-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 105-110
doi:10.5923/j.ajmms.20261601.25
Received: Dec. 22, 2025; Accepted: Jan. 13, 2026; Published: Jan. 16, 2026

Comparative Analysis of Treatment Outcomes in Patients According to CYP11B2 (–344 T/C) Polymorphism Genotypes
Tosheva Kh. B.1, Gadayev A. G.2, Boboev A. T.3, Turakulov R. I.2
1Bukhara State Medical Institute, Bukhara, Uzbekistan
2Tashkent Medical University, Tashkent, Uzbekistan
3Republican Specialized Scientific-Practical Medical Center of Hematology, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This article presents a comparative analysis of treatment outcomes in patients with chronic heart failure (CHF) and cardiorenal syndrome (CRS) according to the aldosterone synthase gene CYP11B2 (–344 T/C) polymorphism. The study included 200 CHF patients (100 with CRS, 100 without CRS) and 40 apparently healthy controls. CYP11B2 –344 T/C genotypes (TT, TC, CC) were determined by PCR–RFLP. Clinical status, 6 minute walk distance, echocardiographic indices, and serum levels of aldosterone, TGF β1 and collagen IV were assessed before and after 6 months of guideline directed medical therapy (ACEI/ARB, β blocker, mineralocorticoid receptor antagonist) combined with the SGLT2 inhibitor dapagliflozin. The TT genotype and T allele were associated with higher aldosterone and profibrotic biomarker levels, more pronounced renal dysfunction and a poorer response to therapy. In contrast, the CC genotype and C allele showed a more favourable clinical and haemodynamic response and lower fibrosis activity. These findings suggest that CYP11B2 (–344 T/C) polymorphism may serve as a useful prognostic marker for risk stratification and personalization of therapy in CHF patients with cardiorenal syndrome.
Keywords: Cardiorenal syndrome, СHF, CYP11B2 polymorphism, Aldosterone synthase gene, TGF-β1, Genetic polymorphism, Fibrosis biomarkers (collagen IV, TGF-β1), Renal dysfunction, Renin–angiotensin–aldosterone system, Mineralocorticoid receptor antagonists, SGLT2 inhibitors
Cite this paper: Tosheva Kh. B., Gadayev A. G., Boboev A. T., Turakulov R. I., Comparative Analysis of Treatment Outcomes in Patients According to CYP11B2 (–344 T/C) Polymorphism Genotypes, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 105-110. doi: 10.5923/j.ajmms.20261601.25.
Article Outline
1. Introduction
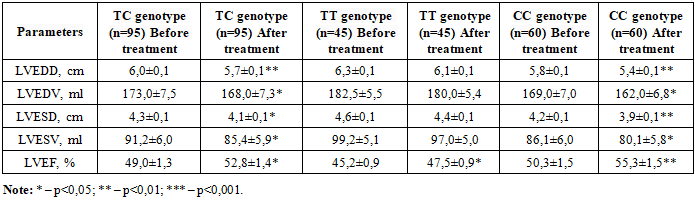
- Chronic heart failure (CHF) is a major global health problem associated with high morbidity, mortality, and healthcare costs [6,11,21]. Cardiorenal syndrome (CRS), characterized by the bidirectional interaction between cardiac and renal dysfunction, significantly worsens the prognosis in CHF patients [7,20]. The renin-angiotensin-aldosterone system (RAAS) plays a central role in the pathophysiology of both conditions [13,17]. Aldosterone, synthesized by aldosterone synthase (CYP11B2) in the adrenal cortex, is the key effector hormone of the RAAS [10,12]. Beyond its classical effects on sodium retention, aldosterone promotes cardiac and renal fibrosis, endothelial dysfunction, and inflammation [4,18]. Elevated aldosterone levels are associated with increased cardiovascular events and accelerated decline in renal function [11,15,16]. Aldosterone breakthrough during ACEI/ARB therapy remains a significant clinical challenge [13].The CYP11B2 gene –344 T/C polymorphism (rs1799998) directly influences aldosterone synthase transcription and aldosterone production [10,12,19]. The T allele has been consistently associated with higher aldosterone levels, elevated blood pressure, reduced glomerular filtration rate, and worse cardiovascular outcomes [15,19,22]. Current guideline-directed medical therapy for CHF includes RAAS inhibitors, beta-blockers, and mineralocorticoid receptor antagonists (MRAs) [2,3,5,21]. MRAs have shown substantial benefits in reducing mortality and hospitalizations [2,3,8]. Novel therapeutic agents, including SGLT2 inhibitors and nonsteroidal MRAs, have demonstrated additional cardioprotective and nephroprotective effects [1,6,7]. However, individual response to these therapies varies considerably among patients.Genetic factors may significantly influence treatment response in CHF patients [9,14]. Given that CYP11B2 polymorphism affects aldosterone levels and RAAS activity, it may also modulate the efficacy of aldosterone-targeted therapies [19,22]. Understanding the relationship between CYP11B2 genotype and treatment outcomes could enable personalized therapeutic approaches and improve clinical results [11,16,18,20]. The aim of this study was to perform a comparative analysis of treatment outcomes in CHF patients with cardiorenal syndrome according to CYP11B2 (–344 T/C) polymorphism genotypes, evaluating the efficacy of standard therapy combined with SGLT2 inhibitor dapagliflozin.The patients included in the study were divided into three groups according to the distribution of CYP11B2 (–344 T/C) aldosterone synthase gene polymorphism genotypes: group 1 – carriers of the TC genotype (n=95), group 2 – carriers of the TT genotype (n=45), and group 3 – carriers of the CC genotype (n=60). In these groups, the efficacy of standard therapy was assessed after 6 months of prospective follow-up based on the analysis of clinical and functional status, quality of life, and central hemodynamic parameters (Figure 1).
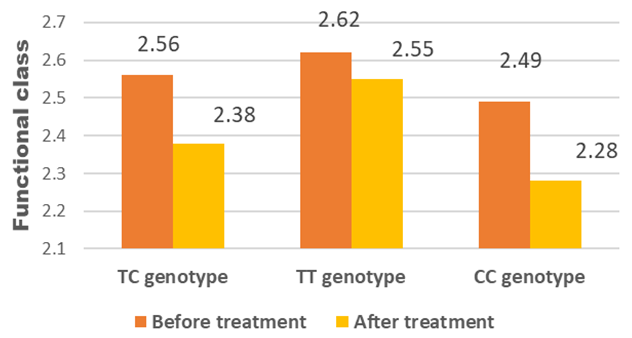 | Figure 1. Dynamics of functional classes in the observed patients |
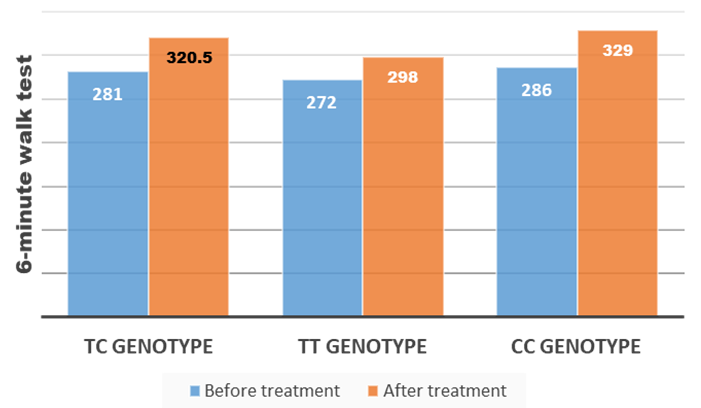 | Figure 2. Dynamics of exercise tolerance (meters) in the observed patients |
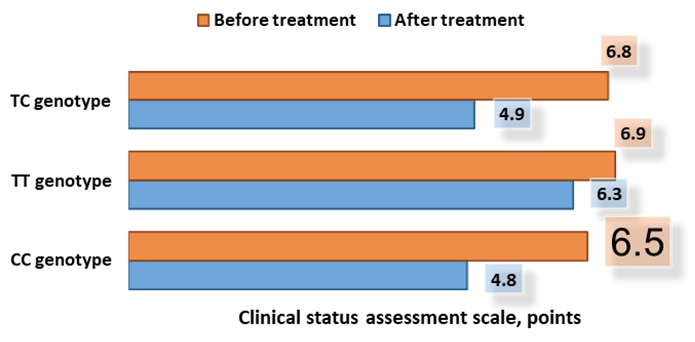 | Figure 3. Dynamics of clinical status (scores) in the observed patients |
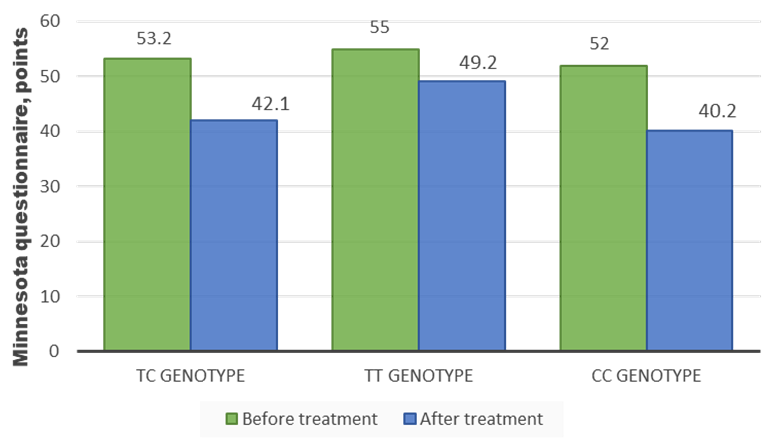 | Figure 4. Dynamics of quality-of-life scores before and after treatment in the observed patients (Minnesota questionnaire) |
|
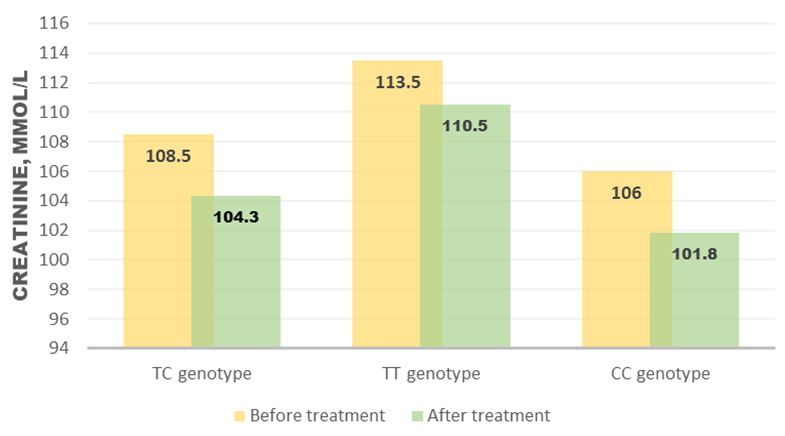 | Figure 5. Dynamics of serum creatinine according to aldosterone synthase gene –344 T/C polymorphism genotypes |
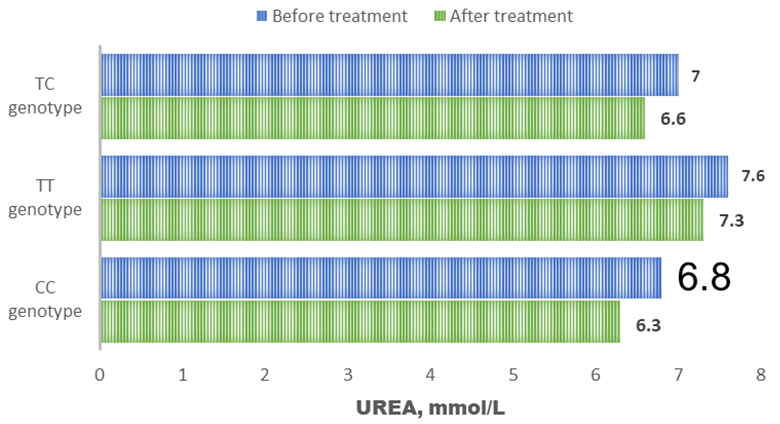 | Figure 6. Dynamics of serum urea under treatment |
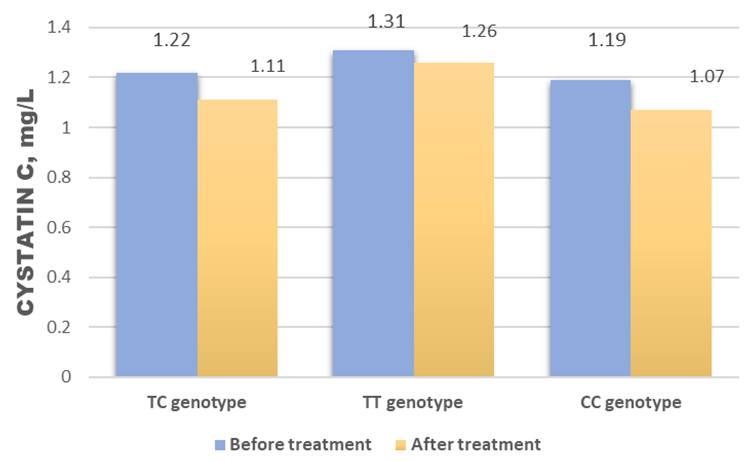 | Figure 7. Dynamics of serum cystatin C under treatment |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML