-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4610-4615
doi:10.5923/j.ajmms.20251512.91
Received: Nov. 13, 2025; Accepted: Dec. 8, 2025; Published: Dec. 25, 2025

Comparative Characteristics of Combined Antihypertensive Drug Therapy in Pregnant Women with Uncontrolled Gestational Arterial Hypertension
M. A. Sadulloeva 1, F. A. Zakirova 2, R. R. Hazratkulov 3
1PhD Student, General Cardiology and Gastroenterology, Republican Specialized Scientific and Practical Medical Center of Cardiology, Tashkent, Uzbekistan
2MD, Dsc, Head of the Department of General Cardiology and Gastroenterology, Republican Specialized Scientific and Practical Medical Center of Cardiology, Tashkent, Uzbekistan
3MD, General Cardiology and Gastroenterology, Republican Specialized Scientific and Practical Medical Center of Cardiology, Tashkent, Uzbekistan
Correspondence to: M. A. Sadulloeva , PhD Student, General Cardiology and Gastroenterology, Republican Specialized Scientific and Practical Medical Center of Cardiology, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

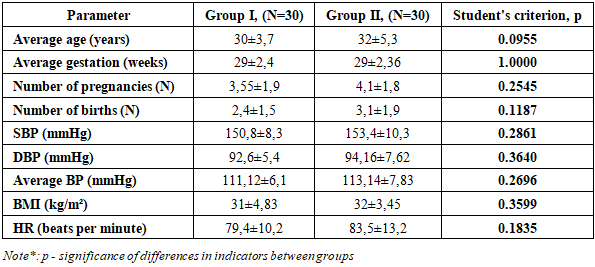
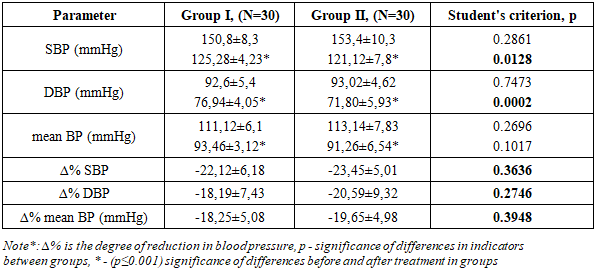
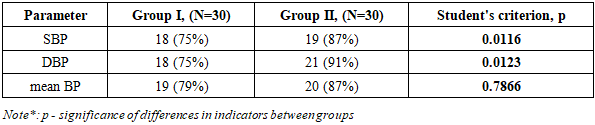
Objective: To evaluate the effectiveness of combination antihypertensive therapy on blood pressure parameters in pregnant women with uncontrolled hypertension. Methods and Results: The study included 60 pregnant women aged 20-24 weeks with a diagnosis of gestational hypertension according to the 2018 ESC Guidelines for the Management of Cardiovascular Diseases during Pregnancy. The women were randomly divided into two groups based on their combination antihypertensive therapy (n=60): Group 1 (n=30) received methyldopa (500-1000 mg) + metoprolol (50 mg), and Group 2 (n=30) received methyldopa (500-1000 mg) + amlodipine (5 mg). Titration of drug doses and monitoring of blood pressure indicators were carried out at 26-28, 30-32, 34-36 and 36-38 weeks of pregnancy. All patients underwent fetal Doppler ultrasonography, signed informed consent, office pressure was measured and ABPM was performed before the prescription of antihypertensive therapy. Analysis of office blood pressure indicators in both groups showed a significant decrease in SBP, DBP and mean BP (p=0.0001). However, the degree of reduction (Δ%) for SBP (p=0.0116) and DBP was statistically higher in Group II, especially for SBP and DBP (p=0.0123), which is probably explained by the vasodilatory effect of amlodipine. But this indicator for mean BP was not significant (p=0.3454), probably due to similar hemodynamic load of both regimens. Target SBP levels were achieved in 75% of patients in the first group and 87% of patients in the second group (χ2=0.98, p=0.32). Target DBP levels were achieved in 75% and 91% of patients, respectively (χ2=2.02, p=0.15), but no statistically significant difference was found between the groups. Conclusion: Both treatment regimens enabled the achievement of target blood pressure (BP) levels. It should be noted that although the number of patients in the second group who achieved target systolic (SBP) and diastolic (DBP) blood pressure was higher, this requires further study in a larger cohort of patients.
Keywords: Gestational hypertension, Pregnancy, Antihypertensive therapy, Target blood pressure
Cite this paper: M. A. Sadulloeva , F. A. Zakirova , R. R. Hazratkulov , Comparative Characteristics of Combined Antihypertensive Drug Therapy in Pregnant Women with Uncontrolled Gestational Arterial Hypertension, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4610-4615. doi: 10.5923/j.ajmms.20251512.91.
1. Introduction
- Hypertensive conditions during pregnancy and their complications remain the leading cause of death among women in developing countries, significantly impacting maternal and perinatal morbidity and mortality, underscoring the importance of timely identification of risk groups during the reproductive period. According to the World Health Organization (WHO), hypertensive disorders occur in approximately 5-10% of all pregnant women worldwide [1]. According to Hitti, J et al., of 7025 pregnant women with hypertensive disorders who delivered at the University of Washington Medical Center from 2013 to 2017, 284 (4%) had severe maternal morbidity; 154 had only a blood transfusion, 27 had other procedures, and 103 women had 149 diagnoses of severe maternal morbidity (26 women had multiple diagnoses). Severe preeclampsia occurred in 438 deliveries (6.2%). Notably, hypertension was associated with severe maternal morbidity in a dose-dependent manner, with the strongest association observed for preeclampsia with severe features (CI 3.9–7.3) [2]. A significant contribution to changing the threshold values for initiating drug therapy during pregnancy was made by Magee L.A., von Dadelszen P., Abalos E., and colleagues. The key studies CHIPS (Control of Hypertension in Pregnancy Study) [3] and CHAP Trial (Chronic Hypertension and Pregnancy Study) [4] completely changed the approach to the introduction of drug therapy in pregnant women with hypertensive conditions, and subsequently formed the basis for clinical recommendations of the ESC (European Society of Cardiology) [6] and ACOG (American College of Obstetricians and Gynecologists) [5]. A Cochrane review on antihypertensive therapy during pregnancy (Abalos E. et al.) summarized the results of 63 randomized trials that included more than 5900 pregnant women with mild to moderate arterial hypertension (SBP 140–159 mmHg, DBP 90–109 mmHg) to determine the efficacy and safety of antihypertensive drugs. The results showed that AHT significantly reduces the risk of developing severe hypertension by approximately half (RR ≈ 0.49; 95% CI: 0.40–0.60), secondary results showed that beta-blockers and calcium channel blockers combined were more effective than methyldopa in preventing episodes of severe hypertension (RR ≈ 0.70; 95% CI 0.56–0.88; 11 studies, 638 women) [7]. The study "Drugs for treating severe hypertension in pregnancy: a network meta-analysis and trial sequential analysis of randomized clinical trials" (Sridharan K, Sequeira R. 2018), which included a review of 51 randomized trials, compared the efficacy and safety of various antihypertensive drugs for severe hypertension in pregnant women. Nifedipine required fewer doses than hydralazine to achieve target blood pressure [8]. The study by Rosemol-George et al. aimed to compare the efficacy and safety of nifedipine with other antihypertensive medications in over 2,500 pregnant women and demonstrated faster achievement of target blood pressure compared with other antihypertensive medications [9]. Yin J. et al., which included 17 randomized controlled trials, compared the efficacy and safety of amlodipine and nifedipine in the treatment of hypertension in pregnant women. Amlodipine was not inferior to nifedipine and demonstrated a pronounced antihypertensive effect (RR = 1.06; 95% CI 1.01–1.10), no statistically significant differences in the incidence of adverse maternal and neonatal outcomes were found between the drugs [10]. Bajpai D. et al. examined key issues in the selection of antihypertensive therapy, citing current guidelines. Furthermore, the lack of evidence supporting the superiority of one drug over another is emphasized [11]. Many large studies are exploring the possibility of using single-agent medications, with subsequent dose adjustments to the maximum tolerated and safe doses for both mother and child. However, this approach does not always allow for rapid achievement of target blood pressure levels, given the limited time frame of pregnancy. The evidence base for combination antihypertensive therapy during pregnancy remains weak.
2. Materials and Methods
- According to the protocol of our prospective comparative study, 60 pregnant women aged 20–24 weeks gestation, diagnosed with gestational hypertension according to the 2018 ESC Guidelines for the Management of Cardiovascular Diseases during Pregnancy, were included.Exclusion criteria included any other form of gestational hypertension after 20 weeks of gestation, acute inflammatory processes or exacerbations of other chronic conditions, severe liver failure (Child-Pugh clinical classification B and C) and renal failure (CKD stages III-V), multiple pregnancies, mental disorders, use of antihypertensive medications at study entry, planned termination of pregnancy, intolerance to antihypertensive medications (methyldopa, metoprolol, amlodipine), known fatal or serious fetal abnormalities, other maternal or fetal systemic diseases, and failure of the woman to consent to inclusion in the study groups. All pregnant women underwent a physical examination, anamnestic data were collected, and blood pressure was measured using the Korotkoff method. To initiate and evaluate the effectiveness of the selected antihypertensive therapy, 24-hour blood pressure monitoring (ABPM) was used using a Cardiospy recorder (LABTechLTD, Hungary). Fetal well-being was assessed using fetal Doppler ultrasound before birth and the Apgar score after delivery. The study was conducted in accordance with the principles of the Declaration of Helsinki on the ethical conduct of clinical trials. Patients were informed, and written consent was obtained.Pregnant women were stratified into two groups based on the type of antihypertensive therapy prescribed: Group 1 (n=30) received methyldopa (500-1000 mg) + metoprolol (50 mg), and Group 2 (n=30) received methyldopa (500-1000 mg) + amlodipine (5 mg). Dynamic observation was carried out prospectively with control visits at 26–28, 30–32, 34–36 weeks, and retrospectively before delivery and in the postpartum period (6–8 weeks), which made it possible to evaluate the effectiveness and safety of the selection of AHT, the dynamics of blood pressure and pregnancy outcomes, until the target values of ∆% SBP, DBP and mean blood pressure were achieved.Statistical analysis was performed using Statistica 10 and a Microsoft Excel 2019 database. Prior to analysis, the data were tested for normal distribution using the Shapiro-Wilk test. Quantitative parameters are presented as mean (M) and standard deviation (SD). Comparisons of parameters between healthy pregnant women and patients with GHT (part one of the study) were performed using the Student's t-test for independent samples (for normally distributed data); the Pearson χ²-test; and the Mann-Whitney U-test (for non-normal distributions). When assessing the dynamics of quantitative parameters within a single group, the paired Student's t-test or the Wilcoxon test were used, depending on the nature of the data distribution. The critical level of statistical significance in all analyses was set at p < 0.05.
3. Results
- Before the study began, the groups were comparable in terms of age, parity and number of pregnancies, gestational age, and pretreatment systolic and diastolic blood pressure (SBP) levels (Table 1). All patients underwent fetal Doppler ultrasound and signed informed consent before initiation of antihypertensive therapy.
|
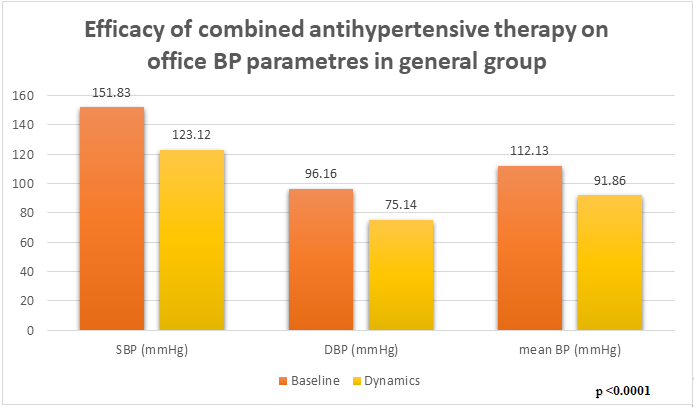 | Picture 1 |
|
|
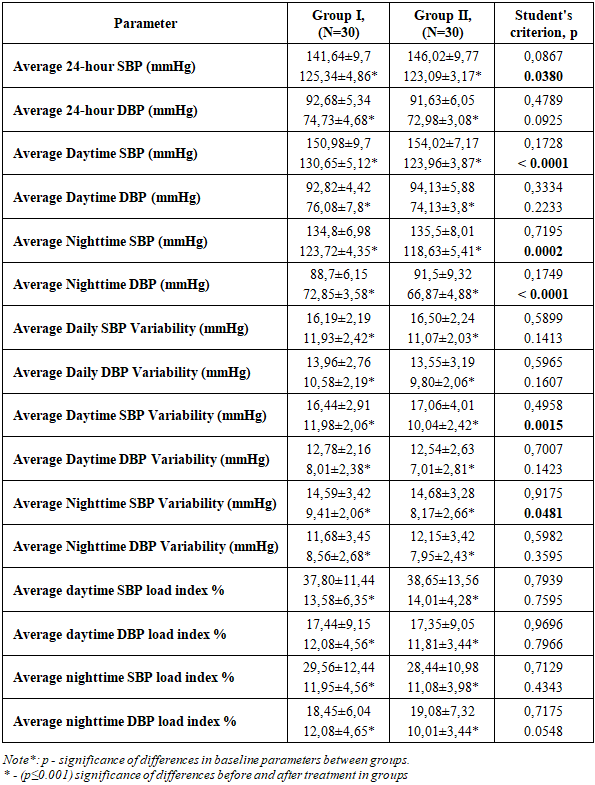
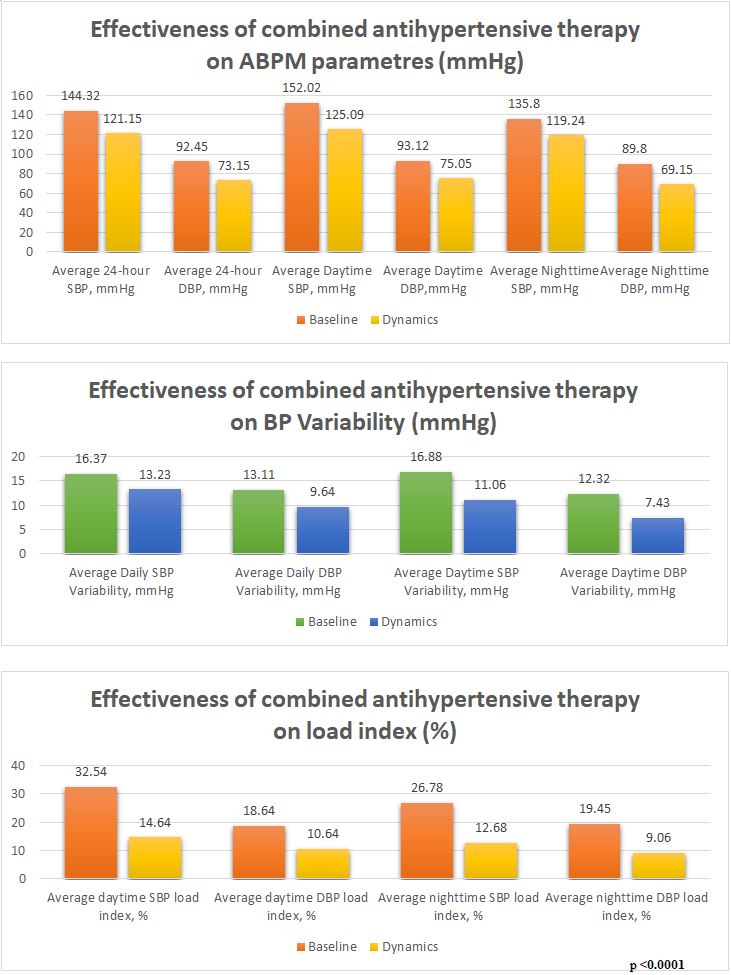 | Picture 2. Effectiveness of combined antihypertensive therapy on ABPM parameters in general group of 60 patients |
|
4. Discussion and Conclusions
- Our clinical experience with two combination therapy regimens, methyldopa/metoprolol succinate or methyldopa/amlodipine, is as follows: both treatment groups demonstrated high antihypertensive efficacy in pregnant women with poorly controlled hypertension at high risk for complications. Both treatment regimens enabled many patients to achieve target blood pressure (BP). Сomparing our findings with major randomized controlled trials such as CHIPS (Control of Hypertension in Pregnancy Study) and CHAP (Chronic Hypertension and Pregnancy trial), several important parallels emerge.The CHIPS trial showed that tight control of BP with a target DBP than 85 mmHg was safe and reduced the risk of hypertension complications compared with less tight control, moreover did not affect perinatal outcomes. In our study both combination regimens gave BP reductions and did not show any negative trends in fetal wellbeing indicators. The results support the concept that properly adjusted therapy, during pregnancy works well and stays safe [3]. The CHAP trial added proof by showing that treating chronic hypertension during pregnancy to a target BP <140/90 mmHg cut the rate of preeclampsia with severe features, preterm birth and maternal complications. Our findings are consistent with CHAP, demonstrating that achieving guideline-recommended BP goals—even with combination therapy—provides tangible clinical benefit and may decrease the likelihood of hypertensive complications [5]. It should be noted that although the second group had a higher number of patients achieving target systolic (SBP) and diastolic (DBP) blood pressure, further study in a larger cohort of patients is warranted. Overall, it can be concluded that initial therapy with a low-dose combination of antihypertensive drugs (AHD) may be more effective than a high-dose combination of a single drug, but requires more careful monitoring of maternal and fetal vital signs.Based on data from numerous studies, the following promising areas can be identified in the selection of antihypertensive therapy for pregnant women with GHT: a personalized approach to therapy, choice between first-line medications, the need for additional research to determine the optimal antihypertensive therapy regimen in a timely manner, the importance of pregnancy planning to ensure the most effective, adequate, and safe treatment for both mother and fetus, and, most importantly, regional standardization to develop uniform national protocols that incorporate best practices and minimize differences in treatment approaches.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML