-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(1): 50-53
doi:10.5923/j.ajmms.20261601.13
Received: Dec. 15, 2025; Accepted: Jan. 3, 2026; Published: Jan. 8, 2026

Morphofunctional Features of the Thymus of White Rats Depending on Age Periods in Alimentary Selenium Deficiency
Turdiyev M. R., Teshayev Sh. J., Sokhibova Z. R.
Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this article, the morphofunctional state of the thymus of white rats with alimentary selenium deficiency was studied depending on age. The study analyzed the histological structure, cellular composition, and functional activity of the thymus tissue of white outbred rats at the age of 6 and 9 months. The results showed that with alimentary selenium deficiency, a violation of parenchymal-stromal balance, a decrease in the morphometric parameters of lymphoid tissue, and a decrease in the number of lymphoid cells were revealed in this organ. These changes manifested as a decrease in the immune response and a weakening of the body's defense capabilities. Morphofunctional changes in the thymus in alimentary selenium deficiency manifest differently depending on the age periods of white rats, which contributes to a deeper understanding of the influence of alimentary factors on immune homeostasis.
Keywords: Alimentary insufficiency, Spleen, Thymus, Morphofunctional characteristics, White rat, Age periods, Immune system
Cite this paper: Turdiyev M. R., Teshayev Sh. J., Sokhibova Z. R., Morphofunctional Features of the Thymus of White Rats Depending on Age Periods in Alimentary Selenium Deficiency, American Journal of Medicine and Medical Sciences, Vol. 16 No. 1, 2026, pp. 50-53. doi: 10.5923/j.ajmms.20261601.13.
1. Actuality
- The study of the biological role of macro- and microelements is one of the most relevant areas of modern medicine. The rapid development of industry, the intensification of agriculture, and the introduction of new technologies necessitate the development of new methods for protecting the biosphere from anthropogenic impacts. A social approach to environmental and medical problems, improvement of methodological data and diagnostics, increases interest in studying the fundamental and practical aspects of the biological role of chemical elements in human life [5].Elements in their free state and in the form of many chemical compounds are part of all cells and tissues of the human body. Chemical elements are the most important catalysts of various biochemical reactions, important and irreplaceable participants in the processes of growth and development of the organism, metabolism, adaptation to changing environmental conditions [4].In recent years, scientific researchers have been increasingly interested in studying the role of selenium in the body. Selenium is one of the most important trace elements that support many biological functions in living organisms [3,8,13].Selenium is a microelement that is part of enzymes and plays an important role in the reproductive, endocrine, immune system, metabolism of the body, as well as in the activity of cell homeostasis and carcinogenesis [6,8,9,12].The thymus is the central organ of immunogenesis. It ensures the maturation and differentiation of T-lymphocytes not only within itself but also in peripheral immune organs, while simultaneously stimulating the integration of various populations of T-lymphocytes and macrophages for the implementation of immune responses [1,2,10].Despite many achievements in the study of the main pathogenetic mechanisms of the effects of selenium deficiency on the human body, there is insufficient data on changes in the structural composition of the organs of the immune system.The aim of the study is to assess the morphofunctional features of the thymus in white rats of different age periods with alimentary selenium deficiency.
2. Material and Research Methods
- The study was conducted on 80 white outbred male rats at the age of 6 and 9 months. In the experiments, the rules of ethics for the use of animals and the requirements of the Helsinki Congress were observed. Rats were kept under normal vivarium conditions. The age, sex, and weight of the experimental laboratory animals correspond to the nutritional conditions of the environment. Two groups of animals were created to determine the morphofunctional indicators of the thymus structure. Group I - normal (n=40); Group II - rats on a selenium-deficient diet (n=40). For the modeling of microelement deficiency, the German company "ALTROMIN Spezialfutter GmbH & Co." Special feed prepared by the firm "KG" was used. The feed was provided with a special official certificate No. 36/2024. The rats of the control group were fed regular feed 2 times a day. In the experimental group, 20 g of special feed was used twice a day, depending on body weight, for 24 weeks.White outbred rats of the control and experimental groups were excluded from the experiment and decapitated under ether anesthesia. The thymus was extracted by opening the rib cage. The parts of the thymus were fixed in 10% neutralized formalin, washed in running water for 2-4 hours, dehydrated in increasingly concentrated alcohols and chloroform, and paraffin blocks were prepared according to generally accepted methods. Paraffin blocks were cut 4-6 μm thick and stained with hematoxylin-eosin and Van Gieson. Morphometric examination of the structural structures of the thymus preparation was carried out using an ocular micrometer. The thickness of the capsule, the depth and diameter of the trabecula in different parts of the thymus, as well as the relative area of the lobes of the thymus, the cortex and medulla (relative to the total area of the incision), and the width of the cortex were measured. Measurements were carried out in five fields of view of each histological section. Fields of vision were selected randomly.To study the cells of the lymphoid structures of the thymus, the number of cells in the components of the thymus (subcapsular, cortical, and medullary) was counted under oil immersion using a NOVEL Model NLCD-307 (China) microscope. The number of cells was counted using a morphometric grid installed in the eyepiece of the microscope.Mathematical processing of histo- and cytomorphometric data obtained during the study was carried out directly using the Microsoft Office "Excel 7.0" application package of the Pentium IV personal computer.Statistical processing of the research data was carried out using variational parametric statistical methods using the Strelkov table (1986). The significance of the differences in the compared values was assessed using Student's t-test. Differences were considered statistically significant at p≤0.05.
3. Results and Analysis
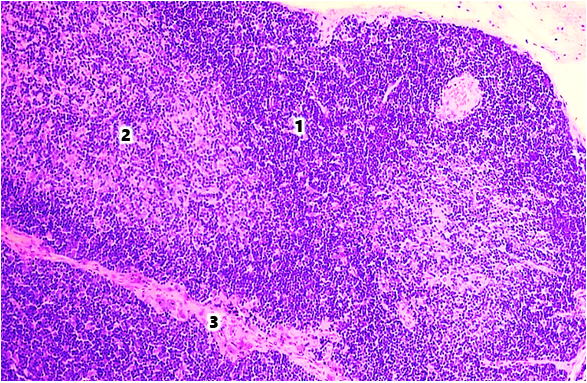
- The thymus of laboratory animals of the control group consists of two interconnected lobes, located in the lower third of the sternum mesentery. The thickness of the capsule in the area of the thymus gate in healthy white outbred rats at 6 and 9 months of age was 5.82±0.32 and 5.97±0.38 μm, respectively. The diameter of the trabecula in the proximal part was 13.36±0.27 and 13.52±0.22 μm, respectively, and in the distal part 10.28±0.14 and 10.43±0.22 μm. The area of the thymus lobules was 64.27±0.12% and 52.27±0.48%, respectively, by age periods.The thickness of the capsule in the thymus gate area of white outbred rats in the group with modeled selenium deficiency increased by 6.87% at 6 months of age and 7.54% at 9 months of age compared to the indicators of white rats in the control group. It was established that the diameter of the proximal and distal parts of the trabecula increases by 1.05 and 1.06 times, respectively, in both age periods. The area of the thymus lobules decreased by 6.1% at 6 months of age, by 7.0% at 9 months of age, and amounted to 60.57±0.32% and 48.83±0.26%, respectively.In the histological preparations of the tissues of the thymus of healthy white outbred rats, the cortical and medullary layers are distinguished. The boundary between the cortical and medullary layers is not clearly defined. Fragments with parenchyma replaced by adipose tissue are identified. In 6- and 9-month-old laboratory animals of the control group, the area of the cortical layer of the thymus was 63.86±0.37% and 58.83±0.26%, respectively, and the area of the medullary layer was 28.52±0.38% and 32.92±0.42%, respectively (Fig. 1).
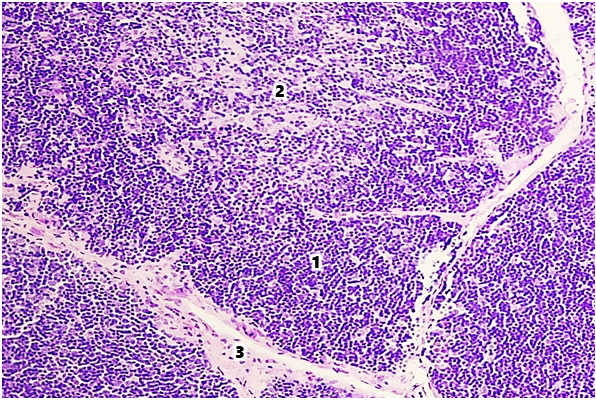 | Figure 1. Thymus of a 9-month-old white rat in the control group. Stained with hematoxylin-eosin. Ok. 10 x vol. 10. 1st cortex, 2nd medulla, 3rd trabecula |
4. Conclusions
- In selenium deficiency, an increase in the amount of connective tissue elements in the thymus was observed, which indicates the intensification of age-related involuntary processes in the organ in this micronutrient deficiency. A decrease in the relative area of the cortex and medulla of the thymus, the cortical-medullary index, the thickness of the cortex, and the number of small, medium, and large lymphocytes in the components of the organ (subcapsular, cortical, and medullary) was revealed. This indicates a decrease in the immune processes associated with the thymus and the protective adaptive properties of the body.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML