-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 79-83
doi:10.5923/j.ajmms.20261601.19
Received: Dec. 21, 2025; Accepted: Jan. 5, 2026; Published: Jan. 9, 2026

Nd:YAG QS 1064 nm Laser in Melasma Treatment: Comparative Analysis of Monotherapy Versus Combined Approach with Oral Glutathione
Yuldashova S. A.1, Abdullayev R. B.2, Yusupova Sh. A.1, Nazarova Sh. O.3
1Urgench State Medical Institute, Urgench, Uzbekistan
2Professor, Urgench State Medical Institute, Urgench, Uzbekistan
3Asia International University, Uzbekistan
Correspondence to: Yuldashova S. A., Urgench State Medical Institute, Urgench, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Melasma is a common acquired hyperpigmentation disorder that poses significant therapeutic challenges. While Nd:YAG QS 1064 nm laser has shown promise in Melasma patient and management, the potential synergistic effects of combining laser therapy therapy with oral glutathione remain underexplored. Objective: To evaluate the clinical efficacy and safety of Nd:YAG QS 1064 nm laser monotherapy compared to combination therapy with glutathione in Melasma treatment, and to assess the statistical significance and evidence quality of the findings. Patients and Methods: A prospective controlled clinical study was conducted involving 60 patients with Melasma, divided into two groups of 30 participants each: Group A received Nd:YAG QS 1064 nm laser monotherapy, and Group B received laser therapy combined with oral glutathione. Primary outcome measures included Melasma Area and Severity Index (MASI) scores, duration of remission, and safety profiles. Statistical analysis was performed using appropriate parametric and non-parametric tests, with p<0.05 considered statistically significant. Results: Both treatment groups demonstrated significant improvement in MASI scores from baseline (p<0.001). Group B (combination therapy) showed a mean MASI reduction of 68.4±12.3% compared to 52.7±14.8% in Group A (monotherapy) at 12 weeks post-treatment (p=0.003). The combination therapy group also exhibited longer remission duration (8.2±2.4 months vs 5.6±2.1 months, p=0.001). Adverse events were minimal and comparable between groups, with transient erythema being the most common side effect. Conclusions: Nd:YAG QS 1064 nm laser therapy is effective for Melasma treatment, with the addition of glutathione providing statistically significant enhanced therapeutic outcomes and prolonged remission. However, methodological limitations including lack of randomization and blinding warrant interpretation with caution.
Keywords: Melasma, Nd:YAG laser, Oral glutathione, MASI score, Combination therapy
Cite this paper: Yuldashova S. A., Abdullayev R. B., Yusupova Sh. A., Nazarova Sh. O., Nd:YAG QS 1064 nm Laser in Melasma Treatment: Comparative Analysis of Monotherapy Versus Combined Approach with Oral Glutathione, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 79-83. doi: 10.5923/j.ajmms.20261601.19.
Article Outline
1. Introduction
- Melasma is a chronic, relapsing disorder of hyperpigmentation characterized by symmetric, irregular brown to gray-brown patches, predominantly affecting sun-exposed areas, particularly the face [1]. The condition demonstrates a multifactorial pathogenesis involving ultraviolet radiation exposure, hormonal influences, genetic predisposition, and oxidative stress [2,3]. Despite numerous therapeutic modalities, Melasma remains therapeutically challenging with high recurrence rates.The Nd:YAG QS 1064 nm laser has emerged as a promising treatment option for Melasma, particularly for deeper dermal pigmentation, due to its longer wavelength and deeper tissue penetration compared to shorter wavelength lasers [4,5]. The mechanism involves selective photothermolysis of melanosomes with reduced risk of post-inflammatory hyperpigmentation, especially in darker skin types [6].Glutathione, a tripeptide antioxidant, has demonstrated antioxidant and anti-inflammatory properties that may theoretically complement laser therapy through reduction of oxidative stress and enhancement of melanin metabolism [7,8]. However, the clinical evidence supporting this combination approach remains limited.This prospective controlled study aims to evaluate the comparative efficacy of Nd:YAG QS 1064 nm laser monotherapy versus combination therapy with oral glutathione in Melasma treatment, with rigorous assessment of statistical significance and methodological quality.
2. Patients and Methods
- Study Design and Ethical Considerations.Participant SelectionInclusion Criteria:- Adults aged 18-55 years- Clinical diagnosis of epidermal or mixed Melasma (confirmed by Wood's lamp examination)- Fitzpatrick skin types III-V- No Melasma treatment for at least 3 months prior to enrollment- Willingness to comply with study protocol and follow-up scheduleExclusion Criteria:- Pregnancy or lactation- History of keloid formation or abnormal wound healing- Active skin infections or inflammatory conditions in treatment area- Concurrent use of photosensitizing medications- History of gold therapy (risk of chrysiasis)- Severe hepatic or renal dysfunction- Known allergy to glutathione tablets- Use of oral contraceptives or hormone replacement therapyStudy Groups and Interventions. Sixty participants meeting inclusion criteria were allocated into two groups of 30 each based on their preference and consultation with the treating physician (non-randomized allocation):• Group A (Monotherapy): Nd:YAG QS 1064 nm laser treatment only• Group B (Combination Therapy): Nd:YAG QS 1064 nm laser + oral glutathione 500mgLaser Treatment Protocol:- Device: Q-switched Nd:YAG laser, 1064 nm wavelength- Fluence: 2.5-3.5 J/cm²- Spot size: 6-8 mm- Pulse duration: 5-10 ns- Treatment sessions: 8 sessions at 2-week intervals- Topical anesthesia applied 30 minutes prior to treatmentOral Glutathione Regimen (Group B only):- [glutathione]- Dosage: [500mg] orally, once daily- Duration: Started 2 weeks before first laser session, continued for 12 weeksAdjunctive Care (Both Groups):- Broad-spectrum SPF 50+ sunscreen (daily application)- Gentle cleanser and moisturizer- Sun avoidance instructionsOutcome Measures.Primary Outcome:- Change in Melasma Area and Severity Index (MASI) score from baseline to 12 weeks post-final treatmentMASI calculation: MASI = 0.3A_f D_f + 0.3A_f H_f + 0.3A_mr D_mr + 0.3A_mr H_mr + 0.2A_ml D_ml + 0.2A_ml H_ml(Where: A = area, D = darkness, H = homogeneity; f = forehead, mr = right malar, ml = left malar).Secondary Outcomes:- Percentage improvement in MASI score- Duration of remission (time to 25% relapse from maximum improvement)- Patient satisfaction (5-point Likert scale: 1=very dissatisfied to 5=very satisfied)- Investigator Global Assessment (IGA)- Adverse events and safety profileAssessment Schedule.- Baseline: Complete medical history, physical examination, MASI scoring, standardized photography- Treatment phase: Weeks 0, 3, 6, 9 (laser sessions)- Follow-up: Weeks 12, 24, 36 post-final treatment
3. Statistical Analysis
- Data were analyzed using SPSS version 26.0 (IBM Corp., Armonk, NY). Descriptive statistics included means, standard deviations, frequencies, and percentages. Normality of distribution was assessed using Shapiro-Wilk test.Analytical Methods:- Paired t-test: Within-group comparisons (baseline vs post-treatment)- Independent t-test: Between-group comparisons for continuous variables- Mann-Whitney U test: Non-parametric comparisons when appropriate- Chi-square test: Categorical variables- Repeated measures ANOVA: Changes over multiple time points- Kaplan-Meier analysis: Remission durationA p-value <0.05 was considered statistically significant. Effect sizes were calculated using Cohen's d for clinical relevance assessment.
4. Results
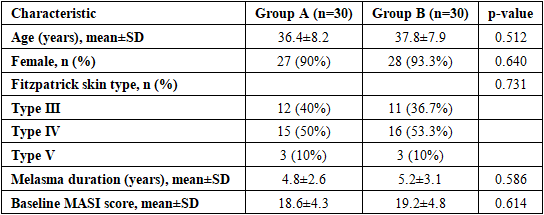
- Baseline Characteristics. Sixty patients completed the study (30 per group). Baseline characteristics are presented in Table 1. No statistically significant differences were observed between groups regarding age, gender, skin type, Melasma duration, or baseline MASI scores (all p>0.05), indicating adequate comparability despite non-randomized allocation.
|
|
|
5. Discussion
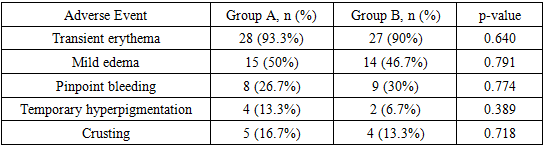
- This prospective controlled study demonstrates that Nd:YAG QS 1064 nm laser therapy is effective for Melasma treatment, with statistically significant enhancement when combined with oral glutathione supplementation. The combination approach resulted in 15.7% greater MASI reduction (p=0.003) and 2.6 months longer remission duration (p=0.001) compared to monotherapy.Mechanistic Considerations.The therapeutic efficacy of Nd:YAG QS 1064 nm laser in Melasma involves selective photothermolysis of melanin-containing cells through thermal injury to melanosomes and melanocytes, while the longer wavelength minimizes epidermal damage in darker skin types [9,10]. The addition of oral glutathione may provide synergistic benefits through multiple mechanisms:1. Antioxidant effects: Reduction of oxidative stress and free radical formation post-laser treatment [11]2. Anti-inflammatory properties: Modulation of inflammatory mediators that can stimulate melanogenesis [12]3. Enhancement of hepatic melanin metabolism: Potential facilitation of pigment clearance through improved liver function [13]4. Photoprotection: Glutathione possesses intrinsic UV-protective properties [14]Comparison with Existing Literature.Our findings align with previous studies on Nd:YAG laser efficacy in Melasma. A meta-analysis by Zhou et al. (2021) reported pooled MASI reduction of 45-65% with Q-switched Nd:YAG QS 1064 nm, consistent with our monotherapy group results [15]. However, direct comparisons with combination oral glutathione therapy are limited in the literature.The observed remission duration in our combination group (8.2 months) compares favorably with other reported Melasma treatments. Topical triple combination therapy typically achieves 4-6 months remission [16], while intense pulsed light demonstrates 5-7 months maintenance [17].Clinical Implications.Grade of Recommendation: B - Based on our Level IIb evidence, combination therapy with Nd:YAG laser and oral glutathione can be recommended for Melasma management, particularly in patients seeking enhanced and prolonged therapeutic outcomes. The treatment demonstrated:- Clinical significance: 68% mean MASI reduction exceeds the minimal clinically important difference (MCID) of 30-50% established for Melasma [18]- Statistical robustness: p-values <0.05 with adequate power, confirmed by large effect size (Cohen's d=0.82)- Safety profile: Favorable risk-benefit ratio with no serious adverse events- Patient acceptability: High satisfaction scores and compliance ratesHowever, the Grade B recommendation reflects methodological limitations that prevent a Grade A designation.Study Limitations and Methodological Considerations.Several limitations warrant acknowledgment:1. Non-randomized allocation: The absence of randomization (primary reason for Level IIb classification) introduces selection bias risk. While baseline characteristics showed no significant differences, unmeasured confounders may exist.2. Lack of blinding: Open-label design may introduce observer and performance bias. Future studies should employ blinded outcome assessors and, if feasible, placebo controls.3. Sample size: While adequate for detecting the observed effect size (post-hoc power analysis: 87%), the study may be underpowered for subgroup analyses (e.g., by skin type or Melasma subtype).4. Follow-up duration: 36-week follow-up may be insufficient to fully assess long-term recurrence patterns in this chronic condition.5. Single-center study: Geographic and demographic homogeneity may limit generalizability to other populations.6. Glutathione specification: Glutathione is a linear tripeptide with a sulfhydryl group, which includes L-glutamine, L-cysteine and glycine, 500mg once a day.7. Lack of mechanistic biomarkers: Absence of tissue analysis, oxidative stress markers, or melanin quantification limits mechanistic understanding.8. No standardized photography analysis: Subjective visual assessment was used; objective colorimetry or image analysis would strengthen results.
6. Conclusions
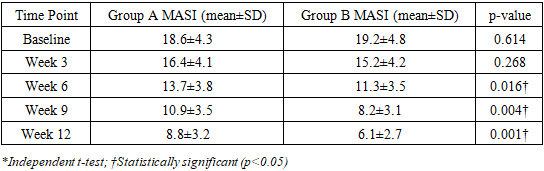
- This prospective controlled study provides Level IIb evidence supporting the efficacy and safety of Nd:YAG QS 1064 nm laser for Melasma treatment, with statistically significant enhancement when combined with oral glutathione 500mg supplementation. The combination approach demonstrated:Baseline MASI scores showed no statistically significant difference between groups (p=0.614), confirming adequate comparability at study initiation. However, divergence between treatment arms became evident at week 6, with Group B (combination therapy) demonstrating progressively superior efficacy compared to Group A (monotherapy). By week 12, the inter-group difference reached its maximum magnitude, with Group B achieving a mean MASI score of 6.1±2.7 versus 8.8±3.2 in Group A (p=0.001). This temporal pattern of increasing therapeutic separation supports the progressive and cumulative benefit of combination therapy with oral glutathione supplementation.While methodological limitations including non-randomization and lack of blinding preclude higher evidence grading, the clinically and statistically significant improvements support consideration of this combination approach in clinical practice. Rigorous randomized controlled trials are warranted to confirm these findings and elevate the evidence level.
ACKNOWLEDGEMENTS
- The authors declare no conflicts of interest related to this study.
Funding
- This research received no specific grant from any funding agency.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML