Mirzakarimov Bahromjon Khalimjonovich, Isakov Nuriddin Zukhriddinovich, Karimov Doniyorbek Karimovich
Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Mirzakarimov Bahromjon Khalimjonovich, Andijan State Medical Institute, Andijan, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
The role of connective tissue dysplasia (CTD) and oxyproline - a biochemical marker of collagen metabolism - in the pathogenesis of pectus excavatum (PEX) in children was investigated. A total of 124 pediatric patients diagnosed with PEX were evaluated using clinical, instrumental, and biochemical methods. The patients were divided into a main group (n=90; 72,5%) and a control group (n=34; 27,5%). In the main group, 14 patients (15.6%) with mild grade I deformity were managed conservatively with orthopedic exercises under observation, while children with severe grade III–IV deformities underwent Vacuum Bell therapy and modern surgical techniques (both with and without fixation devices). In the biochemical analysis, the daily urinary oxyproline concentration was measured using an Agilent 1260 Infinity II LC System HPLC chromatograph. The observed increase in oxyproline levels with advancing age and deformity severity indicates its potential as a reliable biomarker for assessing PEX severity, monitoring disease progression, and evaluating treatment effectiveness.
Keywords:
Oxyproline, Pectus excavatum, Connective tissue dysplasia, Biochemical biomarker, Pediatric surgery
Cite this paper: Mirzakarimov Bahromjon Khalimjonovich, Isakov Nuriddin Zukhriddinovich, Karimov Doniyorbek Karimovich, The Significance of Oxyproline in Determining the Severity of Pectus Excavatum Deformity, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4598-4601. doi: 10.5923/j.ajmms.20251512.88.
1. Introduction
Connective tissue dysplasia (CTD) in children is a complex multifactorial disorder that encompasses genetic, biochemical, and morphological mechanisms, and manifests through decreased structural stability of body tissues, biomechanical weakness of mesenchymal structures, and multisystem functional impairments [1,6,10,17]. Numerous epidemiological studies have demonstrated that the prevalence of CTD among children in the peripubertal and pubertal age groups ranges from 40% to 85% [3,4,11,12,16].Pectus excavatum is one of the most common congenital thoracic deformities encountered in pediatric surgery, and determining its severity, selecting an appropriate treatment strategy, and objectively assessing the patient’s condition during follow-up are of critical importance [5,6,7,9,14]. In addition to the depth of the anatomical defect and the degree of tissue deformation, the biochemical state of the connective tissue - particularly indicators of collagen metabolism - plays a major role. Along with the depth of the anatomical defect and the degree of tissue deformation, the biochemical state of the connective tissue - particularly indicators of collagen metabolism - plays a significant role. Among these indicators, oxyproline (hydroxyproline), the principal amino acid component of collagen, is regarded as a biochemical marker of tissue remodeling and destructive processes [5,8,13,15,17]. An increase in oxyproline concentration indicates an acceleration of collagen degradation, reflecting reduced structural stability of the connective tissue. Several studies in children with pectus excavatum have demonstrated that the progressive depression of the chest wall is associated not only with mechanical factors, such as external pressure or diaphragmatic activity, but also directly with systemic connective tissue weakness (connective tissue dysplasia) [1,2,9,14].Objective. To determine the significance of oxyproline in assessing the severity grades of pectus excavatum.
2. Research Material and Object
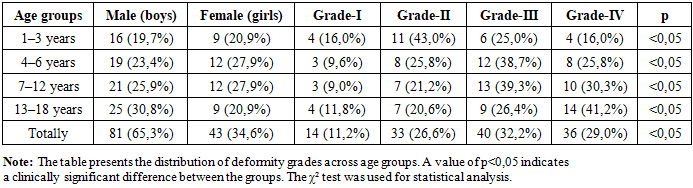
This study is based on the diagnostic and treatment outcomes of 124 pediatric patients with congenital pectus excavatum who were managed at the Department of General and Thoracic Surgery of the Andijan Regional Children’s Multidisciplinary Medical Center - the clinical base of Andijan State Medical Institute during the years 2020–2024. To evaluate the effectiveness of treatment methods, the patients were divided into two groups: a main group consisting of 90 children (72,5%) and a control group comprising 34 children (27,5%). Among the 90 patients in the main group, 14 children (15,6%) with grade I deformity were managed conservatively through supervised orthopedic physical exercises. No indications for surgical intervention were identified in these patients. Of the remaining 76 patients (84,6%) with more advanced deformity grades, 18 children (20,0%) underwent conservative treatment using the Vacuum Bell therapy method, while 58 children (64,4%) received surgical correction - with or without the use of internal fixation devices.Methods of Age Grouping and Severity Assessment. In classifying patients by age groups, we applied the “Standardized Age Groups” recommended by the World Health Organization (WHO), which represent the most widely used international age classification system today. The severity of pectus excavatum was assessed using the Haller-like Pectus Carinatum Index (HPCI, 2021). According to this index, deformity severity was categorized as follows: Grade I – mild deformity: PCI=1,00–1,05, Grade II – moderate deformity: PCI=1,06–1,20, Grade III – severe deformity: PCI=1,21–1,35, Grade IV – very severe, rigid deformity: PCI≥1,35. The distribution of patients by age groups and severity grades is presented below (Table 1).Table 1. Distribution of Children With Pectus Excavatum According to Age Group and Deformity Severity
 |
| |
|
Table-1 encompasses age groups, gender distribution, deformity grades, clinical trends, and statistical significance. These data demonstrate that the severity of pectus excavatum increases with age and was evaluated as statistically significant (p<0,05). In all age groups, the number of male patients remained higher than that of female patients (65,3% vs. 34,6%). This indicates that pectus excavatum is more frequently observed in boys; however, this difference is not considered statistically reliable. Nevertheless, clinical observations show that symptoms of pectus excavatum tend to manifest earlier in male patients. The results of the study demonstrated that the severity of connective tissue dysplasia (CTD) in children changes markedly with increasing age, revealing a clear tendency toward progression of the deformity. In early childhood, mild functional alterations - corresponding to grade I and grade II deformities - predominate. However, beginning from ages 3 to 6, due to increasing tissue fragility and greater mechanical load on the skeletal and joint systems, the incidence of grade III and grade IV deformities rises sharply. Between the ages of 6 and 12, the child’s period of rapid growth and the ongoing morphological remodeling of the musculoskeletal system lead to the highest peak in the occurrence of grade III deformities. During the 13–18-year age range, teenagers showed the highest prevalence of grade IV severe deformities (41,2%). This finding is associated with hormonal restructuring, metabolic imbalance of collagen and elastin tissues, and increased variability within the bone - ligament system, confirming a clinically significant intensification of dysplasia. Overall analysis showed that the combined proportion of grade III and grade IV deformities exceeded 61%, indicating that pectus excavatum tends to present in a more severe form in older children and teenagers. The progressive increase in deformity severity with age was also found to be statistically significant (p<0,05), demonstrating that this trend is not incidental but rather reflects the natural progression of the pathological process. Furthermore, the age-related escalation of deformity grades highlights the critical role of developmental factors in the pathogenesis of connective tissue dysplasia and underscores the importance of early diagnosis and regular dynamic monitoring. Assessing the morpho functional severity of pectus excavatum requires not only anatomical and radiological parameters but also an evaluation of the underlying biochemical mechanisms. Such a combined approach is essential for individualized decision-making, optimal treatment selection, and accurate prognosis for each patient.With this in mind, daily urinary oxyproline concentrations were measured in the study patients and used as a biochemical indicator of deformity severity. The oxyproline level in urine was determined using the Agilent 1260 Infinity II LC System chromatograph. This modular high-performance liquid chromatography (HPLC) platform is widely used for both analytical and preparative applications from routine quality control to complex scientific research - and provides reliable performance for standard HPLC as well as entry-level ultra-high-performance liquid chromatography (UHPLC). Based on the examination results, prophylactic measures were implemented during preoperative preparation and in the postoperative period to prevent recurrence.
3. Results and Discussion
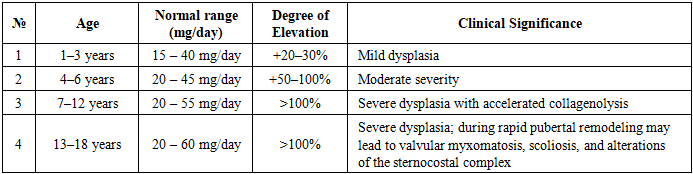
Connective tissue dysplasia (CTD) is considered one of the principal pathogenetic theories in the development of pectus excavatum, and an increase in urinary oxyproline levels is associated with tissue degradation, hyperactivity of metalloproteinases, reduced genetic stability, and oxidative stress. Oxyproline reliably reflects accelerated tissue remodeling and the progression of dysplasia severity. Therefore, in this study, oxyproline levels were used as a clinical and biochemical marker for assessing the severity of pectus excavatum. The daily fluctuation in urinary oxyproline levels in patients was evaluated based on the normative values described in [2] (Table 2).Table 2. Clinical Significance of Daily Variations and Elevation Levels of Oxyproline
 |
| |
|
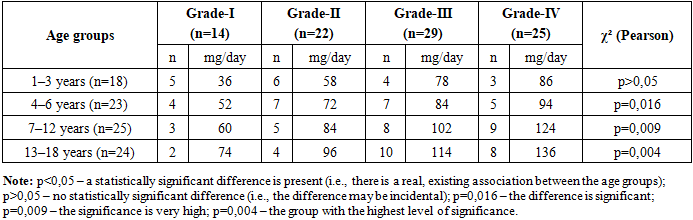
Collagen metabolism in the body varies with age. In children, due to the high activity of bone and connective tissue remodeling, the daily normative range of oxyproline is observed across a broader interval compared with adults. Hormonal changes during the pubertal period, the acceleration of skeletal growth, and increased physical activity contribute to physiological fluctuations in oxyproline levels.In children aged 1–3 years, the normal daily oxyproline level ranges from approximately 15–40 mg/day, and the biochemical changes observed during this period are primarily associated with physiological growth. Values exceeding the norm by 20–30% are characteristic of mild dysplasia, where tissue alterations are minimal. In children aged 4–6 years, the normal range is 20–45 mg/day. During this stage, a 50–100% increase in daily oxyproline indicates a markedly accelerated rate of collagenolysis and corresponds to clinical and biochemical manifestations of moderate dysplasia.In such children, signs including joint hypermobility, skin laxity, and weakness of the sternocostal complex are more commonly observed. In the 7–12-year age group, the normal daily oxyproline level ranges from 20–55 mg/day, and a rise of 100% or more is regarded as an indicator of severe dysplasia. Rapid collagen degradation may lead to systemic skeletal deformities and myxomatous changes in the cardiac valves. For this reason, orthopedic, cardiological, and biochemical monitoring intervals are shortened in these patients.In teenagers aged 13–18 years, the normal daily oxyproline range is 20–60 mg/day. Due to hormonal changes during puberty and remodeling processes occurring in the growth plates, an increase in oxyproline levels by 100% or more indicates a high-risk stage of connective tissue dysplasia. These teenagers may show rapid progression of scoliosis, thoracic deformities (pectus excavatum), and a higher frequency of mitral valve prolapse.When the daily urinary oxyproline levels were examined in 79 patients from the main study group, the following deviation indicators were observed according to the severity grades of pectus excavatum (Table 3).Table 3. Deviation of Daily Urinary Oxyproline Levels According to Deformity Severity in Children With Pectus Excavatum
 |
| |
|
Table 3 presents the severity grades of connective tissue dysplasia (CTD) across different age groups in children and the corresponding dynamics of oxyproline excretion in urine. As the data show, the concentration of oxyproline increases consistently and steadily in parallel with increasing deformity severity. In children aged 1–3 years, the number of cases with grade I and grade II dysplasia is relatively high, and oxyproline levels are recorded at comparatively low values (36–58 mg/day). In patients with grade III and grade IV dysplasia, this indicator rises to 78–86 mg/day. These differences observed at this age are explained by the early manifestations of dysplasia and the relatively low activity of tissue degradation processes. In the 4–6-year age group, dysplasia severity begins to increase markedly. Oxyproline levels, which range from 52–72 mg/day in grades I–II, rise to 84–94 mg/day in grades III–IV. In this age interval, accelerated collagenolysis is observed as a result of intensified skeletal and connective tissue growth and the activation of endogenous developmental factors.In children aged 7–12 years, oxyproline levels increase even further: 60–84 mg/day in grades I–II, and 102–124 mg/day in grades III–IV. This period corresponds to one of the most rapid phases of organ and skeletal system growth in children; therefore, biochemical markers of dysplasia appear with a pronounced elevation.The highest values were recorded in the 13–18-year group. Specifically, oxyproline levels reached 74 mg/day in grade I and up to 136 mg/day in patients with grade IV dysplasia.Hormonal changes during the teenage years, the rapid growth of the musculoskeletal system, and the structural fragility of connective tissue contribute to the progression of dysplasia and a further increase in oxyproline levels. According to the χ² (Pearson) analysis, age-related changes in oxyproline levels in connective tissue dysplasia become statistically significant beginning from the age of 4 years (p<0.05).In the 7–12 and 13–18 age groups, the highly significant p-values of 0.009 and 0.004 indicate a strong correlation between increasing dysplasia severity and elevated daily oxyproline levels. This confirms the scientific validity of using oxyproline as a sensitive biomarker for assessing the severity of pectus excavatum.Overall, the data presented in the table clearly demonstrate the role of collagen degradation in the pathogenesis of pectus excavatum. The gradual increase in oxyproline levels with advancing age and worsening dysplasia severity confirms its value as a clinically and prognostically significant biomarker. Using this biomarker allows for more accurate and reliable assessment of dysplasia severity, monitoring of disease dynamics, and selection of appropriate treatment strategies.An increase in the daily amount of oxyproline in urine above the age-related normal range serves as a biochemical indicator of collagen degradation in connective tissue dysplasia. A 20–30% elevation is observed in mild dysplasia, while a 50–100% increase is considered indicative of moderate dysplasia. When the level rises by 100% or more, severe forms of tissue dysplasia, accelerated collagenolysis, and a high risk of visceral pathologies are likely.In addition, the rapid variability of values during the teenage years is associated with hormonal and mechanical factors and requires regular biochemical monitoring. The dynamics of oxyproline levels serve as an important clinical criterion for evaluating therapeutic effectiveness, predicting outcomes, and determining the intervals for follow-up surveillance.
4. Conclusions
Oxyproline is a key biomarker in the pathogenesis of pectus excavatum in children, and its elevation indicates connective tissue weakness, rapid progression of the deformity, and the presence of severe morphological changes. Integrating clinical, instrumental, and biochemical data provides significant clinical value in the early detection of the deformity, appropriate selection of treatment methods, and improvement of postoperative prognosis.
References
| [1] | American Academy of Pediatrics. Hypermobility disorders and collagen evaluation in children. 2022. |
| [2] | Burtis C.A., Bruns D.E. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics. – 6th ed. – St. Louis: Elsevier, 2018. – 2234 p. |
| [3] | Byers P. H., Belmont J. Ehlers–Danlos syndrome: classification and diagnosis // GeneReviews. 2021. |
| [4] | Castori M. Ehlers–Danlos syndrome and hypermobility spectrum disorders in pediatrics // Current Opinion in Pediatrics. 2021. – 33(6). – P. 632–644. |
| [5] | Demirdas S., Yuen M., et al. Genetic basis of connective tissue fragility syndromes // Journal of Medical Genetics. 2020. – 57(5). |
| [6] | De Paepe A., Malfait F. Hereditary connective tissue disorders: diagnosis and molecular basis // Nature Reviews Genetics. 2019. |
| [7] | European Society of Pediatric Endocrinology. Guidelines on collagen metabolism assessment in growing children. 2021. |
| [8] | Formenti A. M., Doga M., et al. Biochemical bone turnover markers in children and adolescents // Endocrine. 2019. – 63. – P. 349–358. |
| [9] | Gromyko A. I., Sokolov M. A. Oxidative stress and collagen degradation in children with dysplasia // Biomedicine. – 2022. – №1. |
| [10] | Khristenko Yu. N., Gubler E. V. Biochemistry of collagen and connective tissue. – Moscow: Medicine, 2020. – 312 p. |
| [11] | Kolesnikova N. V., Pichugin V. P. Dysplasia of connective tissue in children: clinical manifestations and diagnostic criteria // Pediatria. 2017. – №6. – С. 12–18. |
| [12] | Kovalenko V. N., Radchenko V. A. Undifferentiated connective tissue dysplasia in pediatric practice // Ukrainian Medical Journal. 2020. – №3. |
| [13] | Lukianenko N. S., Petritsa N. A., et al. Congenital malformations of the urinary system as visceral markers of connective tissue dysplasia // Postgraduate Medicine. 2022. – Issue 10. |
| [14] | Malfait F., Symoens S., De Paepe A. The genetics of connective tissue disorders // Clinical Genetics. 2020. – 97(1). – P. 32–54. |
| [15] | Martynov A. I., Nechaeva G. I. Clinical guidelines of the Russian Society of Physicians on diagnosis, treatment and rehabilitation of patients with connective tissue dysplasia (first revision) // Medical Bulletin of the North Caucasus. 2018; 13 (1–2): 1–73. |
| [16] | Nazarenko G. I. Biochemical markers of collagen metabolism in children // Laboratory diagnostics. 2019. №4. |
| [17] | Trzeciak H. I., Kwiatkowska M. Collagen metabolism disturbances in developmental disorders // Pediatria Polska. 2018. – 93(2). – P. 112–118. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML

