-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 70-74
doi:10.5923/j.ajmms.20261601.17
Received: Dec. 23, 2025; Accepted: Jan. 7, 2026; Published: Jan. 9, 2026

Beyond Transaminases: Leptin and C-Peptide as Metabolic Markers of Steatosis Severity
F. R. Mirzakarimova, A. S. Babadjanov, L. V. Kadomseva
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study was to evaluate the diagnostic and prognostic significance of serum leptin and C-peptide levels depending on the degree of hepatic steatosis in patients with non-alcoholic fatty liver disease, as well as to determine their role in non-invasive risk stratification of disease progression. Non-alcoholic fatty liver disease (NAFLD) is a leading cause of chronic liver pathology worldwide, closely associated with obesity, insulin resistance, and metabolic syndrome. The identification of reliable non-invasive biomarkers for assessing steatosis severity remains a clinical challenge. This study aimed to evaluate the diagnostic and prognostic value of serum leptin and C-peptide levels in relation to the degree of hepatic steatosis in NAFLD. A comparative analysis was conducted in 60 patients with ultrasound-confirmed NAFLD (grades I–II steatosis) and 20 apparently healthy individuals. Serum leptin and C-peptide levels were measured using immunochemiluminescent assays. Metabolic parameters, transaminase activity, and body mass index were also assessed. Diagnostic performance was evaluated using ROC analysis. Both leptin and C-peptide levels increased significantly with steatosis severity. Leptin demonstrated high diagnostic accuracy for detecting moderate steatosis (AUC = 0.83), outperforming alanine and aspartate aminotransferases. Elevated C-peptide levels reflected enhanced endogenous insulin secretion and potential impairment of hepatic insulin clearance. A leptin-based nomogram incorporating body mass index enabled effective non-invasive risk stratification. The findings indicate that leptin and C-peptide are valuable non-invasive biomarkers for assessing NAFLD severity and may enhance clinical risk stratification in routine practice.
Keywords: Non-alcoholic fatty liver disease, Leptin, C-peptide, Steatosis, Non-invasive diagnostics
Cite this paper: F. R. Mirzakarimova, A. S. Babadjanov, L. V. Kadomseva, Beyond Transaminases: Leptin and C-Peptide as Metabolic Markers of Steatosis Severity, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 70-74. doi: 10.5923/j.ajmms.20261601.17.
1. Introduction
- Non-alcoholic fatty liver disease (NAFLD) is one of the most common forms of chronic liver disease worldwide and is directly associated with the epidemic of obesity, metabolic syndrome, and insulin resistance. Its prevalence continues to increase, and the disease is now regarded as a significant public health problem due to the risk of progression to non-alcoholic steatohepatitis, fibrosis, and liver cirrhosis. Traditional biochemical markers such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST) have low specificity for assessing the degree of steatosis, which stimulates the search for more sensitive and specific non-invasive biomarkers for the diagnosis and stratification of NAFLD [1].Disturbances in energy metabolism, adipokine dysregulation, and alterations in insulin metabolism play a key role in the pathogenesis of NAFLD. Leptin, a peptide hormone secreted by adipocytes, regulates energy balance and lipid metabolism, and its serum levels are usually elevated in obesity and hepatic steatosis, reflecting metabolic overload of adipose tissue [2]. C-peptide, as a product of proinsulin cleavage, serves as a stable marker of endogenous insulin secretion and correlates with insulin resistance, which is a key factor in the development of NAFLD [3]. Increased C-peptide levels in patients with NAFLD may reflect both enhanced β-cell insulin secretion and reduced clearance due to hepatic dysfunction.The aim of the study was to evaluate the diagnostic and prognostic significance of serum leptin and C-peptide levels depending on the degree of hepatic steatosis in patients with non-alcoholic fatty liver disease, as well as to determine their role in non-invasive risk stratification of disease progression.
2. Material and Methods
- The study included 60 patients with non-alcoholic fatty liver disease who were under observation and examination in a specialized inpatient setting, as well as 20 apparently healthy individuals who formed the control group. The diagnosis of NAFLD was established based on clinical and laboratory data, liver ultrasound findings, and exclusion of other causes of chronic liver disease (alcoholic liver disease, viral hepatitis, autoimmune and hereditary disorders). The degree of hepatic steatosis was assessed by ultrasound, with patients divided into groups with grade I and grade II steatosis.All participants underwent anthropometric assessment with calculation of body mass index (BMI), as well as laboratory testing including blood glucose levels, lipid profile (total cholesterol, triglycerides), and the activity of alanine aminotransferase (ALT) and aspartate aminotransferase (AST). Serum leptin and C-peptide levels were measured using immunochemiluminescent assays with standard commercial test kits in accordance with the manufacturer’s instructions.Statistical analysis was performed using a statistical software package. Quantitative variables are presented as mean ± standard deviation (M ± SD). Intergroup differences were assessed using parametric or non-parametric tests depending on data distribution. Correlation analysis was performed using Pearson’s correlation coefficient. The diagnostic value of leptin, ALT, and AST in detecting grade I–II hepatic steatosis was evaluated using ROC analysis with calculation of the area under the curve (AUC), sensitivity, specificity, positive predictive value, and negative predictive value. Differences were considered statistically significant at p < 0.05.
3. Results
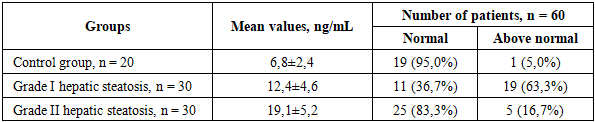
- The study demonstrated that patients with NAFLD showed a significant increase in C-peptide and leptin levels with increasing severity of hepatic steatosis. Serum C-peptide concentration was significantly higher in patients with grade II steatosis compared with the control group and patients with grade I steatosis, with maximum values exceeding control levels by more than fivefold. The degree of C-peptide elevation correlated with steatosis severity, indicating enhanced endogenous insulin secretion and/or reduced insulin clearance as hepatic fat infiltration progressed (Fig. 1).
 | Figure 1. Blood levels of C-peptide and leptin in patients with NAFLD depending on the degree of hepatic steatosis |
|
|
 | Figure 2. Nomogram chart for stratification of leptin cutoff values in NAFLD |
4. Discussion
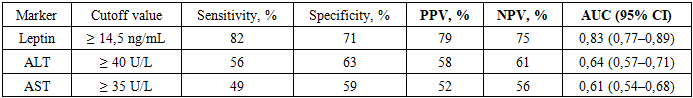
- In our study, leptin concentrations were significantly higher in patients with NAFLD compared with controls. These findings are consistent with the meta-analysis by Polyzos et al., in which a systematic review of 33 studies demonstrated that patients with both simple steatosis and NAFLD had higher leptin levels than healthy individuals, and that higher leptin concentrations were associated with disease severity [4]. Marques et al. also emphasized the diagnostic significance of leptin in NAFLD, reporting significantly higher leptin levels in NAFLD patients and an AUROC of approximately 0.83 for distinguishing patients from healthy controls [5]. Furthermore, leptin concentrations are known to increase with increasing severity of steatosis [6]. At the same time, in patients with NAFLD combined with type 2 diabetes mellitus, leptin levels may be somewhat lower than in patients without diabetes [5], which likely reflects modified metabolic and endocrine processes in combined pathology.Under physiological conditions, leptin secreted by adipocytes exerts anti-steatotic effects by stimulating β-oxidation of fatty acids and suppressing hepatic lipogenesis and gluconeogenesis, thereby preventing excessive fat accumulation and improving insulin sensitivity [7]. However, chronic hyperleptinemia, characteristic of obesity, leads to leptin resistance, resulting in the loss of its protective effects and failure to prevent steatosis [8]. Moreover, leptin has pronounced pro-inflammatory properties: it induces the synthesis of cytokines and chemokines in the liver and activates hepatic stellate cells, thereby promoting inflammation and fibrogenesis [9]. Thus, hyperleptinemia not only reflects adipose tissue overload but may also directly contribute to NAFLD pathogenesis by linking adipokine signaling with insulin resistance and inflammatory progression.Leptin levels are strongly influenced by sex and body mass index. Women physiologically have higher baseline leptin concentrations than men, which is confirmed by significantly lower values in male NAFLD cohorts. We observed a direct correlation between BMI and leptin (p < 0.0001); however, leptin remained an informative marker of NAFLD regardless of obesity stratification. Even at high BMI values, leptin reliably distinguished NAFLD patients from healthy individuals with an AUROC of approximately 0.83. Population and genetic factors (race, ethnicity, polymorphisms), as well as comorbid conditions, may further influence leptin levels. For example, in NAFLD patients with type 2 diabetes mellitus, leptin levels were significantly lower than in non-diabetic patients [5], possibly due to differences in fat distribution and pharmacological treatment. Taken together, these data emphasize that sex, BMI, and metabolic disturbances should be considered when interpreting leptin levels.C-peptide is regarded as a reliable marker of endogenous insulin secretion: it is released by pancreatic β-cells in equimolar amounts with insulin but, unlike insulin, undergoes minimal hepatic degradation, allowing assessment of total β-cell activity. In our study, C-peptide levels were elevated in patients with NAFLD, consistent with published data. Atsawarungruangkit et al., analyzing the U.S. NHANES database, identified fasting C-peptide, along with waist circumference and ALT levels, as three of the most important predictors of NAFLD presence [10]. Similarly, a study by Han et al. demonstrated that higher fasting C-peptide levels in obese children were associated with an increased risk of NAFLD [11]. These findings indicate compensatory hypersecretion of insulin (and thus C-peptide) in NAFLD patients with pronounced insulin resistance. Therefore, measurement of C-peptide may expand the possibilities of non-invasive assessment of metabolic status and disease progression risk.The influence of hepatic function on insulin and C-peptide clearance should also be considered. Insulin undergoes substantial first-pass hepatic degradation, whereas C-peptide is primarily cleared by the kidneys [10]. In chronic liver disease, this results in reduced hepatic insulin extraction and a decrease in the C-peptide/insulin ratio. Classical studies have shown that in patients with chronic liver disease, both C-peptide and insulin levels increase after glucose loading, while the C-peptide/insulin ratio significantly decreases, reflecting reduced hepatic insulin clearance. In NAFLD, a similar trend is observed, although the reduction in the C-peptide/insulin ratio is less pronounced than in cirrhosis [12]. Therefore, when interpreting C-peptide levels in NAFLD patients, hepatic function should be taken into account, as elevated C-peptide may result not only from hypersecretion of insulin but also from impaired hepatic inactivation.Overall, our results confirm that leptin and C-peptide are involved in NAFLD pathogenesis through different but interrelated mechanisms. Leptin, as an adipokine, conveys information about fat mass and metabolic inflammation; hyperleptinemia reflects adipose tissue accumulation and associated insulin resistance. C-peptide signals the degree of β-cell activity and compensatory insulin secretion and indirectly reflects hepatic insulin metabolism. Both parameters may serve as additional non-invasive biomarkers: leptin aids in detecting steatosis and associated inflammation, while C-peptide helps assess disturbances in carbohydrate metabolism and the risk of disease progression. Our observations deepen the understanding of NAFLD molecular mechanisms and support further investigation of leptin and C-peptide in risk stratification and diagnosis of this disease.
5. Conclusions
- The obtained results confirm the significance of leptin and C-peptide as promising non-invasive markers for assessing the degree of hepatic steatosis in patients with NAFLD. Elevated leptin levels significantly correlate with steatosis severity and body mass index, and its diagnostic accuracy (AUC = 0.83) exceeds that of traditional biochemical markers. C-peptide, reflecting both insulin secretion and potential impairment of hepatic clearance, also shows increased levels with steatosis progression. Both markers have clinical value for risk stratification and can be used in the comprehensive assessment of NAFLD patients in outpatient practice.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML