Bekzod Rakhmatov1, Mirza-Ali Gafur-Akhunov1, Akhror Mamadaliev2
1Center for the Development of Professional Qualifications of Medical Workers, Tashkent, Uzbekistan
2Republican Scientific and Practical Medical Center of Oncology and Radiology, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Lung cancer remains one of the most significant challenges in modern oncology and continues to be the leading cause of cancer-related mortality worldwide. Due to the pronounced biological heterogeneity of non-small cell lung cancer (NSCLC), immunohistochemical (IHC) profiling plays a crucial role in characterizing tumor behavior, angiogenic activity, and mechanisms of immune evasion. The aim of this study was to analyze the expression of p53, CD34, and PD-L1 in the major histological subtypes of NSCLC. A retrospective descriptive analysis was performed on immunohistochemical data from 60 patients with morphologically verified NSCLC in the Navoi region. The study included three histological subtypes, with 20 cases each: squamous cell carcinoma, lung adenocarcinoma, and large cell carcinoma. Immunohistochemical staining was conducted using an automated Bond™ system (Leica, Australia) with monoclonal antibodies against p53, CD34, and PD-L1 and DAB visualization. Expression of p53 and PD-L1 was evaluated using a semi-quantitative scale (ALLRED-based, scores 0–3), while CD34 was used to assess tumor microvessel density in representative high-power fields. In squamous cell carcinoma, p53 expression was negative in 20% of cases and positive in 80%, with weak, moderate, and strong expression observed in 20%, 30%, and 30% of patients, respectively. PD-L1 expression in this group was negative in 20% and positive in 80% of cases (weak 25%, moderate 40%, strong 15%). In lung adenocarcinoma, p53 expression was positive in all cases, predominantly at moderate and high levels (35% and 50%, respectively), while PD-L1 demonstrated high positivity, with strong expression detected in 45% of patients. In large cell carcinoma, p53 expression was positive in all cases (weak 25%, moderate 40%, strong 35%), and PD-L1 positivity was observed in 85% of patients. CD34 expression was detected in 100% of cases across all histological subtypes, with higher microvessel density noted in adenocarcinoma and large cell carcinoma. The study demonstrates that immunohistochemical expression patterns of p53, CD34, and PD-L1 differ among histological subtypes of NSCLC and are associated with tumor aggressiveness. Lung adenocarcinoma is characterized by higher p53 and PD-L1 expression combined with pronounced angiogenic activity, indicating a more aggressive biological phenotype. Standardized immunohistochemical assessment of these markers may contribute to improved prognostic stratification and individualized treatment planning in patients with NSCLC.
Keywords:
Lung cancer, Incidence, Immunohistochemical examination, Markers
Cite this paper: Bekzod Rakhmatov, Mirza-Ali Gafur-Akhunov, Akhror Mamadaliev, Analysis of Immunohistochemical Study Results in Lung Cancer, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 115-122. doi: 10.5923/j.ajmms.20261601.27.
1. Introduction
Lung cancer remains one of the most urgent problems of modern oncology and continues to be the leading cause of cancer-related mortality worldwide. According to the GLOBOCAN 2020 database, approximately 2.16 million new cases of lung cancer are diagnosed each year and more than 1.8 million deaths are recorded annually, which places this malignancy first among all cancers in terms of mortality [1]. Despite progress in screening strategies, imaging, surgical techniques, radiotherapy, and systemic treatment, the overall prognosis remains unfavorable in many patients, largely due to late-stage detection, early dissemination, and pronounced biological heterogeneity of tumors [2,3].Non-small cell lung cancer (NSCLC) accounts for the majority of lung cancer cases and encompasses several major histological variants, including squamous cell carcinoma, adenocarcinoma, and large cell carcinoma [2]. These subtypes differ not only morphologically but also in their molecular and immunophenotypic characteristics, which directly determine tumor aggressiveness, metastatic potential, response to treatment, and survival outcomes [3,4]. The rapidly expanding knowledge about tumor biology has demonstrated that traditional histological classification alone is often insufficient for optimal clinical decision-making, as tumors within the same histological subtype may exhibit fundamentally different behavior and treatment sensitivity due to underlying molecular alterations and immune microenvironment features [3–5].In this context, immunohistochemistry (IHC) has become a cornerstone method in contemporary oncopathology, bridging classical morphology with molecular oncology. IHC enables the evaluation of key protein markers in routinely processed biopsy or surgical tissue, thereby supporting diagnostic verification, characterization of tumor histogenesis, assessment of proliferative and apoptotic activity, estimation of angiogenesis, and evaluation of immune evasion mechanisms [2,3,6]. This is particularly relevant for healthcare settings where access to comprehensive molecular testing may be limited; in such circumstances, IHC remains a cost-effective, reproducible, and widely applicable approach for clinically meaningful tumor stratification [4,6].Among the most clinically informative markers in NSCLC are p53, CD34, and programmed death-ligand 1 (PD-L1). Alterations in p53 reflect genomic instability and dysregulation of cell-cycle control, which are central hallmarks of malignant progression and are commonly linked to more aggressive tumor behavior [7]. CD34 is widely used to assess microvessel density and angiogenic activity within the tumor microenvironment, providing indirect evidence of the tumor’s capacity for invasion and hematogenous dissemination [8]. PD-L1 expression is a critical immune checkpoint-related biomarker associated with tumor immune escape and serves as an important predictive factor for the potential efficacy of immune checkpoint inhibitor therapy [6,17]. Importantly, unjustified use of immunotherapy without appropriate biomarker confirmation may not only be ineffective but may also expose patients to unnecessary toxicity and resource burden; therefore, reliable assessment of PD-L1 and related features is increasingly essential in routine practice [6,12,14,15].Given the clinical need to individualize treatment strategies (surgery, neoadjuvant and adjuvant approaches, and immunotherapy), comprehensive immunohistochemical assessment of tumor markers across different histological variants of NSCLC is highly relevant and practically significant. Systematic analysis of p53, CD34, and PD-L1 expression contributes to a deeper understanding of tumor biology, supports prognostic and predictive stratification, and can strengthen therapeutic decision-making aimed at improving clinical outcomes in patients with lung cancer [3,4,6].
2. Materials and Methods
This study was based on a retrospective descriptive analysis of immunohistochemical findings in patients with non-small cell lung cancer (NSCLC). A total of 60 patients with histologically confirmed NSCLC were included in the study. Tumor tissue samples were obtained during diagnostic biopsy or surgical interventions and subsequently subjected to pathological and immunohistochemical examination. All cases were examined under the conditions of the Navoi region, and the diagnosis of lung cancer was verified according to standard histopathological criteria.Histological classification of tumor specimens was performed in accordance with the World Health Organization (WHO) classification of lung tumors. The study cohort comprised three major histological subtypes of NSCLC, each represented by 20 cases: squamous cell carcinoma of the lung, lung adenocarcinoma, and large cell carcinoma of the lung. Such distribution enabled a comparative assessment of immunohistochemical marker expression among different histological variants of NSCLC.Immunohistochemical analysis was carried out on formalin-fixed, paraffin-embedded tissue blocks. Sections 4 μm thick were prepared from paraffin blocks, mounted on poly-L-lysine-coated glass slides, and dried at room temperature for 24 hours, followed by additional drying in a thermostat at 55–60°C for 60 minutes. Deparaffinization was performed using ortho-xylene, after which the sections were rehydrated through graded ethanol solutions. Antigen retrieval was achieved using a demasking buffer in water heated to 98°C for 30–40 minutes. Endogenous peroxidase activity was blocked with a 3% hydrogen peroxide solution.Immunohistochemical staining was performed using an automated immunohistochemical processor Bond™ (Leica, Australia). Monoclonal antibodies against p53, CD34, and programmed death-ligand 1 (PD-L1) were applied in accordance with the manufacturer’s recommendations. Visualization of antigen–antibody reactions was achieved using a diaminobenzidine (DAB) chromogen system, followed by counterstaining with Mayer’s hematoxylin. After staining, the sections were dehydrated, cleared, and mounted for microscopic evaluation.Evaluation of p53 and PD-L1 expression was carried out using the semi-quantitative ALLRED scoring system, which is based on the proportion of positively stained tumor cells. The results were categorized as negative (0 points), weak positive reaction with less than 30% of positive cells (1 point), moderate positive reaction with 30–60% positivity (2 points), and strong positive reaction with more than 60% of positive tumor cells (3 points). CD34 expression was assessed by identifying endothelial cell staining and was used to estimate tumor microvessel density. The number of stained microvessels was evaluated in representative high-power fields (objective ×10, ocular ×40), allowing assessment of angiogenic activity within the tumor microenvironment.The immunohistochemical data obtained were analyzed using descriptive statistical methods. The distribution and frequency of marker expression levels were calculated for each histological subtype of NSCLC and expressed as absolute values and percentages. Comparative analysis was performed to identify differences in the expression patterns of p53, CD34, and PD-L1 among squamous cell carcinoma, adenocarcinoma, and large cell carcinoma of the lung. The stages of the immunohistochemical study are summarized in Table 1.Table 1. Stages of immunohistochemical (IHC) examination
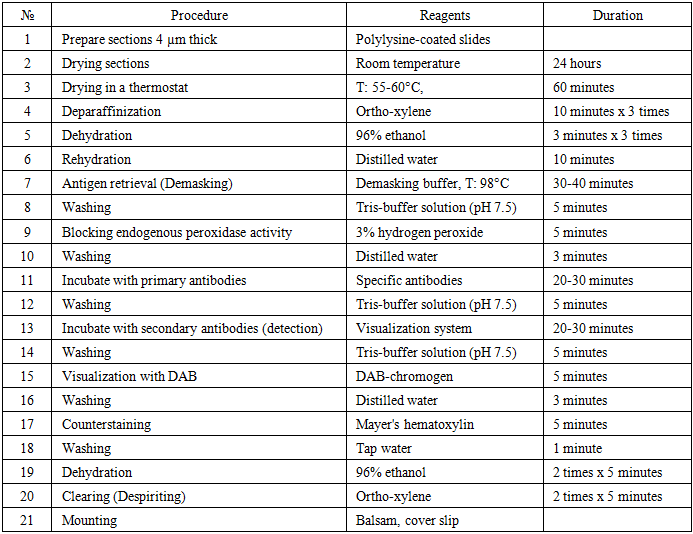 |
| |
|
3. Results and Discussions
In our study, immunohistochemical examination was performed in 60 patients. The expression of the p53 tumor suppressor gene, the CD34 marker, and the PD-L1 marker was evaluated, with each marker being analyzed in 20 patients.Table 2. The following markers were selected for immunohistochemical analysis
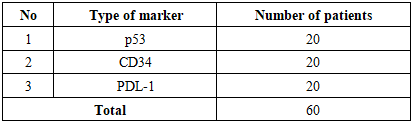 |
| |
|
This investigation is of great importance for identifying molecular structures at the cellular level, studying cellular localization, assessing tumor spread and histogenesis, monitoring the development of precancerous processes, determining prognostically significant complications, defining tumor stage and treatment strategy, enabling dynamic follow-up and control of therapeutic interventions, and identifying risk groups predisposed to the development of malignant neoplasms.Table 3. Histological types of non-small cell lung cancer
 |
| |
|
Immunohistochemical examination was performed in the following histological types of lung cancer: squamous cell carcinoma of the lung in 20 patients, lung adenocarcinoma in 20 patients, and large cell carcinoma of the lung in 20 patients. In patients with non-small cell (squamous cell) lung cancer, the levels of p53 marker expression (high, moderate, and weak) were evaluated. The obtained results were assessed using the ALLRED scoring system, which takes into account the percentage of positively stained tumor cells and the intensity of receptor staining. These parameters were subsequently combined to yield a semi-quantitative score ranging from 1 to 3. The minimum score was defined as 0 (negative), while scores of 1, 2, and 3 corresponded to weak positive reaction (10–30%), moderate positive reaction (30–60%), and strong positive reaction (60–100%), respectively.In this analysis, among 20 patients, weak positive p53 expression was observed in 4 cases (20%), moderate positive expression in 6 cases (30%), strong positive expression in 6 cases (30%), and negative expression in 4 cases (20%) (Table 4).Table 4. p53 expression levels in non-small cell (squamous cell) lung cancer
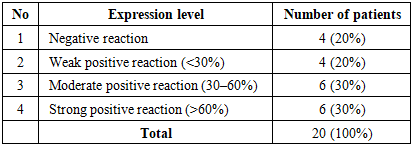 |
| |
|
On microscopic examination, the tumor cells with squamous epithelial differentiation demonstrated marked polymorphism, with hyperchromatic nuclei and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. The tumor cell nuclei exhibited intense dark-brown staining. 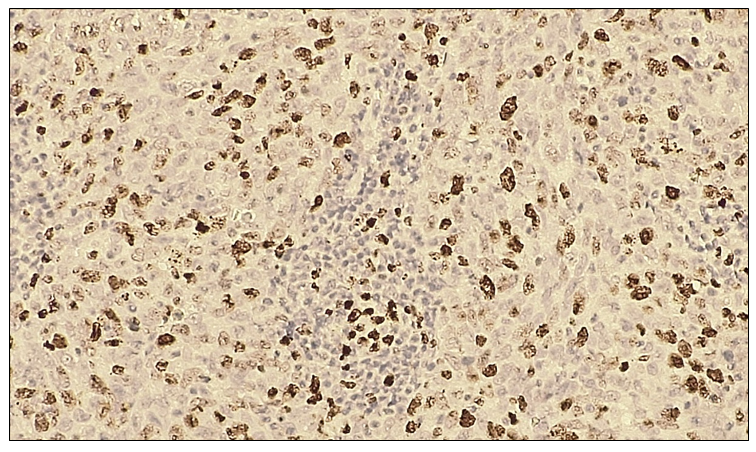 | Figure 1. Negative p53 immunohistochemical reaction in non-small cell (squamous cell) lung cancer. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In non-small cell (squamous cell) lung cancer, CD34 protein expression was observed in all 20 patients, demonstrating a 100% positive reaction. This positivity was characterized by distinct staining of endothelial cells lining the vessel walls and facilitated assessment of microvessel density in the present study. In representative high-power fields (objective ×10, ocular ×40), the number of blood vessels was clearly visualized. The pronounced vascularization of the tumor tissue indicates a high angiogenic activity, which may reflect an increased metastatic potential of the tumor.Microscopically, tumor cells with squamous epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels with intensely dark-brown–stained walls. A microvessel density of approximately 10–15 vessels per field of view was identified.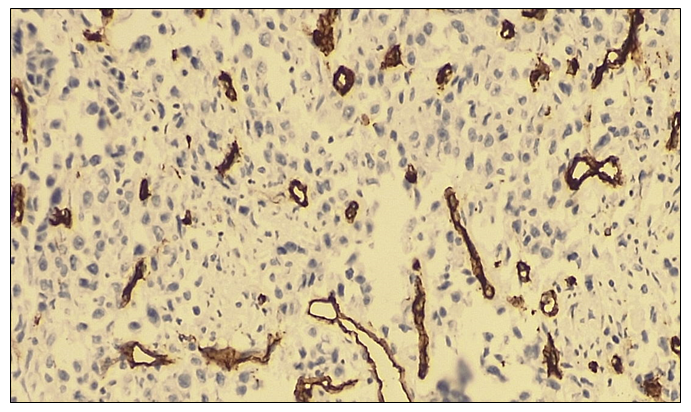 | Figure 2. Negative CD34 immunohistochemical reaction in non-small cell (squamous cell) lung cancer. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In non-small cell (squamous cell) lung cancer, PD-L1 expression was evaluated using the ALLRED scoring system, with the obtained data integrated into a semi-quantitative scale ranging from 1 to 3. A minimum score of 0 indicated a negative reaction, while scores of 1, 2, and 3 corresponded to weak positive (10–30%), moderate positive (30–60%), and strong positive (60–100%) expression, respectively. Among the 20 patients with non-small cell (squamous cell) lung cancer, weak PD-L1 expression was observed in 5 cases (25%), moderate expression in 8 cases (40%), strong expression in 3 cases (15%), and negative expression in 4 cases (20%) (Table 5).Table 5. PD-L1 expression levels in non-small cell (squamous cell) lung cancer
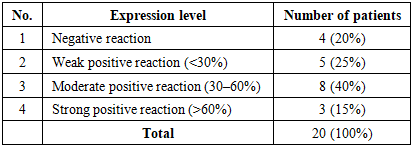 |
| |
|
On microscopic examination, tumor cells with squamous epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. No positive nuclear reaction was observed in the tumor cells. The membranes of malignant tumor cells demonstrated light brown staining.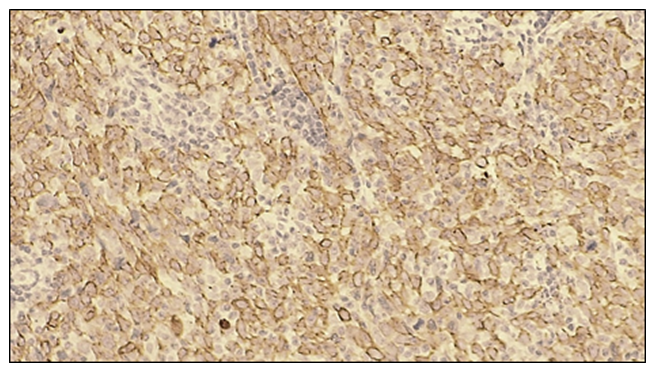 | Figure 3. Moderate positive PD-L1 immunohistochemical reaction in non-small cell (squamous cell) lung cancer. The membranes of tumor cells exhibit intense dark-brown staining. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In 20 patients with non-small cell lung adenocarcinoma, p53 marker expression was evaluated using the ALLRED scoring system. The analysis demonstrated weak positive expression in 3 cases (15%), moderate positive expression in 7 cases (35%), and strong positive expression in 10 cases (50%). No negative p53 expression was observed in this group (Table 6).Table 6. p53 expression levels in non-small cell lung adenocarcinoma
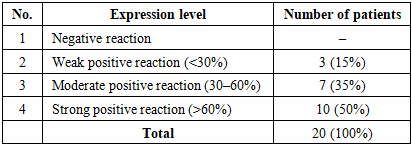 |
| |
|
On microscopic examination, tumor cells with glandular epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. The tumor cell nuclei demonstrated a strong positive reaction with intense dark-brown staining.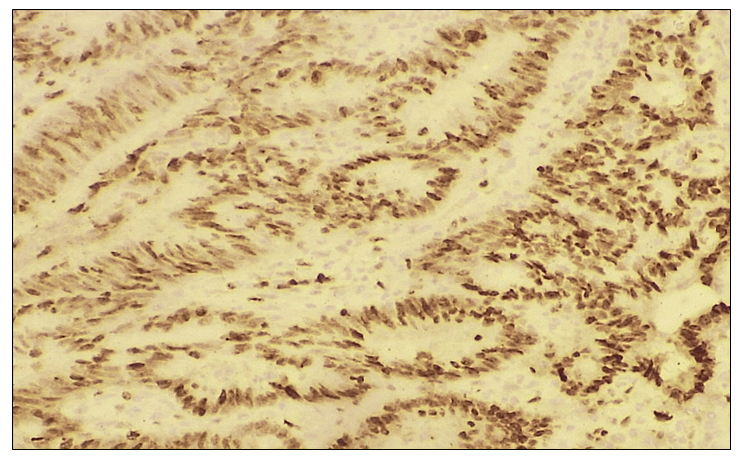 | Figure 4. Negative p53 immunohistochemical reaction in non-small cell lung adenocarcinoma. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In all 20 selected patients with non-small cell lung adenocarcinoma, CD34 expression was evaluated to assess tumor vascularity and its association with metastatic potential. A 100% positive reaction was observed in all cases, characterized by distinct staining of endothelial cells lining the vessel walls. This finding facilitated the assessment of microvessel density in the present study, with a clearly visible number of blood vessels per high-power field (objective ×10, ocular ×40). The pronounced vascularization of the tumor tissue indicates a high angiogenic activity, suggesting an increased potential for tumor metastasis.Microscopically, tumor cells with glandular epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels with intensely dark-brown–stained walls. A high microvessel density, ranging from 30–40 or more blood vessels per field of view, was identified.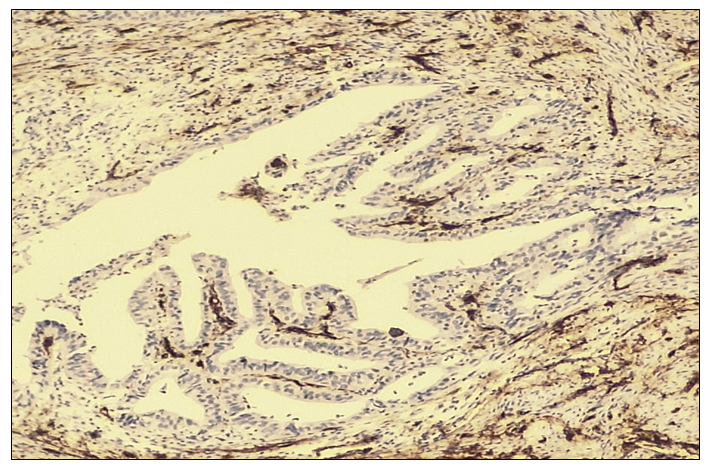 | Figure 5. Negative CD34 immunohistochemical reaction in non-small cell lung adenocarcinoma. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In non-small cell lung adenocarcinoma, PD-L1 expression was evaluated using the ALLRED scoring system. Among the 20 patients with lung adenocarcinoma, weak positive PD-L1 expression was observed in 4 cases (20%), moderate positive expression in 7 cases (35%), and strong positive expression in 9 cases (45%).Table 7. PD-L1 expression levels in non-small cell lung adenocarcinoma
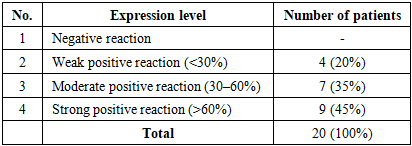 |
| |
|
On microscopic examination, tumor cells with glandular epithelial differentiation demonstrated marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. No positive nuclear reaction was observed in the tumor cells. The membranes of malignant tumor cells exhibited intense dark-brown staining.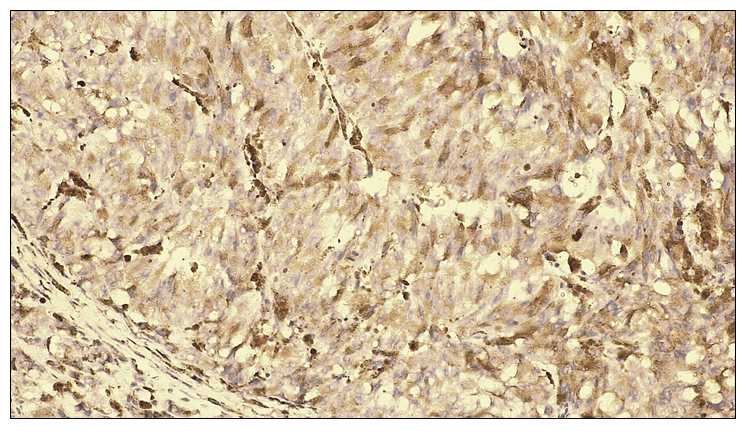 | Figure 6. Strong positive PD-L1 immunohistochemical reaction in non-small cell lung adenocarcinoma. The membranes of tumor cells exhibit intense dark-brown staining. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In 20 patients diagnosed with large cell carcinoma of the lung, p53 marker expression was evaluated using the ALLRED scoring system. The analysis revealed weak positive expression in 5 cases (25%), moderate positive expression in 8 cases (40%), and strong positive expression in 7 cases (35%). No negative p53 expression was observed in this group.Table 8. p53 expression levels in large cell carcinoma of the lung
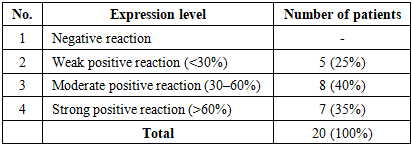 |
| |
|
On microscopic examination, tumor cells with large-cell epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. The tumor cell nuclei demonstrated a strong positive reaction with intense dark-brown staining.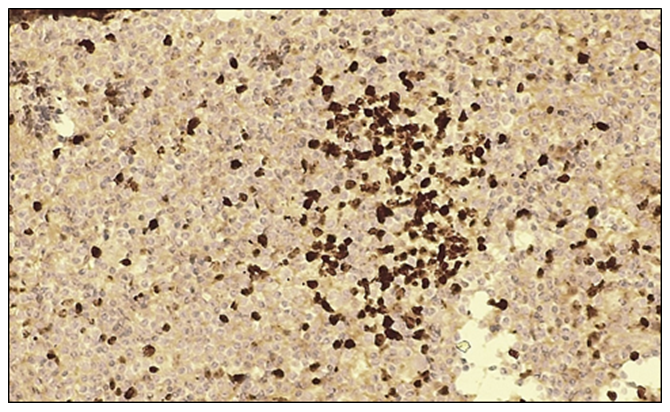 | Figure 7. Moderate positive p53 immunohistochemical reaction in large cell carcinoma of the lung. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In large cell carcinoma of the lung, CD34 marker expression was evaluated to assess tumor vascularity and its association with metastatic potential. The results were interpreted as negative or positive reactions. A positive CD34 reaction was observed in all 20 patients (100%), characterized by distinct staining of endothelial cells lining the vessel walls. This finding facilitated the assessment of microvessel density in the present study, with several blood vessels clearly visualized per high-power field (objective ×10, ocular ×40). The pronounced vascularization of the tumor tissue indicates high angiogenic activity, suggesting an increased metastatic potential of the tumor.Microscopically, tumor cells with large-cell epithelial differentiation exhibited marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels with intensely dark-brown–stained walls. A high microvessel density of approximately 30–40 or more blood vessels per field of view was identified.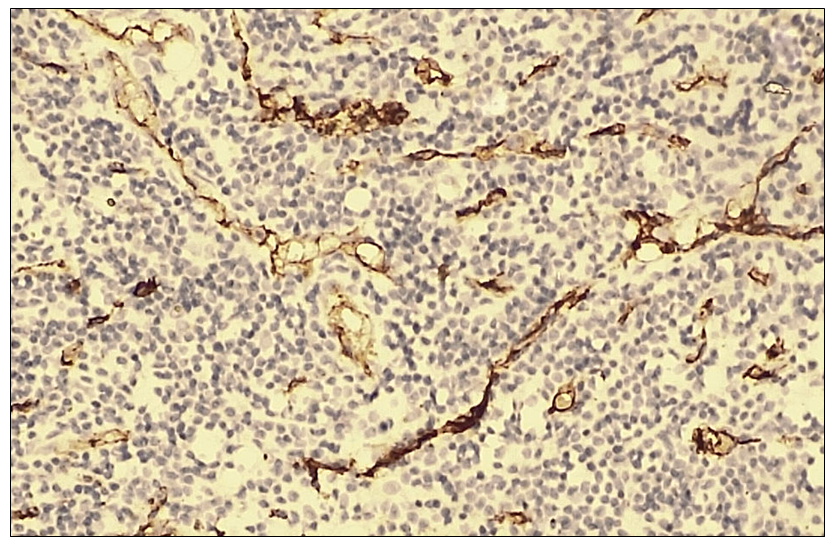 | Figure 8. Negative CD34 immunohistochemical reaction in large cell carcinoma of the lung. IHC, DAB chromogen. Objective ×10, ocular ×40 |
In large cell carcinoma of the lung, PD-L1 marker expression was evaluated in 20 patients. Weak positive PD-L1 expression was observed in 2 cases (10%), moderate positive expression in 10 cases (50%), strong positive expression in 5 cases (25%), and negative expression in 3 cases (15%).Table 9. PD-L1 expression levels in large cell carcinoma of the lung
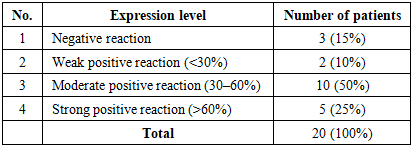 |
| |
|
On microscopic examination, tumor cells with large-cell epithelial differentiation demonstrated marked polymorphism, hyperchromatic nuclei, and numerous pathological mitotic figures. The epithelial stroma contained angiomatous blood vessels and loose fibrous connective tissue. No positive nuclear reaction was observed in the tumor cells. The membranes of malignant tumor cells exhibited intense dark-brown staining.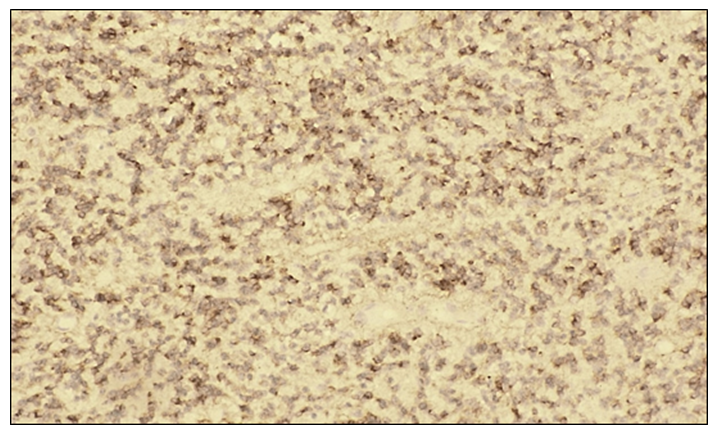 | Figure 9. Strong positive PD-L1 immunohistochemical reaction in large cell carcinoma of the lung. The membranes of tumor cells exhibit intense dark-brown staining. IHC, DAB chromogen. Objective ×10, ocular ×40 |
The results of the immunohistochemical analysis indicate that, across the histological subtypes of non-small cell lung cancer, the expression levels of molecular-genetic markers in tumor cells are closely associated with tumor aggressiveness. These markers reflect the biological behavior of tumor cells and have significant importance for selecting appropriate treatment strategies and determining prognosis in affected patients. Therefore, assessment of immunohistochemical marker expression plays a crucial role in guiding therapeutic decision-making in non-small cell lung cancer.The present study demonstrates that immunohistochemical expression patterns of p53, CD34, and PD-L1 differ significantly among the main histological subtypes of non-small cell lung cancer (NSCLC) and are closely associated with tumor aggressiveness and biological behavior. These findings support the growing body of evidence that immunohistochemical profiling plays a crucial role in understanding tumor heterogeneity and guiding personalized therapeutic approaches [1,2].Altered p53 expression was detected in all NSCLC subtypes examined in this study. Lung adenocarcinoma exhibited predominantly moderate to strong p53 positivity, with no negative cases observed, indicating a higher degree of genomic instability and disruption of cell-cycle regulation. In contrast, squamous cell carcinoma and large cell carcinoma showed heterogeneous p53 expression, including negative reactions in a subset of patients. Similar patterns have been reported in previous studies, where increased p53 expression was associated with aggressive tumor behavior, higher proliferation rates, and unfavorable prognosis in NSCLC [3,10].Angiogenesis, assessed using the CD34 marker, was highly pronounced across all histological subtypes, with 100% positivity observed in the study cohort. However, microvessel density was notably higher in lung adenocarcinoma and large cell carcinoma compared with squamous cell carcinoma. Enhanced angiogenic activity is a key mechanism facilitating tumor growth, invasion, and metastatic spread, and increased CD34 expression has been linked to poor clinical outcomes in lung cancer patients [11,12]. The uniformly high CD34 positivity observed in this study underscores the fundamental role of tumor vascularization in NSCLC progression.PD-L1 expression showed marked variability among histological subtypes, with lung adenocarcinoma demonstrating the highest proportion of strong positive cases. This finding suggests more active immune evasion mechanisms mediated through the PD-1/PD-L1 axis in adenocarcinoma. Previous studies have confirmed that elevated PD-L1 expression is associated with advanced disease stage, increased metastatic potential, and response to immune checkpoint inhibitor therapy [6,7]. The heterogeneous PD-L1 expression observed in squamous cell carcinoma and large cell carcinoma further emphasizes the necessity of biomarker-based patient selection prior to immunotherapy initiation.Taken together, the combined immunohistochemical profile characterized by elevated p53 expression, increased angiogenesis, and high PD-L1 expression indicates a more aggressive biological phenotype in lung adenocarcinoma compared with other NSCLC subtypes. These results highlight the importance of integrating immunohistochemical markers into routine diagnostic workflows to improve prognostic stratification, guide therapeutic decision-making, and advance precision oncology in lung cancer management [2,6].
4. Conclusions
In conclusion, the present study demonstrates that immunohistochemical expression of p53, CD34, and PD-L1 varies significantly among histological subtypes of non-small cell lung cancer and reflects differences in tumor aggressiveness and biological behavior. Lung adenocarcinoma is characterized by higher expression levels of all three markers, indicating increased genomic instability, enhanced angiogenic activity, and more pronounced immune evasion mechanisms compared with squamous cell carcinoma and large cell carcinoma.The findings confirm that immunohistochemical profiling provides valuable prognostic and predictive information that can support clinical decision-making, particularly in the selection of therapeutic strategies such as immunotherapy and anti-angiogenic approaches. Incorporation of p53, CD34, and PD-L1 assessment into routine diagnostic practice may improve individualized treatment planning and contribute to better outcome prediction in patients with NSCLC.Overall, the study highlights the significance of standardized immunohistochemical evaluation within the IMRAD framework and supports its role in advancing precision oncology for lung cancer management.
References
| [1] | Sung H., Ferlay J., Siegel R.L., et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians. 2021; 71(3): 209–249. |
| [2] | Travis W.D., Brambilla E., Nicholson A.G., et al. The 2015 World Health Organization classification of lung tumors. Journal of Thoracic Oncology. 2015; 10(9): 1243–1260. |
| [3] | Herbst R.S., Morgensztern D., Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018; 553(7689): 446–454. |
| [4] | Lindeman N.I., Cagle P.T., Aisner D.L., et al. Updated molecular testing guideline for the selection of lung cancer patients for targeted therapies. Journal of Thoracic Oncology. 2018; 13(3): 323–358. |
| [5] | Pao W., Girard N. New driver mutations in non-small-cell lung cancer. The Lancet Oncology. 2011; 12(2): 175–180. |
| [6] | Yu H., Boyle T.A., Zhou C., Rimm D.L., Hirsch F.R. PD-L1 expression in lung cancer. Journal of Thoracic Oncology. 2016; 11(7): 964–975. |
| [7] | Reck M., Rodríguez-Abreu D., Robinson A.G., et al. Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. New England Journal of Medicine. 2016; 375(19): 1823–1833. |
| [8] | Hanahan D., Weinberg R.A. Hallmarks of cancer: The next generation. Cell. 2011; 144(5): 646–674. |
| [9] | Altorki N.K., Markowitz G.J., Gao D., et al. The lung microenvironment: An important regulator of tumour growth and metastasis. Nature Reviews Cancer. 2019; 19(1): 9–31. |
| [10] | Olivier M., Hollstein M., Hainaut P. TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harbor Perspectives in Biology. 2010; 2(1): a001008. |
| [11] | Weidner N. Tumour angiogenesis: review of current applications in tumor prognostication. Seminars in Diagnostic Pathology. 1993; 10(4): 302–313. |
| [12] | Fontanini G., Boldrini L., Chine S., et al. Angiogenesis in non-small cell lung carcinoma: microvessel density is an independent prognostic indicator. Cancer. 1997; 79(2): 363–369. |
| [13] | Beysenova A.R. The role of immunohistochemical studies in oncology. Medicine and Ecology. 2015; 2: 12–16. (in Russian) |
| [14] | Kaprin A.D., Starinsky V.V., Shakhzadova A.O. The state of cancer care for the population of Russia in 2022. Moscow: P.A. Herzen Moscow Research Institute of Oncology; Russian Center for Information Technologies and Epidemiological Research in Oncology; 2023. (in Russian) |
| [15] | Brierley J.D., Gospodarowicz M.K., Wittekind C. TNM classification of malignant tumours. 8th ed. Oxford: Wiley-Blackwell; 2017. Russian edition: Dubovoy E.A., Pavlova K.A. (eds.). Moscow: Logosfera; 2018. (in Russian) |
| [16] | Tillyashaykhov M.N., Ibragimova Sh.N., Djanklich S.M. State of oncological care for the population of the Republic of Uzbekistan in 2019. Tashkent: Ministry of Health of the Republic of Uzbekistan; Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology; Academy of Sciences of the Republic of Uzbekistan; 2022. (in Russian) |
| [17] | Tillyashaykhov M.N., Salomov M.S. Clinical features of immunohistochemical research methods. Eurasian Journal of Medical and Natural Sciences. 2023; 3(5): 107–111. |








 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML










