-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 234-237
doi:10.5923/j.ajmms.20261601.51
Received: Dec. 20, 2025; Accepted: Jan. 17, 2026; Published: Jan. 22, 2026

The Role of Melatonin Hormone in the Pathogenesis of Sleep-Associated Epileptic Seizures and Sleep Structure Disorders in Patients with Epilepsy
S. Kh. Kalandarova, F. Kh. Muratov, S. R. Kuranbaeva
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Objective: To study chronobiological mechanisms of the relationship between sleep disorders and epileptic seizures, and to evaluate the role of melatonin in the pathogenesis of sleep-associated epilepsy. Materials and methods. A comprehensive examination of 107 patients with epilepsy (61 patients with nocturnal seizures, 46 with diurnal seizures) and 30 healthy volunteers was conducted. Video-EEG monitoring, validated questionnaires (PSQI, ESS, QOLIE-31, NHS3), and determination of melatonin levels by ELISA were used. Results. Patients with sleep epilepsy showed pronounced sleep architecture disorders: decreased efficiency to 81.2±8.9% (p<0.001), increased sleep onset latency to 28.4±15.7 min, reduced deep sleep to 14.2±6.8% and REM sleep to 14.3±5.4%. Melatonin deficiency (<85 pg/ml) was found in 77.0% of patients with nocturnal seizures versus 10.0% in controls (p<0.001). A strong correlation was established between melatonin levels and seizure frequency (r=-0.567, p<0.001), sleep quality (r=-0.634, p<0.001). Structural modeling revealed the mediating role of sleep parameters in the melatonin-epilepsy relationship. The use of melatonin in complex therapy led to a 1.7-fold reduction in seizure severity, a 2.1-fold improvement in sleep quality, and a 1.4-fold improvement in quality of life. Conclusion. Melatonin deficiency is a key pathogenetic link in sleep epilepsy, justifying the inclusion of melatonin preparations in the complex therapy of this form of the disease.
Keywords: Epilepsy, Sleep, Melatonin, Circadian rhythms, Video-EEG monitoring, Quality of life
Cite this paper: S. Kh. Kalandarova, F. Kh. Muratov, S. R. Kuranbaeva, The Role of Melatonin Hormone in the Pathogenesis of Sleep-Associated Epileptic Seizures and Sleep Structure Disorders in Patients with Epilepsy, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 234-237. doi: 10.5923/j.ajmms.20261601.51.
1. Introduction
- The relationship between sleep and epilepsy represents one of the most complex problems in modern neurophysiology. Sleep-related epileptic seizures are observed in 25-45% of patients with epilepsy and are characterized by specific clinical and electroencephalographic manifestations [1]. Current research shows that different stages of sleep have opposing effects on epileptic activity: slow-wave sleep promotes synchronization and spread of epileptic discharges, while REM sleep has an antiepileptic effect [2,3]. Melatonin, the main hormone of the pineal gland, plays a key role in regulating the sleep-wake cycle and possesses multiple neuroprotective effects [4]. Experimental studies demonstrate anticonvulsant properties of melatonin related to modulation of GABAergic transmission, antioxidant action, and stabilization of neuronal membrane potential [5,6]. However, clinical data on the role of melatonin in epilepsy pathogenesis remain contradictory, and the mechanisms of interaction in the "sleep-melatonin-epilepsy" system are insufficiently studied [7,8]. Understanding these relationships is critically important for developing pathogenetically based approaches to diagnosis and treatment of sleep-associated epilepsy.Objective: To study chronobiological mechanisms of the relationship between sleep structure disorders and epileptic seizures, and to evaluate the role of melatonin in pathogenesis and its potential application in therapy of sleep-associated epilepsy.
2. Materials and Methods
- A prospective controlled study of 107 patients with verified epilepsy diagnosis (47 men, 60 women, mean age 36.8±12.4 years) and 30 healthy volunteers (control group) was conducted. Patients were divided into two groups: 61 patients with epileptic seizures occurring predominantly during sleep (sleep epilepsy group), and 46 patients with seizures exclusively during wakefulness (wake epilepsy group). Inclusion criteria: verified epilepsy diagnosis according to ILAE criteria (2022), age 18-60 years, stable antiepileptic therapy for at least 3 months. Exclusion criteria: psychogenic seizures, acute symptomatic seizures, severe somatic diseases, psychotropic medication use. All participants underwent video-EEG monitoring for 4-24 hours with sleep recording, sleep quality assessment using the Pittsburgh Sleep Quality Index (PSQI), daytime sleepiness using the Epworth Sleepiness Scale (ESS), seizure severity using the NHS3 scale, and quality of life using the QOLIE-31 questionnaire. Serum melatonin levels were determined by ELISA during 22:00-23:00. Statistical analysis included descriptive statistics, correlation analysis, and structural modeling. Differences were considered significant at p<0.05.
3. Results
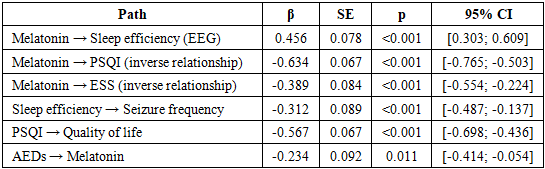
- Analysis of sleep architecture revealed pronounced disorders in patients with epilepsy, especially in those with nocturnal seizures. Sleep efficiency in the sleep epilepsy group was 81.2±8.9% versus 93.4±4.2% in controls (p<0.001). Sleep onset latency was increased to 28.4±15.7 min versus 12.6±8.4 min in healthy individuals (p<0.001). The most significant changes concerned the representation of different sleep stages: the proportion of deep slow-wave sleep (N3) was reduced to 14.2±6.8% versus 23.7±4.2% in controls (p<0.001), REM sleep to 14.3±5.4% versus 21.5±3.6% respectively (p<0.001). Simultaneously, an increase in superficial stages was observed: N1 to 12.8±4.6% versus 6.2±2.1% (p<0.001), N2 to 58.7±8.3% versus 48.6±6.4% (p<0.001). Sleep fragmentation was characterized by increased awakening index to 18.7±6.3 episodes per hour versus 7.2±2.9 in controls (p<0.001) and microarousals to 24.6±8.1 versus 9.4±3.7 respectively (p<0.001). Subjective sleep quality assessment using PSQI showed pronounced disorders in the sleep epilepsy group: total score was 12.8±3.4 versus 4.2±1.8 in controls (p<0.001). Pathological daytime sleepiness according to ESS (>10 points) was found in 96.7% of patients with nocturnal seizures versus 10.0% in controls. Investigation of melatonin levels revealed significant deficiency in patients with epilepsy. In the sleep epilepsy group, melatonin concentration was 67.3±18.9 pg/ml versus 98.7±12.4 pg/ml in controls (p<0.001), representing a 31.8% decrease. In patients with wake epilepsy, melatonin levels were intermediate - 82.4±16.2 pg/ml (p=0.001 compared to the sleep epilepsy group). Reduced melatonin levels (<85 pg/ml) were found in 77.0% of patients with nocturnal seizures, 50.0% with diurnal seizures, and only 10.0% of healthy individuals (χ²=34.7, p<0.001). Correlation analysis (Table 1) revealed multiple significant relationships between melatonin levels and clinical parameters. The strongest correlations were observed with seizure frequency (r=-0.567, p<0.001), total PSQI score (r=-0.634, p<0.001), and sleep efficiency (r=0.456, p<0.001).
|
4. Discussion
- The obtained results confirm the concept of complex bidirectional relationships between sleep and epilepsy, where epileptic activity disrupts sleep architecture, and sleep disorders, in turn, contribute to increased seizure frequency [9]. Pronounced changes in sleep architecture in patients with epilepsy, especially the reduction in deep slow-wave and REM sleep proportions, are consistent with data from modern polysomnographic studies [10,11]. These changes are critically important since deep sleep stages play a key role in memory consolidation, neuroplasticity, and cognitive function restoration [12]. The melatonin deficiency found in 77% of patients with nocturnal seizures represents an important pathophysiological link that may explain the mechanisms of sleep epilepsy development and maintenance. Melatonin, besides regulating circadian rhythms, possesses multiple neuroprotective effects: antioxidant, anti-inflammatory, and membrane-stabilizing actions [13,14]. Its level reduction may contribute to increased neuronal excitability and disruption of the balance between excitatory and inhibitory neurotransmitters, creating conditions for epileptic activity development [15]. The strong correlation between melatonin levels and seizure frequency (r=-0.567) confirms the hypothesis about antiepiletic properties of this neurohormone, which is consistent with experimental research results [16,17]. Structural modeling results reveal mechanisms through which melatonin exerts its antiepileptic action. The greatest mediating role of subjective sleep quality assessment (51.2%) indicates the importance of not only objective improvement of sleep parameters but also subjective perception of sleep quality by patients. This may be related to melatonin's influence on psychoemotional state, anxiety levels, and general well-being [18]. Clinical effectiveness of melatonin use in complex therapy of sleep epilepsy confirms the pathogenetic rationale of this approach. A 1.7-fold reduction in seizure severity, 2.1-fold improvement in sleep quality, and 1.4-fold improvement in quality of life demonstrate significant advantages of including melatonin in therapeutic regimens. These results are consistent with data from small randomized studies showing melatonin effectiveness in epilepsy [19,20,21,22]. The developed multifactorial seizure risk prediction model has important practical significance, allowing patient stratification by risk groups and personalization of preventive approaches. Including easily determinable parameters such as melatonin levels and standardized questionnaire results makes it applicable in routine clinical practice.
5. Conclusions
- The conducted study revealed fundamental disorders in the sleep-wake system in epilepsy, characterized by melatonin deficiency in 77% of patients with nocturnal seizures, pronounced changes in sleep architecture, and complex mediating relationships between components of the "melatonin-sleep-epilepsy" system. Melatonin deficiency is a key pathogenetic link in sleep epilepsy, closely related to seizure frequency and sleep quality. The use of melatonin in complex therapy demonstrates high effectiveness, justifying the inclusion of melatonin preparations in treatment standards for sleep-associated epilepsy. The developed diagnostic and treatment algorithm, based on integration of clinical, electroencephalographic, and biochemical parameters, opens new possibilities for personalized medicine in epileptology.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML