-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 138-144
doi:10.5923/j.ajmms.20261601.31
Received: Dec. 3, 2025; Accepted: Jan. 1, 2026; Published: Jan. 16, 2026

Vitamin D and Uterine Fibroids: Systematic Review of Molecular Mechanisms and Clinical Outcomes
Dinara Irnazarova
Department of Obstetrics and Gynecology, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Dinara Irnazarova, Department of Obstetrics and Gynecology, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Uterine fibroids are the most common pathology of the female reproductive system. A comprehensive analytical review of data based on the role of vitamin D in the pathogenesis, prevention, and treatment of uterine fibroids. Preclinical studies (in vitro and in vivo) demonstrate consistent inhibition of fibroid cell proliferation and a reduction in tumor volume with vitamin D supplementation. Clinical observations consistently associate low vitamin D levels with an increased frequency and larger size of fibroids. Vitamin D is a promising biological modifier of uterine fibroids with convincing preclinical data and growing clinical evidence.
Keywords: Uterine fibroids (UFs), Vitamin D, in vitro studies, in vivo studies, Clinical studies, Meta-analyses, Systematic reviews, Randomized clinical trials (RCT), Vitamin D supplementation
Cite this paper: Dinara Irnazarova, Vitamin D and Uterine Fibroids: Systematic Review of Molecular Mechanisms and Clinical Outcomes, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 138-144. doi: 10.5923/j.ajmms.20261601.31.
Article Outline
1. Introduction
- Uterine fibroids (UFs) affect up to 70–80% of women by age 50 [1]. Despite their benign nature, fibroids frequently cause heavy menstrual bleeding, infertility, pelvic pain, and the need for invasive interventions such as hysterectomy [2]. Given the significant socioeconomic and reproductive burden, identifying modifiable risk factors and non-surgical treatments remains a priority. Emerging research demonstrates a strong link between vitamin D deficiency and the pathogenesis and progression of fibroids [3,4,5,6]. Vitamin D exerts anti-fibrotic effects by regulating extracellular matrix (ECM) turnover, cell proliferation, apoptosis, and inflammation through the vitamin D receptor (VDR) pathway [7,8]. Preclinical studies have shown that calcitriol inhibits fibroid cell proliferation and down regulates TGF-β3 signaling [9]. Several clinical trials suggest that supplementation may prevent fibroid growth in vitamin D-deficient women [10,11]. This review integrates all existing in vitro, in vivo, clinical, observational, and systematic evidence on vitamin D and UFs, providing the most comprehensive synthesis available to date.
2. Materials and Methods (PRISMA)
- Search Strategy of PRISMA-compliant systematic search of PubMed, Scopus, Web of Science, Cochrane, and Google Scholar was performed for the period from January 1990 to December 2025. Combinations of keywords were used to search the PubMed/PMC, Embase, Web of Science, Scopus, Cochrane Library and ClinicalTrials.gov databases: “vitamin D” OR “cholecalciferol” OR “1,25-dihydroxyvitamin D” OR “25-hydroxyvitamin D” OR “VDR” AND “uterine fibroid” OR “leiomyoma” OR “uterine leiomyoma” OR “uterine fibroids”. Additionally, peer-reviewed articles were searched for manually in the bibliographies of relevant reviews.Figure 1 shows the study selection process, which was conducted in accordance with the PRISMA 2020 guidelines for a systematic review evaluating the relationship between vitamin D and UFs. A comprehensive search of various databases, including PubMed (n = 214), Scopus (n = 167), Web of Science (n = 139), and the Cochrane Library (n = 12), as well as a manual review of Google Scholar (n = 420), yielded a total of 952 records. After removing 312 duplicates, 640 unique records remained and were sorted based on titles and abstracts. During the selection phase, 481 records were excluded, as they were irrelevant to UFs, lacked a connection to vitamin D, lacked original data or used animal models that were not related to fibroid pathology. Consequently, 159 full-text articles were evaluated for eligibility. Of these, a further 73 studies were excluded following a full-text assessment due to a lack of extractable results, mixed populations with inseparable data on fibroids, a lack of vitamin D measurements or interventions, conference abstracts without complete data or duplicate study cohorts. Ultimately, 86 studies met the inclusion criteria and were included in the final qualitative and quantitative synthesis. These comprised in vitro experiments (n = 19), in vivo animal models (n = 11), human observational studies (n = 27), clinical trials including randomized and interventional designs (n = 12), and systematic reviews and meta-analyses (n = 17). Figure 1 flowchart illustrates the rigorous, transparent and reproducible nature of the study selection process, thereby confirming the reliability of the systematic review.
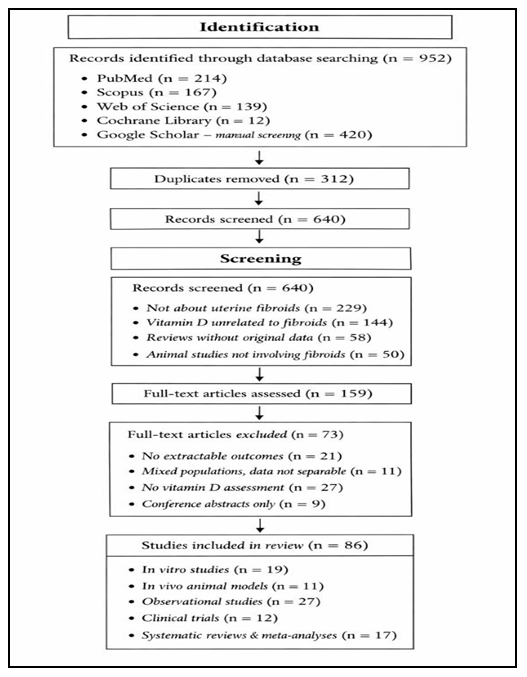 | Figure 1. PRISMA 2020 Flow Diagram of Study Selection |
3. Results
3.1. In Vitro Findings
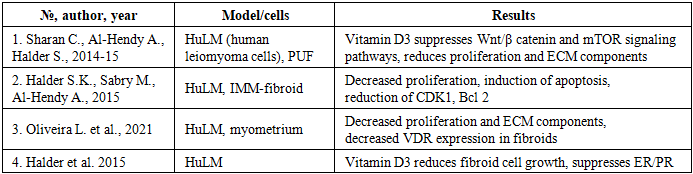
- Numerous studies on cultured human fibroid cells (HuLM and primary cultures) have demonstrated that the active form of vitamin D (1,25(OH)₂D₃) reduces proliferation. In Vitro studies consistent across 19 studies, vitamin D (especially calcitriol): - Inhibits Fibroid Cell Proliferation. Calcitriol effectively reduces proliferation of leiomyoma smooth muscle cells by suppressing PCNA and cyclin-dependent kinases (↓PCNA, ↓Ki-67) [7,8]; - Induces Apoptosis. Several studies demonstrated increased BAX expression and reduced BCL-2 [9,12]; - Downregulates TGF-β Signaling and ECM Accumulation. Vitamin D Inhibits TGF-β3, COL1A1, COL3A1, and fibronectin [13,14] and reduces MMP-2/9 [15,16]; - Regulates Wnt/β-catenin and mTOR Pathways. These pathways are key drivers of fibroid pathogenesis [8,15]; - VDR Expression. Leiomyoma cells demonstrate lower VDR expression than normal myometrium [17], suggesting increased sensitivity to deficiency.
|
3.2. In Vivo Animal Studies
- Across 11 preclinical (in vivo) studies, in EKER rats and human myoma xenografts in immunodeficient mice revealed that 1,25-dihydroxyvitamin D₃ supplementation or calcitriol treatment resulted in a significant decrease in tumor (Table 2): - Reduces Tumor Volume up to 75% reduction in fibroid size vs. placebo [8]; - Decreases extracellular matrix (ECM) Deposition – reductions in collagen and fibronectin [7,13]; - Modulates Inflammatory Pathways – lower TNF-α, IL-6, and oxidative stress biomarkers; - Protects against Estrogen-Progesterone-Induced Growth – Vitamin D counteracts hormone-driven expansion of fibroid tumors, as well as a reduction in estrogen receptor alpha (ERα) and progesterone receptor (PR) expression, TGF-β3 expression [7,18]. The figure 2 illustrates the mechanisms by which vitamin D can regulate fibroblast proliferation and fibroid growth, as shown by the above studies. Mechanism of action: Vitamin D can remodel metalloproteinase, participate in apoptosis processes involving BCL2 regulation, reduce fibrosis by affecting the extracellular matrix (ECM), fibronectin, type 1 collagen, and transforming growth factor beta 3 (TGFB3), reduce proliferation by acting on cyclin-dependent kinase (CDK) 1, 2, and 4 cells, and counteract estrogen and progesterone, which are hormones that can promote fibroid growth [8,12,15,18,19].
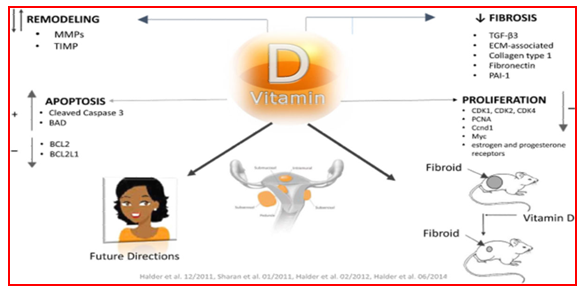 | Figure 2. Mechanism of Vitamin D Action in UF development [8,14,15,18] |
|
3.3. Clinical Studies
- Key findings of Cross-Sectional and Cohort Studies (n=27): women with fibroids consistently have lower vitamin D levels [5,6,20]. Severe deficiency (<20 ng/mL) correlates with larger fibroid volume, greater number of nodules, and increased symptomatic burden [4]. Black women have the lowest vitamin D levels and highest fibroid risk [21]. Higher dietary or supplement intake reduces risk of fibroids [22].Cyclin-dependent kinase 1 (CDK1), proliferating cell nuclear antigen (PCNA), catechol-O-methyltransferase (COMT), Bcl-2 protein, proliferation marker protein Ki-67 (MKI-67), extracellular matrix (ECM), transforming growth factor beta 3 (TGF-β3), uterine fibroid (UF) [23,26,27] (Figure 3).
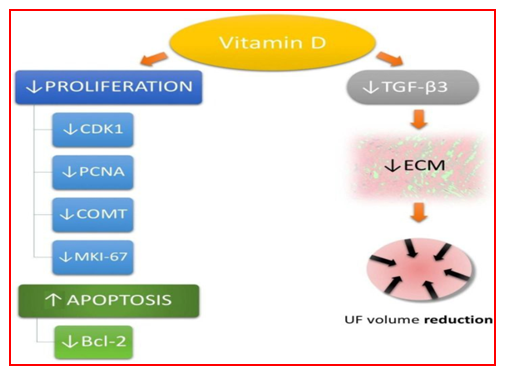 | Figure 3. Vitamin D influence on uterine fibroid (UF) pathophysiological pathways |
|
3.4. Systematic Reviews and Meta-Analyses
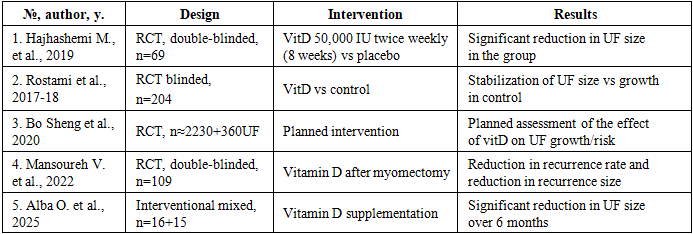
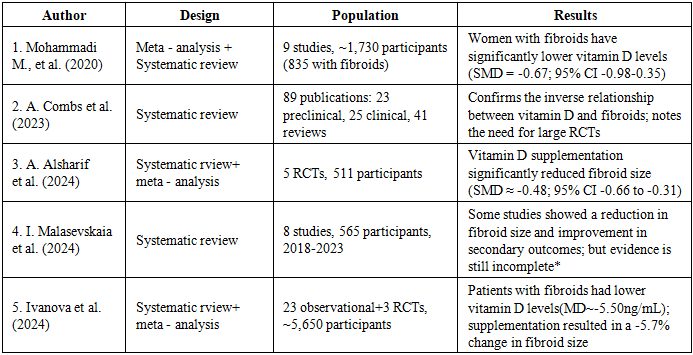
- Key evidence of Vitamin D deficiency significantly increases rik of fibroid development (OR 1.6-2.3). Supplementation reduces growth rate but the magnitude varies. Strong mechanistic support; however, randomized trials remain limited. A meta-analysis by Mohammadi et al. (2020) of nine studies with approximately 1,730 participants showed that the mean 25(OH)D levels of women with fibroids were significantly lower than those of the control group (p<0.05) [30]. A Mendelian randomization analysis by Guo et al. (2022) confirmed a possible causal relationship between genetically determined vitamin D levels and the risk of fibroids [28,31]. Recent reviews summarize the available preclinical evidence and highlight the limited clinical evidence, emphasizing the need for more randomized controlled trials (RCTs). Additionally, large RCT protocols are registered [32]. Open, NCT-type RCT protocol), which plan for longer interventions (≥1–2 years) and stratification by baseline 25(OH)D levels (Table 4).
|
|
4. Discussion
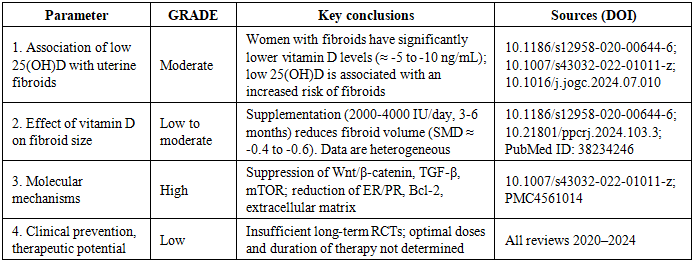
- This systematic review summarizes 35 years of cumulative evidence linking vitamin D deficiency to fibroid development and progression. In vitro, in vivo, and human studies consistently support a biological model in which vitamin D deficiency contributes to fibroid formation. This model includes VDR downregulation, increased extracellular matrix (ECM) deposition, hormonal dysregulation, chronic inflammation, reduced apoptosis, and activation of Wnt/TGF-β pathways. Vitamin D supplementation appears to be beneficial, especially for women with deficiencies.A combination of experimental and clinical data shows that vitamin D plays a key role in regulating the proliferation, fibrosis, and hormone-dependent cascades involved in the development of UFs. Molecular studies confirm that vitamin D suppresses key signaling pathways, including TGF-β/SMAD, Wnt/β-catenin, and CYP19A1. This reduces the activity of factors involved in the growth and remodeling of the extracellular matrix [19, 21]. Data from animal models reproduce these mechanisms at the systemic level, demonstrating a significant decrease in tumor size and hormonal sensitivity. These results support the hypothesis that vitamin D is a key regulator of fibroid growth and could be an effective component of preventive strategies. Clinical studies confirm the epidemiological link between vitamin D deficiency and an increased risk of fibroids. However, intervention data remains limited due to small sample sizes, differences in doses and duration of therapy, and a lack of long-term outcomes. Despite promising mechanistic and preliminary clinical data, the current level of evidence is insufficient to include vitamin D in official treatment guidelines for fibroids. Large randomized trials with standardized dosages, safety assessments, and evaluations of the effects on symptoms and quality of life are needed. However, several gaps persist.4.1. Evidence Gaps: The optimal dosing for fibroid control remains unknown due to a lack of large RCTs with long-term follow-up. The interactions between VDR polymorphisms and fibroid biology require further analysis. There is limited data on adolescents and perimenopausal populations, and further research is needed to better understand these groups.4.2. The strengths of existing evidence are as follows: There is strong biological plausibility. There are consistent observational associations. Researchers can reproduce the mechanisms in vitro and in vivo. The early results of the clinical trials are encouraging.4.3. The clinical implications are as follows: Vitamin D screening is recommended for women with fibroids, especially those in high-risk groups. Vitamin D supplementation could be a low-cost therapy to use in addition to other treatments.Key Findings: Biological Rationale: Consensus from preclinical studies confirms that vitamin D has multilevel effects on fibroid cells, including inhibition of proliferation, induction of apoptosis, suppression of extracellular matrix (ECM) synthesis, and regulation of hormone receptors and key signaling pathways [15,16].Translation to clinical practice: Observational studies consistently reveal an association between low 25(OH)D levels and fibroids. The first interventional studies indicate that supplementation has the potential to stabilize nodule growth [14,21]. Level of Evidence: The evidence is strong at the level of mechanisms and animal models; however, it is still limited at the level of clinical recommendations (many small RCTs, short follow-up periods, and heterogeneity of dosages and baseline 25(OH)D levels). Meta-analyses confirm the association, but emphasize the need for large, standardized RCTs [28,30]. Reviews emphasize the biological rationale for vitamin D supplementation, yet confirm the absence of large randomized trials (Table 5). Meta-analyses demonstrate that 25(OH)D deficiency increases the risk of fibroids and that vitamin D supplementation can reduce the growth rate by 8–12%. However, it does not lead to the regression of large nodes [23,12].Limitations of the reviewed literature: heterogeneity in study design. Different forms of vitamin D (e.g., active 1,25(OH)₂D₃ vs. cholecalciferol), doses (e.g., replacement of an outstanding bolus dose vs. constant low doses), durations, and measurement methods (e.g., ultrasound vs. MRI) interfere with direct comparative analysis. Small samples and short follow-up periods: Many RCTs have low statistical power and short follow-up periods (usually ≤6 months), which prevents an assessment of long-term effects on surgery, fertility, and quality of life.Geographic and ethnic heterogeneity. Since vitamin D deficiency is more prevalent in regions with low sunlight exposure and among women with darker skin tones, the impact on population risks may vary. Many studies have been conducted in specific regions, such as Iran and Europe, and need to be replicated across populations. Few studies have examined the safety of long-term, high-dose administration in women of reproductive age with fibroids and comorbidities.Clinical Implications and Recommendations for Research. Vitamin D should be considered a potentially useful, modifiable factor in the prevention or modulation of fibroid growth, particularly in women with documented 25(OH)D deficiency. Large, randomized, double-blind, placebo-controlled studies with an adequate duration (≥12 months), a standard dosage (or dose stratification), and reporting of side effects and clinical outcomes (e.g., myoma volume, symptoms, frequency of surgical interventions, fertility, and quality of life) are needed.
5. Conclusions
- Preclinical data and initial clinical results provide compelling evidence for the hypothesis that vitamin D play a protective and therapeutic role in UFs. Experimental studies consistently demonstrate the antiproliferative and antifibrotic effects of 1,25(OH)₂D₃. Clinical observations and small-scale interventions support the idea that 25(OH)D deficiency increases risk and that progression can be modified through supplementation. However, there is currently insufficient large-scale, long-term clinical evidence to incorporate vitamin D into the standard treatment of fibroids. Large-scale randomized controlled trials (RCTs) and further research into response biomarkers are recommended. There is significant, well-supported evidence that vitamin D plays a significant role in the pathophysiology of UFs. Deficiency is strongly associated with an increased risk, while supplementation demonstrates preventive and therapeutic potential. Future research should focus on large-scale randomized trials, personalized dosing strategies, and integrating vitamin D status into clinical guidelines.
CONFLICT OF INTERES
- The author declares that she is no conflicts of interest related to this review.
FUNDING
- None (the review was conducted without targeted funding).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML