-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 75-78
doi:10.5923/j.ajmms.20261601.18
Received: Dec. 23, 2025; Accepted: Jan. 8, 2026; Published: Jan. 9, 2026

Metabolic and Inflammatory Laboratory Markers of Non-Alcoholic Fatty Liver Disease Severity
F. R. Mirzakarimova, A. S. Babadjanov, L. V. Kadomseva
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study was to evaluate the diagnostic and prognostic significance of laboratory markers of inflammation, cytolysis, cholestasis and metabolic disturbances in the development and progression of NAFLD depending on the degree of hepatic steatosis. The article presents the results of a comprehensive analysis of metabolic and inflammatory laboratory markers in patients with non-alcoholic fatty liver disease (NAFLD) depending on the degree of hepatic steatosis. Hemogram parameters, markers of systemic inflammation, cytolysis, cholestasis, lipid and carbohydrate metabolism were evaluated in patients with grade I and grade II steatosis compared with a control group. It was established that the progression of hepatic steatosis is accompanied by a significant increase in C-reactive protein levels, leukocyte count, ALT, AST and γ-glutamyltransferase activity, as well as the development of atherogenic dyslipidemia and signs of insulin resistance. Significant correlations were identified between the degree of hepatic steatosis and key laboratory parameters, confirming the role of systemic inflammation and metabolic disturbances in NAFLD progression. The obtained data emphasize the diagnostic and prognostic value of routine laboratory markers for non-invasive assessment of disease severity and risk stratification of NAFLD progression.
Keywords: Non-alcoholic fatty liver disease, Hepatic steatosis, Systemic inflammation, Metabolic disorders, Laboratory markers
Cite this paper: F. R. Mirzakarimova, A. S. Babadjanov, L. V. Kadomseva, Metabolic and Inflammatory Laboratory Markers of Non-Alcoholic Fatty Liver Disease Severity, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 75-78. doi: 10.5923/j.ajmms.20261601.18.
1. Introduction
- Non-alcoholic fatty liver disease (NAFLD) is one of the most common chronic liver disorders worldwide and represents an important problem of modern medicine due to its high prevalence and serious clinical consequences. Epidemiological data indicate that the prevalence of NAFLD among the adult population reaches approximately 25–30% and continues to increase in parallel with the global rise in obesity, metabolic syndrome and type 2 diabetes mellitus. NAFLD not only increases the risk of hepatocellular injury and progression to non-alcoholic steatohepatitis (NASH), fibrosis and cirrhosis, but is also associated with an increased risk of cardiovascular and metabolic complications, highlighting its significance as a multisystem disease [1].Despite the widespread use of instrumental methods such as ultrasound for detecting hepatic steatosis, laboratory markers remain crucial for diagnosis, risk stratification and prediction of disease progression. Biochemical parameters, including cytolysis enzymes, inflammatory markers and lipid metabolism indicators, reflect not only the degree of liver injury but also the underlying metabolic imbalance involved in NAFLD pathogenesis. The study and validation of such laboratory predictors are of clinical importance for early identification of patients at high risk of disease progression and optimization of monitoring and therapeutic strategies [2].The aim of the study was to evaluate the diagnostic and prognostic significance of laboratory markers of inflammation, cytolysis, cholestasis and metabolic disturbances in the development and progression of NAFLD depending on the degree of hepatic steatosis.
2. Material and Methods
- The study included patients with NAFLD who underwent examination and treatment in clinical settings. The diagnosis of NAFLD was established based on clinical and anamnestic data, laboratory findings and ultrasound examination of the liver, with exclusion of other causes of liver disease (alcoholic liver disease, viral hepatitis, autoimmune and drug-induced hepatopathies). All examined individuals were divided into groups according to the degree of hepatic steatosis: grade I and grade II. The control group consisted of apparently healthy individuals without ultrasound signs of hepatic steatosis and without clinical or laboratory evidence of liver disease.All patients underwent comprehensive laboratory evaluation, including complete blood count with assessment of hemogram parameters (hemoglobin level, leukocyte count, erythrocyte sedimentation rate), as well as biochemical blood analysis. The biochemical profile included alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), total bilirubin, C-reactive protein (CRP), lipid profile parameters (total cholesterol, low-density and high-density lipoproteins, triglycerides) and fasting glucose concentration. All laboratory tests were performed using standardized methods with certified reagent kits.Statistical analysis was carried out using applied statistical software packages. Quantitative data are presented as mean ± standard error of the mean (M ± m). Parametric and non-parametric statistical methods were used to assess intergroup differences depending on data distribution. Correlation analysis was performed using Pearson or Spearman correlation coefficients. Differences were considered statistically significant at p < 0.05.
3. Results
- Analysis of hemogram parameters revealed statistically significant differences between the study groups depending on the degree of hepatic steatosis. Patients with grade II steatosis had significantly higher hemoglobin levels compared with both grade I steatosis patients and the control group (p < 0.05), which may reflect changes in blood rheology and compensatory responses in the presence of pronounced metabolic disturbances. Leukocyte counts in patients with grade II steatosis were significantly higher than in controls, and erythrocyte sedimentation rate increased progressively with steatosis severity (p < 0.05), indicating the development of subclinical systemic inflammation.Biochemical analysis demonstrated progressive increases in inflammatory and cytolytic changes. CRP levels in grade I steatosis remained close to reference values, whereas in grade II steatosis they increased almost threefold compared with controls (p < 0.017), indicating activation of the systemic inflammatory response. The most pronounced changes were observed in liver transaminases: in patients with grade II steatosis, ALT activity increased almost fourfold and AST more than twofold compared with controls (p = 0.001). In contrast, ALT and AST values in grade I steatosis were mostly within normal ranges.Cholestasis markers also showed a tendency to increase with steatosis progression. ALP levels were significantly higher in grade II steatosis compared with controls (p = 0.044), while GGT activity increased more than fourfold (p = 0.032), indicating combined hepatocellular and biliary tract involvement. Total bilirubin levels did not differ significantly between groups (p > 0.05), confirming its low sensitivity as an early marker of hepatic steatosis.Lipid profile analysis revealed characteristic atherogenic dyslipidemia in NAFLD. With increasing steatosis severity, levels of total cholesterol, low-density lipoproteins and triglycerides increased, with the most pronounced changes observed in grade II steatosis. High-density lipoprotein levels were significantly lower in grade I steatosis compared with controls (p = 0.05), indicating a reduction in anti-atherogenic potential even at early stages of the disease. Fasting glucose levels showed a moderate but statistically significant increase in grade I steatosis (p = 0.001), reflecting the development of insulin resistance; in grade II steatosis, glycemic values remained comparable, possibly due to compensatory mechanisms or interindividual variability in metabolic response.Correlation analysis revealed strong associations between the degree of hepatic steatosis and key laboratory parameters. A high positive correlation was found between steatosis grade and ALT (r = 0.72–0.78; p < 0.001) and AST levels (r = 0.68–0.75; p < 0.001), confirming increased hepatocellular damage with progression of fatty infiltration. Significant positive correlations were also observed for GGT and ALP (p < 0.01), indicating an increasing cholestatic component in more severe steatosis. CRP levels positively correlated with steatosis grade (r = 0.60–0.68; p < 0.01), confirming the role of chronic low-grade inflammation in NAFLD pathogenesis (Table 1).
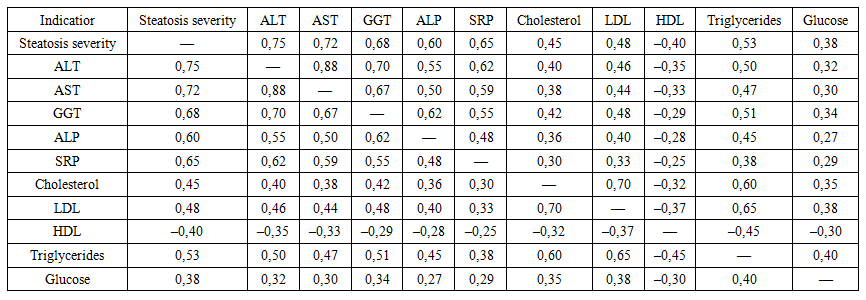 | Table 1. Correlation matrix between the degree of hepatic steatosis and key biochemical parameters |
4. Discussion
- In our study, markers of systemic inflammation—C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and leukocyte count—were significantly higher in patients with grade II steatosis than in those with grade I steatosis. This finding is consistent with current evidence on the role of low-grade inflammation in the pathogenesis of NAFLD. Numerous studies have demonstrated that elevated hs-CRP levels are closely associated with the presence and severity of NAFLD. For example, a cross-sectional study showed that the prevalence of NAFLD increases markedly with rising hs-CRP concentrations, and high levels of this marker remain an independent risk factor even after adjustment for confounders [3]. Similarly, our patients with more severe steatosis exhibited higher leukocyte counts, supporting the findings of Chao et al., who demonstrated a significant association between steatosis severity and leukocyte levels, with a stepwise increase in WBC counts as steatosis worsened (p < 0.001) [4].On the other hand, the impact of NAFLD on ESR is less consistent in the literature: some studies report minimal changes in ESR in NAFLD patients without overt inflammation, whereas ESR may increase with the development of non-alcoholic steatohepatitis (NASH). Nevertheless, the overall concept that “subclinical” inflammation accompanies NAFLD is supported by large cohort studies and clinical observations [5].Regarding cytolytic markers, patients with grade II steatosis in our study had significantly higher levels of transaminases—alanine aminotransferase (ALT) and aspartate aminotransferase (AST)—compared with grade I steatosis. This pattern is typical for NAFLD, as ALT elevation usually predominates over AST, reflecting a predominantly hepatocellular type of injury. Our findings are consistent with previous studies showing that the AST/ALT ratio decreases with NAFLD progression and correlates with steatosis severity and the presence of fibrosis [6]. For instance, a cross-sectional analysis of NHANES data (USA) demonstrated a positive association between the ALT/AST ratio and both NAFLD risk and the degree of hepatic steatosis [7]. Thus, increased transaminase levels, particularly ALT, reflect hepatocellular dysfunction and indicate a more active liver injury process in progressive NAFLD.Among cholestasis markers, γ-glutamyltransferase (GGT) and alkaline phosphatase (ALP) were assessed. In agreement with the literature, GGT levels were higher in patients with more severe steatosis. Recent studies suggest that serum GGT is not only a marker of chronic liver injury but also an indicator of metabolic syndrome, with NAFLD considered its hepatic manifestation. Even in the absence of overt cholestasis, moderately elevated GGT may indirectly indicate NAFLD and lipid metabolism disturbances [8]. A recent U.S. population-based study showed that an increased GGT/HDL-C ratio is independently associated with NAFLD risk and steatosis severity [7]. Accordingly, our findings support the prognostic value of GGT in NAFLD. In contrast, ALP levels showed less pronounced changes, which is consistent with reports indicating that ALP is not a key marker in non-alcoholic steatosis.The lipid profile in NAFLD patients was characterized by typical atherogenic dyslipidemia: patients with grade II steatosis more frequently exhibited elevated total cholesterol, LDL cholesterol and triglycerides, along with reduced HDL cholesterol compared with those with grade I steatosis. These results align well with published data showing increased TG and LDL cholesterol and decreased HDL cholesterol in NAFLD. Large population studies indicate that atherogenic dyslipidemia in NAFLD is associated not only with classical risk factors (obesity, insulin resistance) but also represents an independent risk signal. The predominance of LDL cholesterol and reduction of HDL cholesterol have been confirmed by other authors as well [9]. The correlations observed in our study between steatosis severity and worsening lipid profile further support the close relationship between NAFLD, metabolic syndrome and dyslipidemia.Glycemic levels (or comparable HbA1c values) were significantly higher in patients with grade II steatosis in our observations. This finding is consistent with the well-established association between NAFLD, insulin resistance and type 2 diabetes mellitus, as patients with impaired glucose metabolism are more likely to develop progressive hepatic steatosis. Studies show that even in non-diabetic individuals, elevated HbA1c correlates with an increased risk of NAFLD. For example, an Italian study demonstrated that each 1% increase in HbA1c approximately doubled the risk of NAFLD in individuals without diabetes, while higher HbA1c levels in diabetic patients were associated with a greater prevalence of advanced fibrosis [10]. Overall, our glycemic findings confirm the metabolic nature of NAFLD, indicating that elevated glucose and HbA1c levels may serve as markers of disease progression risk.Comparing our data with those of other authors, it can be concluded that our results generally confirm established patterns of biochemical changes in NAFLD. Elevated markers of inflammation and liver injury accompany the progression of NAFLD from simple steatosis to steatohepatitis. In particular, our observations of increased CRP and leukocyte counts in higher steatosis grades support the concept of chronic low-grade inflammation in NAFLD. Increased ALT/AST reflects ongoing hepatocellular injury and the activity of the cytolytic component of the disease, consistent with evidence on the prognostic value of the ALT/AST ratio for assessing steatosis and fibrosis severity. Elevated GGT levels also support the concept of hepatic metabolic overload and may serve as an additional marker of NAFLD. Finally, the presence of atherogenic dyslipidemia and hyperglycemia in patients with grade II steatosis corresponds to the known association between NAFLD, metabolic syndrome and increased cardiovascular risk.The diagnostic and prognostic significance of the evaluated biochemical markers lies in their use for stratifying the risk of NAFLD progression. Increases in transaminases, GGT and CRP may prompt further evaluation, such as calculation of non-invasive fibrosis indices (FIB-4, NAFLD fibrosis score), as these markers reflect inflammation and fibrogenesis in the liver. Atherogenic dyslipidemia and impaired glycemia should be considered when assessing cardiovascular risk and the need for metabolic correction. Our findings emphasize that even at the stage of simple steatosis, a set of laboratory abnormalities may already indicate an increased risk of progression to NASH and fibrosis. Therefore, regular monitoring of these parameters in patients with NAFLD enables identification of high-risk groups and timely modification of treatment and lifestyle.Overall, the obtained data are consistent with current publications and confirm the key roles of systemic inflammation (CRP, leukocytes), hepatocellular injury (ALT, AST), cholestasis (GGT) and metabolic disturbances (dyslipidemia, hyperglycemia) in the pathogenesis and progression of NAFLD. This suggests that a comprehensive assessment of biochemical parameters in combination with clinical evaluation is useful for risk stratification and prognostic assessment in patients with NAFLD.
5. Conclusions
- Progression of non-alcoholic fatty liver disease is accompanied by increasing systemic inflammation, hepatocellular injury and metabolic disturbances, manifested by elevated CRP, transaminases, γ-glutamyltransferase, atherogenic dyslipidemia and impaired glucose metabolism. The identified laboratory markers correlate significantly with the degree of hepatic steatosis and can be used for non-invasive assessment of NAFLD severity and stratification of progression risk.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML