 6th day. Macrophages increased in the peritrabecular area, lymphocytes and plasma cells decreased, and oligonucleated lymphoblasts increased, and stroma was found to be disrupted. Vascular dilation and collapse were detected. Staining G.E. Size 40x10 |
In the paracortical areas, a small number of T-lymphocytes are found, and in the space around the vessels, destroyed and various cell fragments are found. This fluid moves directly to the regional lymph nodes. The lymph node capsule is tense, of uniform thickness, macrophages are relatively reduced in the subcapsular space, lymphocytes are detected.Postcapillary venules were full-blooded, with foci of free lymphocyte migration in the focus. The perimeter of the postcapillary venules was covered with fibrinoid thickening.It was found that the histiotopographic shape of the lymph node was deformed in the areas of thickening and destruction of the trabecular stroma components, and the morphofunctional parameters of the lymph node were out of whack.Lymphostasis in the lymph nodes, lymphothrombosis, lymphadenitis in the lymph nodes, delymphatization and the development of foci of neutrophil infiltration in the postcapillary venules, complete blockage of the lymphatic drainage system in the lymph node, leading to the development of secondary complications (sepsis, septicemia, septicopyemia, etc.).Degranulation of mast cells in the microcirculatory bed, as a result of which plasmorrhagia and leukodiapedesis are enhanced, leading to the formation of inflammatory infiltrates in the parietal and visceral layers. (See Fig. 5, 6). As a result, venous congestion occurs, which leads to the development of interstitial edema and a sharp functional strain of the drainage system in the lymph nodes, which leads to lymph node enlargement and lymph node edema (See Fig. 7).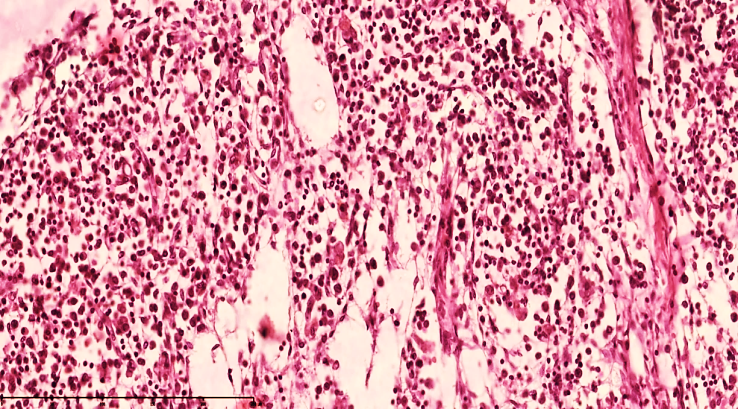 | Figure 5. Mesenteric lymph node of a 28-day-old infant with peritonitis. In the cortical area, lymphocytes and plasma cells are reduced, lymphoblasts are increased with nucleoli, and stroma is disrupted. Delymphatic foci are detected. Staining G.E. Size 40x10 |
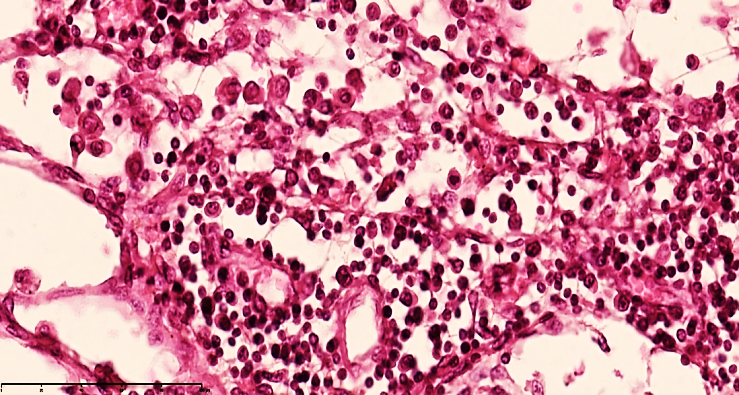 | Figure 6. Mesenteric lymph node of a 28-day-old infant with peritonitis. Treated on the 3rd day. In the cortical area, lymphocytes and plasma cells are reduced, mononuclear lymphoblasts are increased, and stroma is disintegrated. In the form of a cone, foci of reticulocytosis are detected. Staining GE. Size 40x10 |
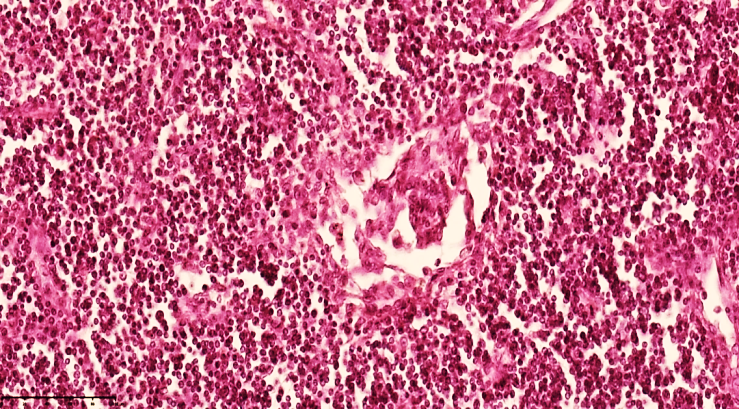 | Figure 7. Mesenteric lymph node of a 28-day-old infant with peritonitis. Treated on the 13th day. Lymphocytes and plasma cells decreased in the cortical area, mononuclear lymphoblasts increased, and stroma wasting was detected. Delymphatic foci were detected. Staining G.E. Size 40x10 |
Sharp expansion of lymph nodes and parallel enlargement of regional lymph nodes, sometimes lymphangitis was found. These changes were found in the lymph nodes, the tightening of the capsule, the expansion of the subcapsular space, the increase of bare areas in the stroma, the perivascular edema around the lymphatic blood vessels, and the vertical arrangement of reticulocytes.Increased migration of T and B lymphocytes in the active areas of the lymph nodes through the postcapillary venules is determined. Stasis and subcapsular edema in the lymph nodes, pleural effusion in the lymph nodes and migration of T and B lymphocytes around the postcapillary venules are determined. In the lymph node cavity, the destruction of cellular and tissue components, consisting of fibrous structures, and the development of lymphostasis foci are detected. In the foci of inflammation, segmented nuclear neutrophils predominate. Stasis is preserved in the lymph nodes, the capsule is of varying thickness, tumors are preserved in the subcapsular space, lymphoid follicles are reduced in size and their cellular composition is reduced (see Fig. 8-9). This indicates that lymphocytes are intensively mobilized to the focus of inflammation, but their return is impaired.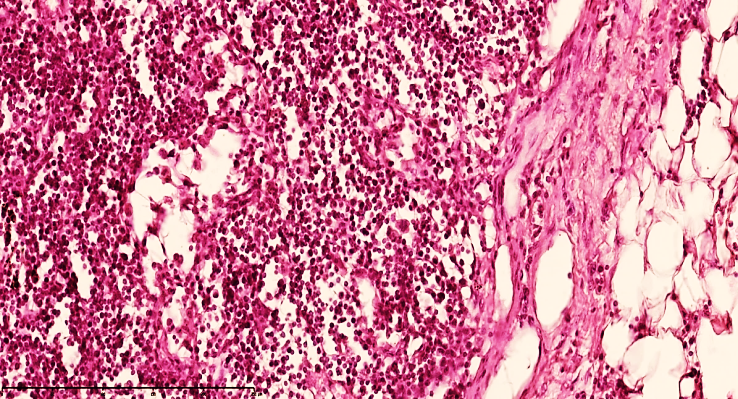 | Figure 8. Mesenteric lymph node of a 28-day-old infant with peritonitis. The capsule is markedly thickened, traces of lymphoid follicles are visible, and cystic spaces are formed in the space. Stained with GE. Size 40x10 |
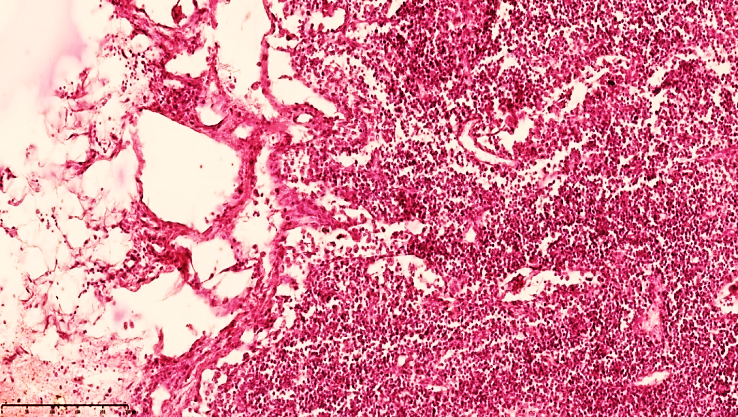 | Figure 9. Mesenteric lymph node of a 28-day-old infant with peritonitis. Treated on the 15th day. Fragmented capsule with cystic expansion in the space between. The boundaries of the cortex and medulla are determined. The main hypercellularity is formed by lymphoblasts. Staining G.E. Size 40x10 |
In the peritoneal sheets, a relative decrease in neutrophils and a subsequent increase in the number of macrophages were detected as a result of the reduction of the infectious (bacterial) factor under the influence of antibiotics and antiseptics. The foci of phagocytosis of destroyed tissue components by macrophages are detected. In the peritoneal tissue, interstitial edema and the full-blooded appearance of blood vessels with the preservation of perivascular edema are detected. Stasis and lymphodynamic disorders are detected in the lymphatic system, the process of lymphocyte recruitment and the ongoing process of delymphaticization are detected in active areas (see Fig. 9).Stasis and fullness of the lymphatic vessels, their cavities filled with fragments consisting of disintegrated tissue and cells, and foci of lymphothrombosis are detected. In the active areas of the lymph node, a decrease in T and B lymphocytes and an increase in their place by a large number of macrophages and reticulocytes were detected.In the exposed areas of the parietal and visceral peritoneal surfaces, fibrinous inclusions and fibrinous exudate formed adhesions with healthy serous tissue. In the interstitial space of the newly formed sparse fibrous connective tissue, an inflammatory infiltrate was still present (see Fig. 10-11).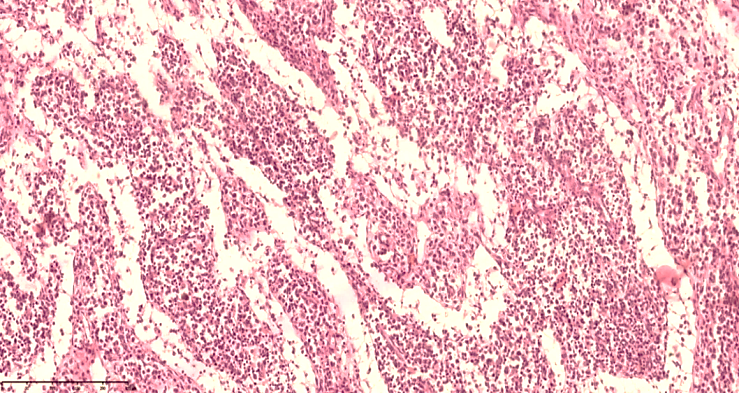 | Figure 10. Mesenteric lymph node of a 28-day-old infant with peritonitis. In the corticomedullary area, there are scattered foci along the trabecular border, with the appearance of interstitial space.Stained with GE. Size 10x10 |
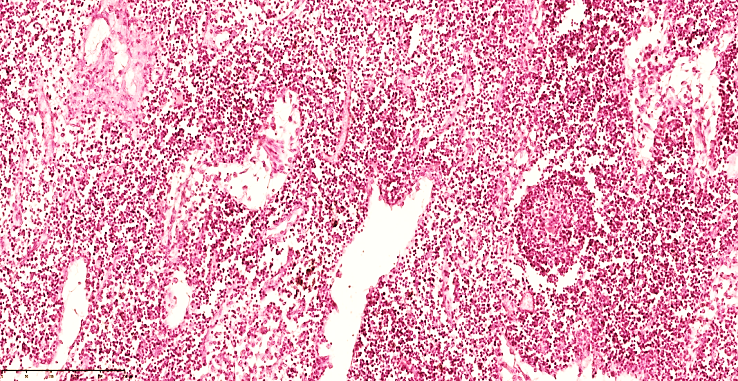 | Figure 11. 28-day-old infant. Mesenteric lymph node of a 28-day-old infant with peritonitis. Single atrophic lymphoid follicles are detected in the cortex. A small number of macrophages and reticulocytes are detected in the germinal center. Most of the blood vessels are dilated, and foci of reticulocytosis are detected in the stroma. Staining G.E. Size 10x10 |
In this area, fibrinoid necrosis and fibrinoid thickening are detected on the exposed basal layer of the sheet surface. In this area, small-caliber blood vessels and foci resembling hyaline thrombi are detected in the microcirculatory tract. In parallel, perivascular edema and hyperemia in the lymphatic vessels are detected.Inflammatory foci are macrophages and segmented neutrophils, lymphocytes and mast cells. This is manifested by a sharp deficiency of angioprotectors and membrane-stabilizing factors, which, in the traditional treatment, are aimed at preventing blood stasis and lymphostasis in the vessels, determining the dynamics of inflammation. As a result, the absorption of intermediate metabolites and toxic substances throughout the peritoneum is characterized by endogenous intoxication. The visceral layer covers the surface of the intestines and is rich in blood vessels, and peritonitis is characterized by a sharp expansion of blood vessels in the serous membrane, the formation of fibrinous purulent exudate on its surface.Stasis in the lymph nodes, foci of microthrombosis, subcapsular edema of the lymph nodes, an increase in the number of reticulocytes, their vertical location, a strong manifestation of the filter-barrier phenomenon indicate the retention of macrophages in the barriers, which indicates a morphofunctional blockage of the lymphatic drainage system. As a result, the lymphocytes in the active areas of the lymph nodes lag behind the acute response to the process and a relative decrease in B lymphocytes in the cortical areas and T lymphocytes in the paracortical areas is detected.The preservation of integrity in the lymph nodes is due to the absence of a factor necessary for the drainage of the exudate formed under the influence of peritonitis, which is manifested by massively active macrophages, along with intermediately destroyed small dispersed structures, clogging the lymph nodes and subcapsular spaces in the form of microthrombi, which manifests itself as subcapsular edema, fullness in the postcapillary venules, cystic and ponassimon-like enlarged spaces in the areas of the lymphatic gate and medullary bands.
4. Conclusions
In infants who died of peritonitis at the age of 28 days, a sharp decrease in lymphocytes, emptying of the sinuses, unclear boundaries between the cortex and medulla, lack of formation of lymphoid follicles, and accumulation of homogeneous protein substrates in the stroma of the lymph node, mainly consisting of macrophages, lymphoblasts, reticular cells, massive necrotic masses, are detected in the mesenteric lymph nodes. Blood vessels are unevenly filled, reticulocytosis, delymphatization and lymph node histioarchitectonic disorders were found.It is determined that symptoms of lymphangitis in lymph nodes and lymphadenitis in lymph nodes are preserved. The fact that the number of immunocompetent cells in the active areas of the lymph node has not yet fully occupied the lost volume and the presence of infiltrating foci of neutrophils and macrophages around the postcapillary venules requires the addition of other methods to the treatment tactics, requiring the protection of the parallel connection in the vein-lymph drainage system in the damaged areas.
References
| [1] | Gorbatyuk, O. M., Martynyuk, T. V., & Shatrova, K. M. Clinical and morphological characteristics of gastrointestinal perforations in newborns. Russian Bulletin of Pediatric Surgery, Anesthesiology and Reanimatology, 2013, Vol. 3, No. 2, pp. 31–36. |
| [2] | Gorbatyuk O. M., Shatrova K. M., Martynyuk T. V. Morphological diagnosis of gastrointestinal perforation and newborn baby // Neonatology, surgery and perinatal medicine. – 2015. – no. 5, No. 3. - S. 62-66.Aires J, Ilhan ZE, Nicolas L, Ferraris L, Delannoy J, Bredel M, Chauvire-Drouard A, Barbut F, Rozé JC, Lepage P, Butel MJ, ClosNEC Study Group. Occurrence of Neonatal Necrotizing Enterocolitis in Premature Neonates and Gut Microbiota: A Case-Control Prospective Multicenter Study. // Microorganisms. 2023 Sep 29; 11(10): 2457. |
| [3] | Alsaied A, Islam N, Thalib L. Global incidence of necrotizing enterocolitis: a systematic review and meta-analysis. // BMC Pediatr 2020; 20: 344. |
| [4] | Andrade JAB, Freymüller E, Fagundes-Neto U. Pathophysiology of enteroaggregative Escherichia coli infection: an experimental model utilizing transmission electron microscopy. // Arq Gastroenterol. 2010; 47: 306–12. |
| [5] | Aujla S.J., Chan Y.R., Zheng M., Fei M., Askew D.J., Pociask D.A., Reinhart T.A., McAllister F., Edeal J., Gaus K. IL-22 mediates mucosal host defense against Gram-negative bacterial pneumonia. Nat. Med. 2008; 14: 275–281. |
| [6] | Aydemir G, Cekmez F, Tanju IA, Canpolat FE, Genc FA, Yildirim S, Tunc T, Sarici SU. Increased fecal calprotectin in preterm infants with necrotizing enterocolitis. // Clin Lab. 2012; 58(7-8): 841-4. |
| [7] | Berry MJ, Port LJ, Gately C, Stringer MD. Outcomes of infants born at 23 and 24 weeks' gestation with gut perforation. // J Pediatr Surg. 2019 Oct; 54(10): 2092-2098. |
| [8] | Bethell GS, Knight M, Hall NJ. BAPS-CASS B-CNIGobo. Surgical necrotizing enterocolitis: association between surgical indication, timing, and outcomes. // J Pediatr Surge. 2021; 56(10): 1785–90. |
| [9] | Black RE, Cousens S, Johnson HL, et al. Global, regional, and national causes of child mortality in 2008: a systematic analysis. // Lancet. 2010; 375: 1969–87. |
| [10] | Brusselaers N, Simin J, E Lilja H. Risk of neurodevelopmental impairment in Swedish preterm children treated for necrotizing enterocolitis: retrospective cohort study. // BJS Open. 2024 Oct 29; 8(6): zrae131. |
| [11] | Brusselaers N, Simin J, E Lilja H. Risk of neurodevelopmental impairment in Swedish preterm children treated for necrotizing enterocolitis: retrospective cohort study. // BJS Open. 2024 Oct 29; 8(6): zrae131. |
| [12] | Burnand KM, Zaparackaite I, Lahiri RP, Parsons G, Farrugia MK, Clarke SA, DeCaluwe D, Haddad M, Choudhry MS. The value of contrast studies in the evaluation of bowel strictures after necrotising enterocolitis. // Pediatr Surg Int. 2016 May; 32(5): 465-70. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML









