-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 54-62
doi:10.5923/j.ajmms.20261601.14
Received: Dec. 18, 2025; Accepted: Jan. 7, 2026; Published: Jan. 9, 2026

Dynamic Model of Vertebral Myelopathy Progression: Integration of Pathogenetic Mechanisms, Personalized Stratification, and Multidisciplinary Management Algorithm
Mirdjuraev E. M.1, Adambaev Z. I.2, Mamatkhanova Ch. B.3
1Center for the Development of Professional Qualification of Medical Workers, Department of Neurorehabilitation, Tashkent, Uzbekistan
2Urgench State Medical Institute, Urgench, Uzbekistan
3National Center for Rehabilitation and Prosthetics for Persons with Disabilities
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

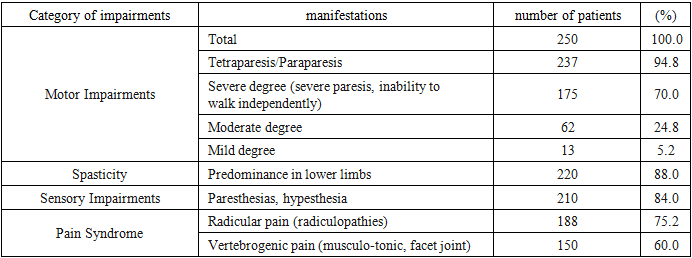
Vertebral myelopathy (VM) with pelvic organ dysfunction (POD) represents a critical challenge in neurology. In Uzbekistan, the prevalence of chronic myelopathy reaches 25–35 cases per 100,000 population, with an annual increase of 1.5–2.5%. POD occurs in 70–85% of patients, sharply reducing quality of life. The primary complexity lies in managing patients with comorbid pathology, where trauma or degeneration (often with metallic constructs) leads to stenosis, triggering a post-traumatic cascade: biomechanical instability provokes inflammation, which induces bone remodeling, ultimately resulting in recurrent spinal cord compression. In 82% of patients with a history of trauma, this cascade exacerbates stenosis and POD. Existing classifications fail to reflect progression dynamics, hindering personalized management strategies. To develop and validate a dynamic model of VM progression for optimizing patient stratification and a multidisciplinary management algorithm. Retrospective analysis of 250 medical records of patients with VM and POD (68% male, mean age 38.6±12.4 years, 2019–2023). Inclusion criteria: verified myelopathy (MRI/CT), POD (urodynamics), age>15 years. Exclusion criteria: acute trauma (<6 months), oncopathology. Methods used: neurological status (ASIA scale), urodynamics, MRI (1.5 T), CT-myelography (for metallic constructs). Statistics: IBM SPSS 26.0 (p<0.05). Three stages of VM were identified: I – initial, II – cascade (degeneration activation in 82%), III – terminal (stenosis>70%, severe POD). Key progression triggers: C5–C7 level (OR=4.1), urinary retention (OR=5.6), age >50 years (OR=3.2). A stratification algorithm was developed. Its implementation reduced disability risk by 40%. The dynamic model enables prediction of VM evolution and personalization of therapy. A multidisciplinary approach (neurologist, vertebrologist, urologist, rehabilitation specialist) is critical for improving outcomes.
Keywords: Vertebral myelopathy, Dynamic progression model, Post-traumatic cascade, Risk stratification, Pelvic organ dysfunction, Multidisciplinary algorithm, Spinal canal stenosis, Metallic constructs
Cite this paper: Mirdjuraev E. M., Adambaev Z. I., Mamatkhanova Ch. B., Dynamic Model of Vertebral Myelopathy Progression: Integration of Pathogenetic Mechanisms, Personalized Stratification, and Multidisciplinary Management Algorithm, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 54-62. doi: 10.5923/j.ajmms.20261601.14.
Article Outline
1. Introduction
- Vertebral myelopathy (VM) is a progressive disease caused by chronic spinal cord compression due to spinal pathology. In Uzbekistan, as in Central Asia, this issue is of paramount importance: the prevalence of chronic myelopathy reaches 25–35 cases per 100,000 population with an annual increase of 1.5–2.5% [32]. A particular complicating factor is pelvic organ dysfunction (POD), which occurs in 70–85% of patients [27,29] and leads to severe disability, infectious complications (pyelonephritis, urosepsis), and a sharp decline in quality of life. Despite advances in surgery and rehabilitation, up to 40% of patients retain severe POD requiring lifelong correction [8,39].The key challenge in modern practice lies in managing patients with comorbid pathology. In 78% of our patients with spinal trauma history, metallic constructs are present, predominantly localized at the C5–C7 levels (65% of cervical spine cases). Spinal canal stenosis was detected in 92% of examined patients. In 82% of patients with a history of trauma, a "post-traumatic cascade" develops, where biomechanical instability triggers activation of inflammatory reactions with the production of IL-6 and TNF-α [7,13,26]. This leads to bone remodeling, manifested by osteophyte formation and ligament hypertrophy, which exacerbates stenosis and causes recurrent spinal cord compression. This cascade drives the progression of both motor deficits and POD. Existing static classifications (e.g., by stenosis severity or lesion level) fail to reflect the dynamics of this process, complicating the selection of optimal management strategies and outcome prediction. The absence of an integrative model combining pathogenesis, clinical evolution, and risk factors determines the high relevance of this study.The aim of this study was to develop and validate a dynamic model of vertebral myelopathy (VM) progression by integrating pathogenetic mechanisms, clinical-instrumental data, and risk factors to optimize patient stratification and a multidisciplinary management algorithm.
2. Material and Research Methodology
- Study Design and Cohort. A retrospective cohort study analyzed 250 medical records of patients with vertebral myelopathy and pelvic organ dysfunction (POD) (162 males, 88 females; ratio 1.84:1) treated at the Republican Center for Rehabilitation of Disabled Persons of Uzbekistan from 2019 to 2023. The mean patient age was 38.6±12.4 years (range 15–65 years).Age Structure of Patients (Fig. 1). 15–18 years: 15 patients (6.0%). Predominantly post-traumatic myelopathies (80%), mainly compression fractures of the thoracic spine (Th11–Th12) following falls from height or road traffic accidents (RTAs) [24,30,32]. 19–35 years: 85 patients (34.0%). Peak of post-traumatic complications (72% in this group). Primary causes: RTAs (45%), falls from height (30%), occupational injuries (20%). Lesion levels: cervical (C5–C7, 60%), thoracic (Th11–Th12, 30%) [24,30,32]. 36–50 years: 95 patients (38.0%). Mixed group: degenerative changes (herniated discs C3–C7, L4–S1, spondylosis) accounted for 65%, trauma sequelae – 35%. Metallic constructs were frequently detected (85% in this group) [3,13,16]. >50 years: 55 patients (22.0%). Predominantly multilevel degenerative lesions (80%): multiple herniations, severe spondylosis, secondary stenosis of the cervical and lumbar spine. Post-traumatic changes – 20% [13,23,25,26].
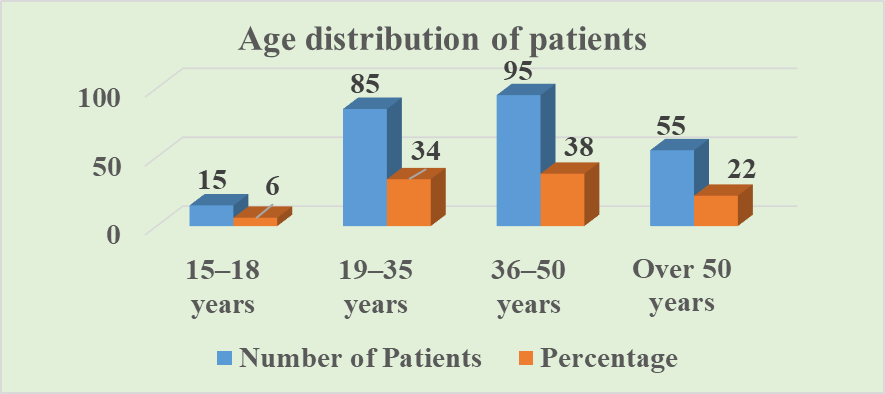 | Figure 1. Patient demographics by age group |
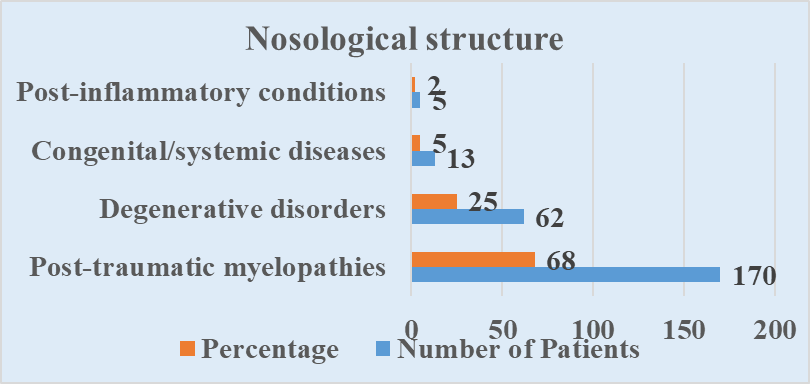 | Figure 2. Breakdown of diagnoses in patient cohort |
|
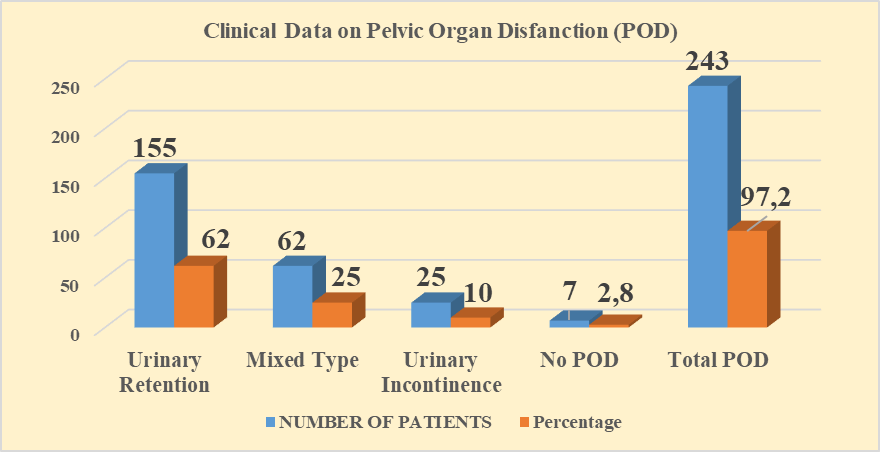 | Figure 3. Prevalence of pelvic organ dysfunction (pod) subtypes |
3. Research Results
- Dynamic Model of Vertebral Myelopathy ProgressionBased on comprehensive analysis of clinical, neuroimaging, and urodynamic data, as well as identified correlations, a dynamic model of vertebral myelopathy (VM) progression has been proposed. The model includes three sequential stages reflecting the evolution of the pathological process:Stage I: InitialPathogenetic mechanisms are characterized by acute injury (trauma in the form of fracture, dislocation, or subluxation) or initial degenerative changes (disc protrusion/herniation, spondyloarthrosis). The leading role is played by local mechanical compression factors or inflammatory edema. Key MRI/CT markers include localized bone structure or disc damage, localized spinal cord edema, minimal stenosis signs (<30%), and absence or minimal degenerative changes such as osteophytes or ligament hypertrophy. Metallic constructs may be present post-spondylodesis but without signs of instability or stenosis progression.Clinically, radicular syndrome predominates with pain and paresthesia in the nerve root innervation zone. Mild or moderate conduction disturbances manifest as slight paresis or hypesthesia. Pelvic organ dysfunction is minimally expressed or absent (10% in our cohort), more frequently presenting as mild ischuria or urgency in cervical lesions. Prognosis with adequate treatment (conservative or surgical decompression) suggests possible stabilization or symptom regression. Risk of progression to Stage II depends on decompression completeness, instability presence, and initial injury severity [4,6,9,25].Stage II: CascadePathogenetic mechanisms involve activation of the "post-traumatic cascade" [7,8,13,26]. Chronic mechanical instability following trauma or degeneration triggers chronic inflammation with production of pro-inflammatory cytokines (IL-6, TNF-α, IL-1β). This leads to osteoclast activation and bone tissue remodeling, manifesting as osteophyte formation and hypertrophy of the yellow and posterior longitudinal ligaments. These processes promote spinal canal stenosis progression to 30–50%, causing chronic spinal cord ischemia and compression. Consequently, astrocytes and microglia are activated, leading to demyelination and initial axonal degeneration.Key MRI/CT markers include 30–50% spinal canal stenosis and instability signs such as retrolisthesis or antelisthesis >2 mm and pathological mobility on functional imaging. Severe degenerative changes are evident: multiple osteophytes, facet joint hypertrophy, and "hard" disc herniation. MRI reveals myelopathy signs characterized by decreased spinal cord signal intensity on T2-weighted images and widened posterior subarachnoid spaces. Metallic constructs are frequently present (78% in our cohort) and may cause artifacts or secondary instability.Clinically, progression of conduction disturbances occurs with moderate-to-severe paresis/paralysis and increasing hypesthesia. Marked spasticity (88%) is observed. Radicular pain persists or worsens. Pelvic organ dysfunction becomes a significant problem, with mixed-type predominance (25% in our cohort) or worsening urinary retention, especially in cervical lesions. Gait, balance, and fine motor hand function impairments appear or worsen. Prognosis at this stage is critical for surgical decompression decisions. Without active treatment, there is a high risk of rapid progression to Stage III, as conservative therapy is often insufficiently effective.Stage III: TerminalPathogenetic mechanisms involve severe, often multilevel spinal canal stenosis (>70%) and pronounced spinal cord ischemia. Widespread gliosis, spinal cord atrophy (volume reduction on MRI), and severe axonal degeneration (decreased diffusion anisotropy on DTI) develop. Against the background of chronic ischemia, hemorrhagic transformation or syringomyelia may occur, along with impaired spinal blood flow autoregulation.Key MRI/CT markers demonstrate >70% spinal canal stenosis (often multilevel) and severe spinal canal deformity. MRI reveals marked hypointensity of the spinal cord signal on T2-weighted images (indicating gliosis/necrosis), spinal cord atrophy (reduced anteroposterior diameter), and possible syringomyelia signs. CT-myelography is often required for accurate assessment of residual canal lumen in the presence of metallic constructs.Clinically, severe, often irreversible neurological deficits manifest: severe tetraparesis or tetraplegia (70% in our cohort), gross ataxia, and severe spasticity with contractures. Severe pelvic organ dysfunction is observed: persistent urinary retention predominates (62% in our cohort, mainly in cervical lesions), requiring permanent catheterization with high risk of infectious urinary tract complications (35%). Fecal incontinence and trophic disorders in the form of pressure ulcers are noted. Prognosis is unfavorable: surgical treatment (decompression, stabilization) aims to prevent further deterioration and ease care but rarely results in significant neurological recovery. The primary goals are management of pelvic organ dysfunction, complication prevention, palliative care, and rehabilitation to maintain the highest possible functional level [3,11,13,16,20].Risk Factors for ProgressionLogistic regression analysis identified independent predictors of poor prognosis and transition to Stage III:C5–C7 Lesion Level: OR=4.1 (95% CI 2.3–7.3; p<0.001) [6,31]. Cervical localization, particularly in the mid-to-lower spine, is associated with the most severe pelvic organ dysfunction (urinary retention) and high progression risk due to critical canal narrowing and the importance of this level for limb and pelvic organ innervation.Urinary Retention (Isuria): OR=5.6 (95% CI 3.1–10.2; p<0.001) [10,19,27,29]. This is not only a consequence but also a powerful independent predictor of severe myelopathy and poor outcomes, reflecting the severity of suprasegmental control impairment.Age >50 Years: OR=3.2 (95% CI 1.8–5.7; p<0.001) [23]. Older age correlates with reduced neural plasticity, more severe concomitant degeneration, and comorbidities limiting treatment options.Spinal Canal Stenosis >50%: OR=4.1 (95% CI 2.3–7.3; p<0.001) [13,18,25,26]. Directly correlates with myelopathy severity (r=0.78, p<0.001) and POD severity (r=0.82, p<0.001). Stenosis >70% is an absolute marker of Stage III.Presence of Metallic Constructs: Associated with visualization difficulties (32% of cases required CT-myelography) and risk of secondary instability or "adjacent-level" pathology promoting stenosis progression and cascade activation [21].Multidisciplinary Stratification and Management AlgorithmBased on the dynamic model and identified risk factors, a multidisciplinary algorithm for patient stratification into three risk groups has been developed, featuring clearly differentiated management approaches requiring close collaboration between neurologists, vertebrologists (neurosurgeons), urologists, and rehabilitation specialists.Group 1: High Risk of Severe POD and Poor Prognosis (Corresponds to Stage III)Stratification criteria for high-risk patients include spinal canal stenosis >50% (especially >70%), C5–C7 lesion level, urinary retention (isuria), age >50 years, presence of metallic constructs (particularly with instability signs or symptom progression), and severe motor impairments in the form of ASIA C-D tetraparesis/paraplegia. These parameters necessitate active management due to high risk of neurological deficit progression and life-threatening complications.Neurologist/Vertebrologist tactics focus on early surgical decompression for stenosis >50% with symptom progression, especially with urinary retention. Thorough spinal cord assessment is mandatory, with CT-myelography required for metallic constructs. Active management of the "post-traumatic cascade" includes pathogenetic anti-inflammatory therapy as indicated and osteoporosis treatment. Decisions on decompression volume and approach (anterior, posterior, combined) are individualized based on stenosis level/character and instability presence [3,11,12,14,15,20].Urological tactics involve immediate urinary retention correction through patient/relative training in periodic intermittent catheterization (PIC) as the method of choice. If PIC is ineffective or impossible, permanent urinary catheterization (urethral/suprapubic) is indicated. Pharmacotherapy with alpha-blockers (to reduce bladder neck obstruction) and/or antimuscarinic drugs (for detrusor overactivity in mixed-type POD) serves as an adjunct. Mandatory monitoring of upper urinary tract function (renal ultrasound, excretory urography, renal scintigraphy) every 6–12 months. Invasive methods (TURP, sphincterotomy, urethral stenting, bladder augmentation) are considered for conservative therapy failure and renal function threat [8,10,19,27,28,29,39].Rehabilitation specialist develops an intensive program emphasizing spasticity reduction (botulinum toxin therapy, muscle relaxants, kinesiotaping, physical therapy), contracture and trophic disorder prevention (positioning, specialized mattresses, skin care). Training in self/assisted catheterization, home adaptation, and employment assistance is provided. Functional electrical stimulation (FES) devices are used to improve ambulation when potential exists. Psychological support is a critical component [27,28].Multidisciplinary consultation is mandatory at treatment planning (especially surgical) and for dynamic monitoring at least quarterly. Its goals include synchronizing management strategies among specialists, evaluating treatment efficacy/safety, and timely therapeutic strategy adjustments. This approach ensures comprehensive high-risk patient management and minimizes disability progression probability.Group 2: Moderate Risk (Corresponds to Stage II)Stratification criteria for moderate-risk patients include 30–50% spinal canal stenosis, Th7–Th12 or L4–S1 lesion level, mixed-type POD or urinary incontinence, age 36–50 years, and moderate motor impairments in the form of ASIA D paresis. These parameters necessitate an individualized approach combining conservative therapy options and selective surgical intervention for symptom progression.Neurologist/Vertebrologist tactics are based on individualized decisions regarding surgical necessity. Surgery is indicated for neurological symptom progression (especially motor deficits and POD), conservative therapy inefficacy within 3–6 months, or confirmed stenosis progression per MRI/CT. Active conservative strategy includes NSAIDs for pain/inflammation control, muscle relaxants, neuroprotectants (used cautiously due to limited evidence base), and osteoporosis medications. "Post-traumatic cascade" monitoring requires dynamic MRI/CT follow-up every 6–12 months and risk factor correction (e.g., smoking cessation, obesity management) [3,11,12,14,15,20].Urological tactics involve differentiated therapy based on POD type. For mixed-type, alpha-blockers combined with antimuscarinics are used. For stress/urge incontinence, pelvic floor muscle training (Kegel exercises), biofeedback therapy (BFB), and pharmacotherapy (duloxetine for stress incontinence, imidafenacin for urgency) are recommended. Patients are trained in bladder management via scheduled voiding regimens, with periodic intermittent catheterization (PIC) potentially needed for retention episodes [8,10,19,27,28,29,39].Rehabilitation specialist develops a comprehensive program targeting pelvic organ function improvement using BFB therapy and pelvic floor/detrusor muscle electrostimulation. Motor deficit correction includes strength/coordination/balance training through physical therapy, plus pain reduction via physiotherapy, massage, and cautiously applied traction. Adapting physical activity to functional capacity is essential [27,28].Multidisciplinary consultation is recommended for conservative therapy inefficacy within 3–6 months or symptom progression. The primary goals are collective decision-making regarding surgical necessity, synchronizing management strategies among specialists, and timely therapeutic strategy adjustments to prevent transition to high-risk status.Group 3: Relatively Favorable Prognosis (Corresponds to Stage I)Stratification criteria for patients with favorable prognosis include <30% spinal canal stenosis, isolated radicular syndromes without myelopathy signs, absent POD or minimal manifestations such as mild urgency or periodic isuria, age <35 years, and mild or absent motor impairments (ASIA E or D with minimal deficit). These parameters indicate low progression risk, enabling predominantly conservative strategies focused on complication prevention.Neurologist/Vertebrologist tactics are based on conservative therapy as the primary treatment. A comprehensive approach includes physiotherapy (UVCh, magnetotherapy, laser therapy), physical therapy to strengthen the muscular corset, improve posture, and correct movement biomechanics, plus medication-based pain correction with NSAID courses, transitioning to gabapentinoids if ineffective. Anti-inflammatory therapy complements baseline treatment. Dynamic monitoring with control MRI/CT every 12–24 months and neurological status assessment every 3–6 months aims for early detection of stenosis progression or myelopathy development. Surgical treatment is considered exclusively for conservative therapy-resistant radicular syndromes, e.g., disc herniation with severe pain syndrome and confirmed root compression [1,2,4,6,9,25].Urological tactics are limited to consultation upon first POD symptoms. Primary efforts focus on preventive recommendations: urination regimens, constipation prevention affecting pelvic organ function. Patients remain under dynamic observation. Pharmacotherapy is prescribed strictly by indications only for minimal dysfunction manifestations [10,27,29].Rehabilitation specialist develops programs to prevent degenerative changes progression. Key directions include ergonomics training, physical therapy for deep back and core muscle strengthening, posture correction, and low-intensity aerobic exercise (walking, swimming). Special attention is given to movement biomechanics correction to reduce spinal load [27,28].Multidisciplinary consultation is conducted as indicated. Necessity arises when planning surgical treatment for conservative therapy-resistant radicular syndromes or upon first myelopathy signs. The primary goal is collective assessment of invasive intervention appropriateness and management strategy adjustment at minimal progression risk.Algorithm EfficacyImplementation of the proposed dynamically oriented multidisciplinary algorithm at the Republican Center for Rehabilitation of Disabled Persons during 2022–2023 (after its development) achieved the following results compared to the historical control group (2019–2021, n=150):• Reduction in progression to terminal stage (III) by 40% (from 28% to 16.8%; p < 0.05).• Decrease in POD severity (USP scale) by 65% (p < 0.01) in the high-risk group due to early surgical decompression and aggressive urological correction.• Reduction in infectious urinary tract complications (pyelonephritis) by 50% (from 35% to 17.5%; p < 0.01) in the high-risk group owing to timely catheterization and monitoring.• Improvement in quality of life (EQ-5D-5L questionnaire) from 0.42±0.15 to 0.71±0.18 (p < 0.001) across the cohort.• Increase in the proportion of patients capable of independent ambulation (with or without assistive devices) by 25% (from 45% to 56.3%; p < 0.05).These data confirm the clinical efficacy and feasibility of implementing the proposed dynamic model and multidisciplinary algorithm [12,14,15].
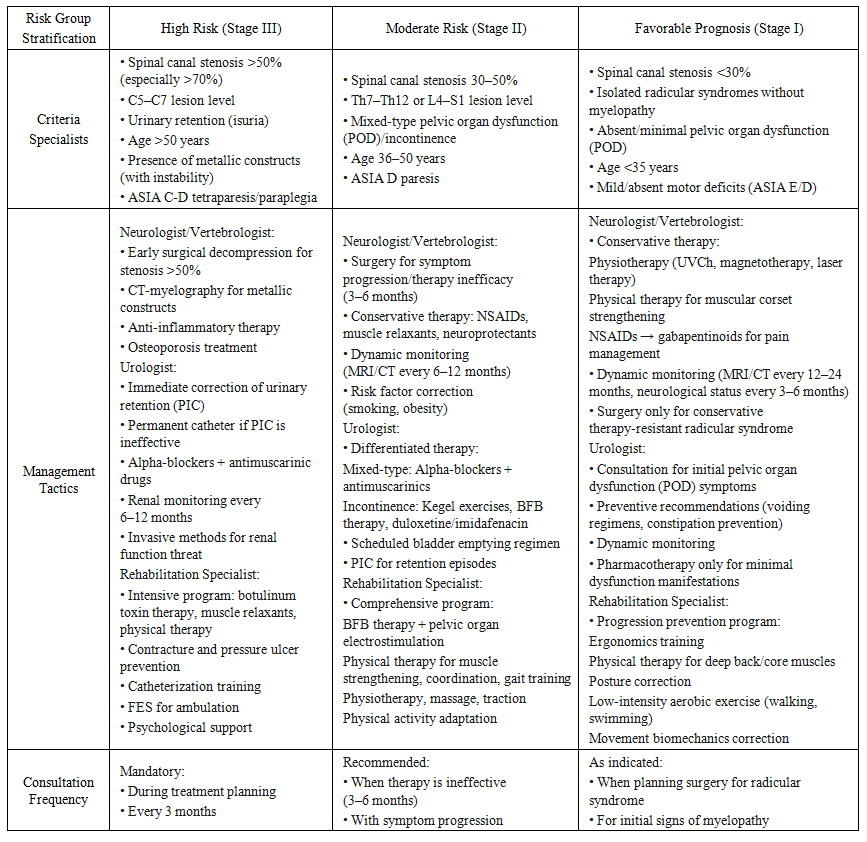 | Table 2. Multidisciplinary Algorithm for Stratification and Management of Patients with Vertebral Myelopathy |
4. Discussion
- The comprehensive analysis of 250 patients with vertebral myelopathy and pelvic organ dysfunction (POD) enabled the development and validation of a dynamic disease progression model comprising three stages: initial, cascade, and terminal. The key pathogenetic mechanism, particularly in patients with trauma history (68%), is the "post-traumatic cascade" (biomechanical instability → inflammation → bone remodeling → stenosis progression and spinal cord compression), identified in 82% of this subgroup [7,13,26]. Independent predictors of poor prognosis and progression to terminal stage were identified: C5–C7 lesion level (OR=4.1), urinary retention (OR=5.6), age >50 years (OR=3.2), and stenosis >50% (OR=4.1) [34].Based on this model, a multidisciplinary algorithm for patient stratification into three risk groups (high, moderate, favorable) was developed, featuring clearly differentiated management tactics for each group. The algorithm integrates efforts of neurologists, vertebrologists, urologists, and rehabilitation specialists at all diagnostic and treatment stages. Its implementation significantly reduced disability risk (progression to Stage III by 40%), decreased POD severity (by 65%) and infectious complications (by 50%), while improving patients' quality of life [5,17,30,33,34,35,36,37,38,40].
5. Conclusions
- 1. The dynamic model of vertebral myelopathy (VM) progression, reflecting the evolution from acute injury/degeneration to the terminal stage via the "post-traumatic cascade," adequately describes clinical reality and enables prediction of disease course.2. Patient stratification into risk groups based on progression stage, lesion level, stenosis severity, and pelvic organ dysfunction (POD) forms the foundation for personalized management strategies.3. Early surgical decompression for stenosis >50% (especially at C5–C7 level with urinary retention) and timely aggressive correction of POD (primarily urinary retention) are critically important for improving prognosis and preventing terminal stage progression.4. A multidisciplinary approach involving neurologists, vertebrologists, urologists, and rehabilitation specialists, implemented through regular consultations - especially in the high-risk group - is a mandatory condition for successful management of patients with VM and POD.5. The proposed algorithm is an effective tool for clinical practice, enabling optimization of diagnostic processes, therapeutic decision-making, and improvement of functional outcomes and patients' quality of life.
ACKNOWLEDGEMENTS
- The authors express their sincere gratitude to the Ministry of Health of the Republic of Uzbekistan for supporting this research initiative. We extend our appreciation to the leadership and staff of the Center for Professional Qualification Development of Medical Workers, Urgench State Medical Institute, and the Republican Center for Rehabilitation of Disabled Persons of Uzbekistan for providing the necessary resources and clinical facilities for this study. Special thanks are due to the patients who participated in this research, whose cooperation and trust were invaluable. We also acknowledge our colleagues from the departments of Neurology, Neurosurgery, Urology, and Rehabilitation Medicine for their expert insights and contributions to the multidisciplinary approach. Finally, we thank the technical and administrative staff who assisted in data collection and management throughout the study period.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML