-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 220-228
doi:10.5923/j.ajmms.20261601.49
Received: Dec. 19, 2025; Accepted: Jan. 22, 2026; Published: Jan. 22, 2026

Polypill-Based Therapy for Reduction of SCORE2 and Cardiovascular Risk in Patients with Arterial Hypertension and Dyslipidemia
Shakhlo S. Fayzullaeva1, Gulnoz A. Khamidullaeva1, Guzal Zh. Abdullaeva1, Zarina T. Mashkurova1, Khafiza F. Yusupova1, Nargiza R. Khanberdiyeva2
1Republican Specialized Scientific and Practical Medical Center of Cardiology, Tashkent, Uzbekistan
2Tashkent Medical Academy, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The objective of this study was to compare the effectiveness of a 6-month fixed-dose combination polypill versus free combinations of antihypertensive drugs and statins in achieving target blood pressure, lipid profile parameters, SCORE2 risk reduction, and organ-protective effects in patients with AH and high or very high cardiovascular risk. Methods and Results: The study included 119 patients aged 40–75 years with grade 1–2 AH and dyslipidemia were randomized into two groups. Group 1 (n=60) received a polypill containing lisinopril/amlodipine/rosuvastatin, while Group 2 (n=59) received separate antihypertensive drugs and rosuvastatin. Clinical assessment included office and ambulatory blood pressure monitoring, lipid profile analysis, SCORE2 evaluation, arterial stiffness parameters, echocardiography, renal function, and metabolic indices at baseline, 3 and 6 months. After 6 months of therapy, both treatment strategies significantly improved blood pressure, lipid parameters, and SCORE2 values (P<0.001). However, the polypill group demonstrated significantly higher rates of achieving target blood pressure (81.3% vs. 57.8%, P=0.005), greater reductions in LDL-C, non-HDL-C, ApoB, and SCORE2, improved circadian blood pressure profile, and more pronounced reductions in arterial stiffness parameters. Treatment adherence and organ-protective effects were also superior in the polypill group, without adverse metabolic effects. Conclusion: Fixed-dose combination polypill therapy provides superior blood pressure and lipid control, improved circadian rhythm restoration, enhanced organ protection, and better adherence compared with free drug combinations in hypertensive patients with high and very high cardiovascular risk.
Keywords: Arterial hypertension, Dyslipidemia, Polypill, Fixed-dose combination, Cardiovascular risk, SCORE2, Blood pressure control, Lipid profile
Cite this paper: Shakhlo S. Fayzullaeva, Gulnoz A. Khamidullaeva, Guzal Zh. Abdullaeva, Zarina T. Mashkurova, Khafiza F. Yusupova, Nargiza R. Khanberdiyeva, Polypill-Based Therapy for Reduction of SCORE2 and Cardiovascular Risk in Patients with Arterial Hypertension and Dyslipidemia, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 220-228. doi: 10.5923/j.ajmms.20261601.49.
Article Outline
1. Introduction
- CVD remains the leading cause of disease burden and death worldwide with the greatest burden in low, low-middle, and middle SDI regions. As of 2023, there were 437 million (95% UI: 401 to 465 million) CVD DALYs globally, a 1.4-fold increase from the number in 1990 of 320 million (292 to 344 million). Ischemic heart disease, intracerebral hemorrhage, ischemic stroke, and hypertensive heart disease were the leading cardiovascular causes of DALYs in 2023 globally. [1]. According to the results of the WHO STEPS study on the prevalence of risk factors for non-communicable diseases in the Republic of Uzbekistan in July 2019, conducted among 4,320 people aged 18-69 years: In the Republic of Uzbekistan, 83.5% of deaths were from non-communicable diseases, of which 63.3% of deaths were from CVD [2].A wide range of risk factors are associated with IHD, including high systolic blood pressure (SBP), high low-density lipoprotein cholesterol (LDL-C), high fasting plasma glucose (FPG), high body mass index (BMI), smoking, dietary risks, and air pollution [3], [4]. The Framingham Heart Study data on the hypertensive population reported that more than 80% had at least one additional cardiovascular disease risk factor and these risk factors were dyslipidemia. Studies have consistently indicated that hypertension and hypercholesterolemia frequently coexist, causing what is known as dyslipidemic hypertension (DH) [5], [6].In a scientific article published in 2012, Dalal et al. [7] introduced the term of “LIPITENSION” into clinical practice, considering the prevalence of AH and dyslipidemia, the impact of these risk factors on patient prognosis, and the importance of early control.Several studies have indicated that the management of dyslipidemia may exert favorable effects on blood pressure (BP). Study [8] demonstrated that patients treated simultaneously with statins and antihypertensive agents exhibited additional BP reductions that could not be fully accounted for by either the cholesterol-lowering properties of statins or the isolated action of the antihypertensive drugs. Hypercholesterolemia itself may worsen BP regulation by amplifying the endothelial effects of vasoconstrictors such as endothelin-1 and angiotensin II. [9] Consequently, reduced nitric oxide availability together with an intensified vasoconstrictive response can promote increased BP in patients with dyslipidemia.In recent years, the concept of the “polypill” has been actively discussed. In 2003 by Wald [10] and Law has been actively discussed at international forums. In a meta- analysis, the authors showed that lowering low-density lipoprotein cholesterol (LDL-C) with statins by 1.8 mmol/L can reduce the risk of coronary heart disease (CHD) by 61% and stroke by 17%. Antihypertensive drugs included in the polypill at half the dosage help reduce diastolic blood pressure (DBP) by 11 mmHg, which leads to a 46% reduction in the risk of CHD and 63% of stroke. May substantially reduce the of CHD by 88% and stroke by 80%. In 2022, the results of the (NEPTUNO) study: patients were with confirmed CVDs and showed that the CNIC polypill significantly reduced the overall incidence of serious CVDs compared with three alternatives [11]. The results show that compared with the control group, patients in the polypill group had fewer cardiovascular events (22%; p = 0.017, 25%; p = 0.002, 27%; p = 0.001), achievement of BP and LDL- cholesterol targets was significantly higher in the CNIC polypill group than in the other groups. (BP: + 12.5% vs. + 6.3%; p < 0.05, + 2.2%; p < 0.01, + 2.4%; p < 0.01.), (LDL-C: +10.3% vs. +4.9%; p < 0.001, +5.7%; p < 0.001, +4.9%; p < 0.001 significant). By the end of the study, the CNIC-Polypill group showed a significant increase in treatment adherence compared to the other groups (72.1% vs. 62.2%, 60.0% vs. 54.2% significant; p < 0.001) [12].The 2018 ESC/ESH guidelines for hypertension management recommend to initiate therapy with a combination of two antihypertensive drugs, preferably in a single-pill fixed-dose combination (FDC), for most patients. [13] [14] In most clinical situations, the optimal starting fixed combination is a combination of an angiotensin II receptor blocker (ARB) or angiotensin-converting enzyme inhibitor (ACEI) with calcium channel blocker (CCB) or a diuretic, which significantly increases the effectiveness of antihypertensive therapy. An example is the combination of perindopril (4-8 mg) or lisinopril (10-20 mg) with amlodipine (5-10 mg).The objective of this study was to compare the effectiveness of a 6-month course of therapy using an FDC(polypill) and a free combination of antihypertensive drugs and statins in achieving target levels of SCORE-2 scale scores, lipid parameters by CVD group, blood pressure (BP) and evaluate organoprotective effectiveness of pharmacotherapy in AH patients with high and very high cardiovascular risk (CVR).
2. Materials and Methods
- The study included 119 patients with AH Grades 1-2 (ESC/ESH, 2018) and dyslipidemia aged 40 to 75 years, of both sexes. The exclusion criteria were: diabetes mellitus type 1 and 2, chronic kidney disease, genetic disorders of lipid metabolism (familial hypercholesterolemia), secondary arterial hypertension, gestational hypertension, atherosclerotic diseases of the cardiovascular system, including: stable angina FC II-IV, all types of unstable angina, post-infarction cardiosclerosis, acute cerebrovascular accident in history, as well as conditions after revascularization, chronic heart failure stage (NYHA FC>II), cardiac arrhythmia, severe metabolic diseases, kidney and liver diseases, as well as patient refusal to participate in the study. Office BP was measured using a mercury sphygmomanometer, according to Korotkov’s method.Blood pressure was measured three times, and the mean of these measurements was used in the analyses. The 24-hour ambulatory blood pressure monitoring (ABPM) was performed using the Cardiospy recorder (LABTechLTD, Hungary).Vascular stiffness was assessed by applanation tonometry using the SphygmoCor device (AtCor Medical, Australia), Blood levels of lipids, urea, creatinine, uric acid, glucose, ALT, AST, and CK-MB fraction were determined using a Daytona autoanalyzer (RANDOX, UK). The estimated glomerular filtration rate (eGFR) was calculated according to the CKD-EPI (2021) equation. Microalbuminuria (MAU) in morning urine was assessed by enzymatic analysis on the MindrayBS 380 biochemical analyzer (China), with a measurement range from 30 to 300 mg/L and higher. The 10-year risk of fatal and non-fatal cardiovascular events was assessed using the SCORE2 scale.In the study, patients were randomized into two groups by sealed-envelope randomization. Patients in both groups received dual combination antihypertensive therapy with statins (ACEI+CA+statin) The first group (n = 60) was recommended the combination drug "Polypill: " containing lisinopril/amlodipine/rosuvastatin. The second group (n = 59) was recommended a separate form of the combination of perindopril/amlodipine and rosuvastatin.During treatment, the dose of antihypertensive drugs was titrated every 2 weeks to achieve systolic blood pressure (SBP < 140 mmHg), diastolic blood pressure (DBP < 90 mmHg), low-density lipoprotein cholesterol (LDL < 100-70 mg/dL), non-HDL < 100-85 mg/dL) and triglycerides (< 150 mg/dL), as well as a 10% reduction in SCORE2 scores, and a 50% reduction in LDL-C from baseline, as assessed at the 12-week control visit, and the dose of statins was titrated. The drugs were prescribed at the following therapeutic doses: perindopril (4-8 mg/day), lisinopril (10-20 mg/day), amlodipine (5-10 mg/day) and rosuvastatin. Initially, rosuvastatin was administered at a moderate-intensity dose (10 mg/day), and if the target LDL-C levels was not achieved within 12 weeks of observation, the rosuvastatin dose was increased to a high-intensity dose of 20 mg/day, in accordance with ESC/EAS recommendations [15]. If adverse events occurred, patients were excluded from the study and completed a questionnaire indicating the reasons. All patients were examined before, after 3 month and 6-month pharmacotherapy.Statistical analysis was performed using the statistical Microsoft Excel and SPSS statistics version 26.0. Baseline characteristics were summarized as frequencies and percentages for categorical variables and as mean±standard deviation (SD) for continuous variables. The Mann-Whitney U test was used to compare the differences between the two independent groups, and the Wilcoxon test (W) was used to compare the mean values of dependent samples. Group comparisons for categorical variables were performed using a chi-square test. The probability value of P<0.05 was considered statistically significant.
3. Results
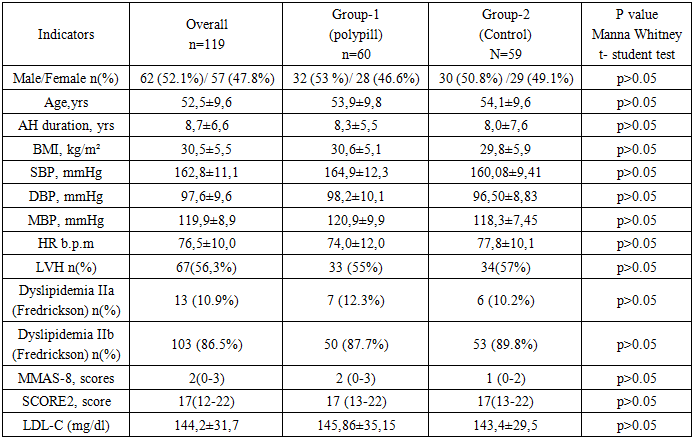
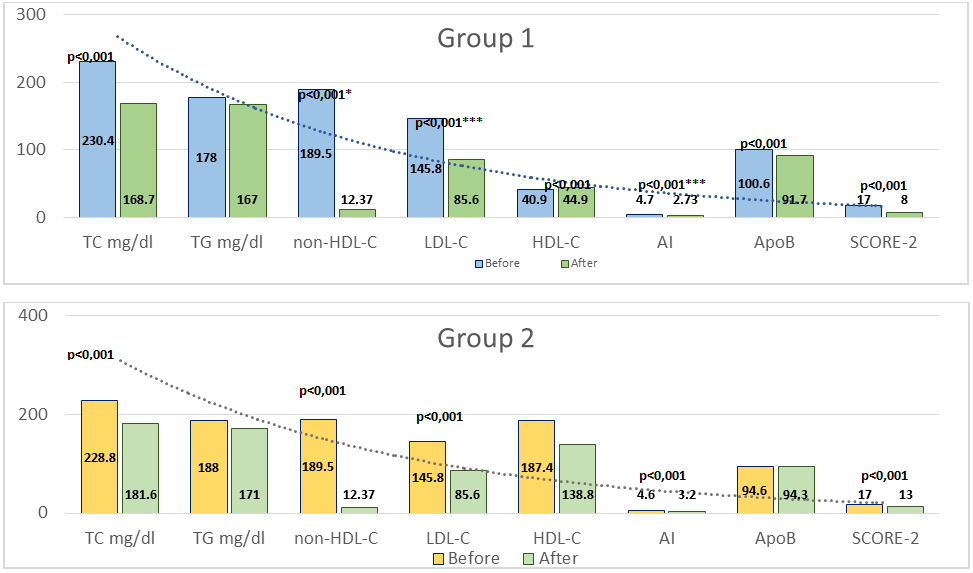
- Before the start pharmacotherapy the mean SBP value of patients was 162.8±11.1 mmHg, and the mean DBP value was 97.6±9.6 mmHg, the mean heart rate (HR) was 76.5±10.0 b.p.m. The mean age of patients was 52.5±9.6 years, and the mean duration of hypertension was 8.7±6.6 years. Of them, 62 (52.1%) were men and 57 (47.8%) were women. When calculating the body mass index of patients (Kettle formula ≥30 kg/m2), 78 (65.5%) patients were overweight and 67 (56.3%) patients had left ventricular hypertrophy according to the 2018 ESC/ESH criteria. In addition, all patients had dyslipidemia, with 13 (10.9%) patients having class IIa dyslipidemia and 103 (86.5%) patients having class IIb dyslipidemia according to the WHO Fredrickson classification. The average score of patients in assessing therapy adherence using the MMAS-8 scale was 1.6±1.5, and when stratified by CVD risk using the SCORE-2 scale, it was 17.9±7.9 points. Between the clinical characteristics of patients in the first and second groups no statistically significant differences were observed, and based on the clinical characteristics of the patients, all patients included in our study were classified as patients with a high and very high degree of CVD Table 1.
|
|
|
|
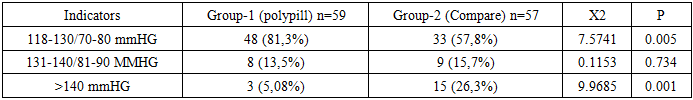
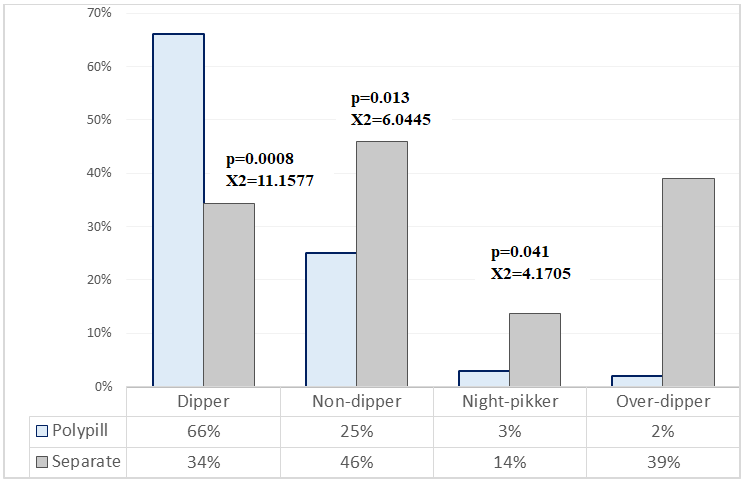 | Figure 1. Achievement of circadian rhythm parameters |
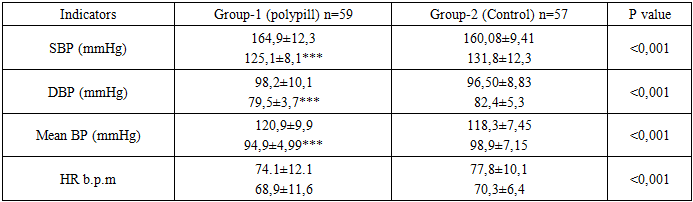
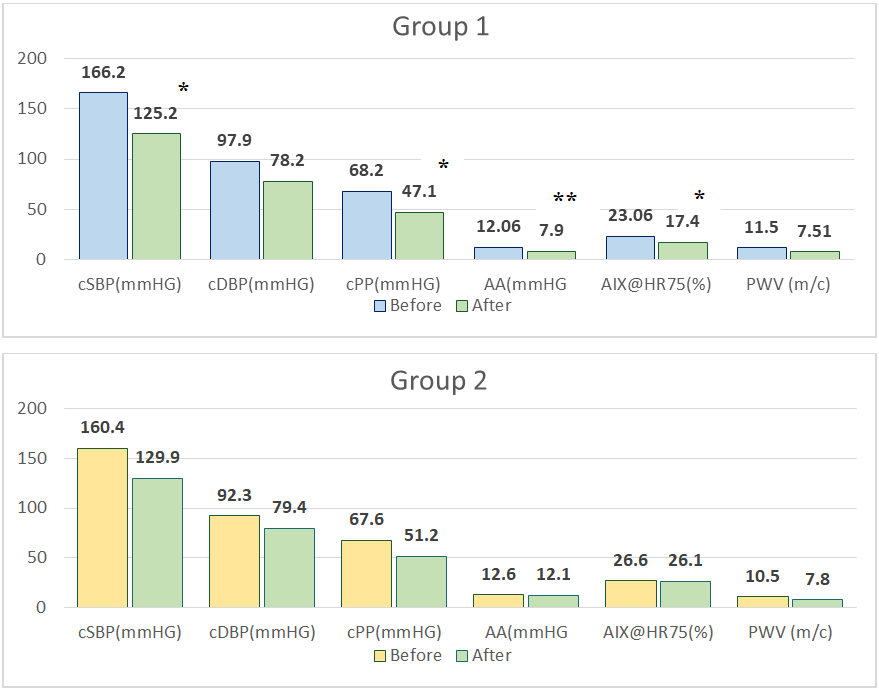 | Figure 2. Parameters of central hemodynamic and vascular stiffness before and after 6-month therapy in the study groups *, **, *** - P <0,05; P <0,01; P <0,001 between 1st and 2nd group |
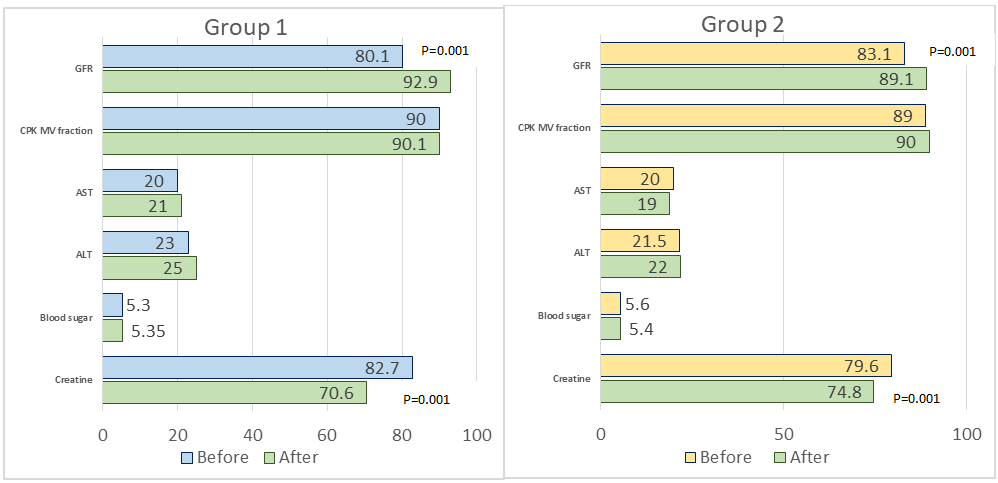 | Figure 3. Metabolic parameters before and after 6-month therapy in the study groups |
4. Discussion
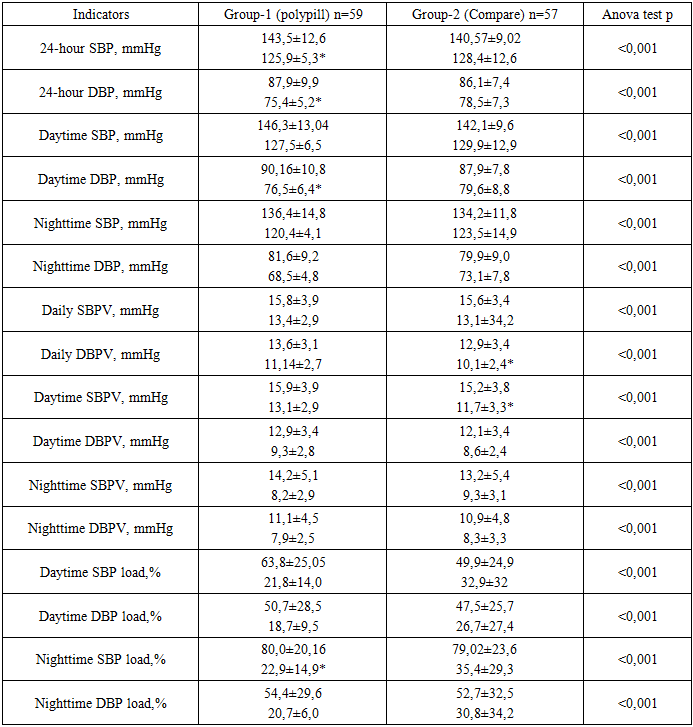
- The results of the present study confirm that fixed-dose combination (FDC) polypill therapy provides significant advantages over free combinations of antihypertensive drugs and statins in patients with arterial hypertension and dyslipidemia at high and very high cardiovascular risk. Our findings demonstrate superior blood pressure control, more pronounced lipid lowering, greater reduction in SCORE2 values, improved circadian blood pressure profile, and enhanced organ-protective effects in patients receiving polypill therapy. The superiority of combination antihypertensive therapy over monotherapy has been consistently demonstrated in large randomized clinical trials and meta-analyses. [16], [17], [18]. The ASCOT-BPLA study showed that the combination of a calcium channel blocker (CCB) with an angiotensin-converting enzyme inhibitor (ACEI) was significantly more effective in reducing cardiovascular events compared with traditional β-blocker–diuretic regimens. This benefit extended beyond blood pressure reduction and included lower rates of stroke, coronary events, new-onset diabetes, and renal impairment. The present study supports these observations by demonstrating that an ACEI–CCB-based fixed-dose regimen achieves more stable and effective blood pressure control, including superior nocturnal and 24-hour blood pressure regulation. Ambulatory blood pressure monitoring data in our study revealed a more favorable circadian blood pressure profile in the polypill group, with a higher prevalence of the “dipper” pattern [19] and a significant reduction in non-dipper and night-peaker phenotypes. Abnormal circadian blood pressure patterns are recognized independent predictors of cardiovascular morbidity and mortality [20]. Our findings extend this evidence by demonstrating that polypill therapy is particularly effective in restoring physiological circadian rhythm in high-risk patients. The favorable vascular effects observed in our study, including significant reductions in central systolic blood pressure, pulse pressure, augmentation index, and pulse wave velocity, further support the organoprotective potential of polypill therapy. Central hemodynamic parameters and arterial stiffness are increasingly recognized as more accurate markers of cardiovascular risk than peripheral blood pressure [19]. The observed improvements are consistent with previous reports indicating that ACEIs and CCBs exert synergistic effects on vascular compliance and endothelial function [21]. In addition, statin therapy has been shown to reduce arterial stiffness through pleiotropic mechanisms, including anti-inflammatory effects and improved nitric oxide bioavailability. The lipid-lowering effects of polypill therapy observed in our study were significantly greater than those achieved with free drug combinations. Reductions in LDL-C, non-HDL-C, apolipoprotein B, and atherogenic index were more pronounced in the polypill group, resulting in a greater decrease in SCORE2 cardiovascular risk estimates. These findings are consistent with the ASCOT-LLA study, which demonstrated substantial cardiovascular risk reduction following the addition of statin therapy to antihypertensive treatment [22]. Importantly, improved lipid control contributes not only to atherosclerotic plaque stabilization but also to improved vascular function, which may enhance blood pressure control [23]. Treatment adherence represents a critical determinant of long-term cardiovascular outcomes. Poor adherence is particularly common in patients requiring multiple medications. In the present study, higher adherence rates observed in the polypill group likely contributed to the superior clinical outcomes. These findings are in agreement with previous studies, including the NEPTUNO and CNIC-polypill investigations, which demonstrated improved adherence and reduced cardiovascular events with fixed-dose combination therapy compared with usual care [24], [25].
5. Conclusions
- In patients with arterial hypertension and dyslipidemia at high and very high cardiovascular risk, fixed-dose combination polypill therapy demonstrated superior efficacy compared with free drug combinations. Over a 6-month period, polypill therapy resulted in better blood pressure and lipid control, greater reduction in SCORE2 risk, improved circadian blood pressure patterns, and enhanced vascular and renal protection, without adverse metabolic effects. These findings support the use of polypill-based strategies for comprehensive cardiovascular risk reduction in high-risk hypertensive patients.
Ethical Considerations
- The study protocol was reviewed and approved by the Ethics Committee of the Republican Specialized Centre of Cardiology. All participants provided written informed consent. The data was only used for study purposes without individual details identifying the patient.
Competing Interests
- The authors declare that they have no competing interests.
Sources of Funding
- This research received no external funding
Abbreviations
- AH – Arterial hypertensionBP – Blood pressureSBP – Systolic blood pressureDBP – Diastolic blood pressureABPM – Ambulatory blood pressure monitoringCVD – Cardiovascular diseaseCVR – Cardiovascular riskLDL-C – Low-density lipoprotein cholesterolHDL-C – High-density lipoprotein cholesterolNon-HDL-C – Non-high-density lipoprotein cholesterolFDC – Fixed-dose combinationPWV – Pulse wave velocityAIx – Augmentation indexLVH – Left ventricular hypertrophyLVMI – Left ventricular mass indexCIMT – Carotid intima-media thicknesseGFR – Estimated glomerular filtration rateMAU – MicroalbuminuriaMMAS-8 – Morisky Medication Adherence ScaleESC – European Society of CardiologyESH – European Society of Hypertension
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML