-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4578-4580
doi:10.5923/j.ajmms.20251512.82
Received: Nov. 22, 2025; Accepted: Dec. 17, 2025; Published: Dec. 25, 2025

Dynamics of QT Interval and QT Dispersion as Predicts of Life-Threating Aritmies in Patients with Oncological Diseases Receiving Cardiotoxic Treatment
Ergashov B. B.
Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Ergashov B. B., Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

A clinical and diagnostic study of the frequency and structure of arrhythmias in patients with oncological diseases receiving cardiotoxic chemotherapy was conducted. Electrolyte imbalance, prolongation of the QTc interval, and an increase in QT variance were significantly associated with an increased risk of life-threatening arrhythmias. Active correction of potassium and magnesium levels reduced the frequency of rhythm disturbances, and combined prevention using cardioprotective therapy provided maximum protective effect and prevented the development of clinically significant arrhythmias. The obtained results confirm the feasibility of dynamic monitoring of electrolytes and QT/QTc indicators to improve the safety of antitumor treatment.
Keywords: Heart arrhythmias, QT interval, QT dispersion, Chemotherapy, Cancer patients, Electrolyte balance, Hypokalemia, Hypomagnesemia
Cite this paper: Ergashov B. B., Dynamics of QT Interval and QT Dispersion as Predicts of Life-Threating Aritmies in Patients with Oncological Diseases Receiving Cardiotoxic Treatment, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4578-4580. doi: 10.5923/j.ajmms.20251512.82.
1. Introduction
- Modern advancements in oncology related to the introduction of anthracyclines, tyrosine kinase inhibitors, anti-HER2-preparations, and immuno-oncological therapy have led to a significant increase in the survival rate of cancer patients. At the same time, the expansion of the range of anti-tumor drugs is accompanied by an increase in the frequency of cardiotoxic complications, among which heart rhythm disorders, including potentially life-threatening ventricular arrhythmias, occupy a special place [1,2].One of the key electro-physiological markers of arrhythmogenic risk is the QT interval and its dispersion (QTd), reflecting the heterogeneity of myocardial repolarization processes. The prolongation of QTc and the increase in QT variance are considered independent predictors of the development of torsade de pointes, polymorphic ventricular tachycardia, and sudden cardiac death [3,4]. Under chemotherapy conditions, these changes can form both due to direct toxic effects on cardiomyocyte ion channels and against the background of electrolyte disturbances and inflammatory damage to the myocardium [5,6].Dynamic monitoring of QT/QTc and QTd is of particular clinical importance in patients receiving targeted and immuno-oncological therapy, as these drugs are often associated with subclinical changes in repolarization without a pronounced decrease in myocardial contractility [7,8]. It has been shown that an increase in QT variance can precede the clinical manifestation of arrhythmias and serve as an early diagnostic marker for an unfavorable prognosis [9].The recommendations of the European Society of Cardiologists for CardioOncology emphasize the need for regular monitoring of the QT/QTc interval in oncological patients receiving drugs with proarrhythmogenic potential, however, the issues of clinical interpretation of QT-dispersion dynamics, its prognostic significance, and threshold values remain debatable [10]. This determines the relevance of conducting research aimed at assessing the dynamics of the QT interval and its variance as predictors of life-threatening arrhythmias in patients with oncological diseases receiving cardiotoxic therapy.The purpose of the study is to assess the dynamics of the QT interval and QT variance as predictors of life-threatening arrhythmias in patients with oncological diseases receiving cardiotoxic anti-tumor therapy, with the aim of early stratification of the arrhythmogenic risk and optimization of preventive measures.
2. Materials and Methods of Research
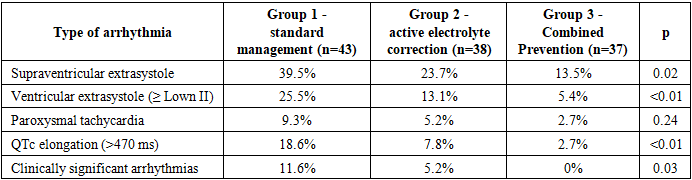
- The study included patients ≥18 years old with confirmed oncopathology receiving cardiotoxic chemotherapy. Patients were divided into three groups: 1st group - standard management without preventive electrolyte correction; 2nd group - active monitoring and correction of K+ and Mg2+ levels before each cycle; 3rd group - combined prevention, including electrolyte correction and cardioprotective therapy (β-blockers, ACEI/ARB). All patients underwent a clinical examination, standard ECG registration with an assessment of the QT/QTc interval, and, if necessary, Holter ECG monitoring, echocardiography, as well as laboratory blood electrolyte levels.The main endpoints of the study were the frequency of electrolyte imbalance disorders, the frequency of heart arrhythmias, and the dynamics of the QTc interval. Statistical processing of the data was carried out using standard methods of variation statistics; the differences were considered statistically significant at p<0.05.
3. Research Results
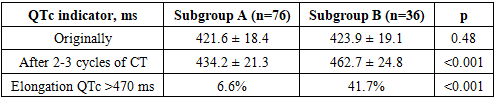
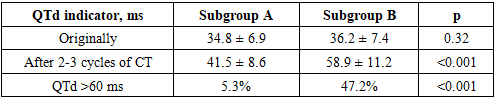
- To assess the prognostic significance of the QTc interval, a comparative analysis of its initial values and dynamics during chemotherapy was conducted in patients without clinically significant arrhythmias (subgroup A) and in patients with registered clinically significant or life-threatening arrhythmias (subgroup B). The QTc assessment was conducted before the start of antitumor treatment and after 2-3 cycles of chemotherapy (Table 1).
|
|
|
4. Conlcusions
- The conducted study showed that in patients with oncological diseases receiving cardiotoxic chemotherapy, electrolyte imbalance and changes in myocardial repolarization indicators (prolongation of QTc and increase in QT dispersion) are significant predictors of arrhythmia development. Standard management without preventive measures was associated with the highest frequency of supraventricular and ventricular rhythm disturbances, prolongation of the QTc interval, and clinically significant arrhythmias. Active control and correction of electrolytes significantly reduced the frequency of arrhythmias, while combined prevention with the inclusion of cardioprotective therapy provided maximum protective effect and practically completely prevented the development of clinically significant rhythm disorders. The obtained data justify the need to implement comprehensive monitoring of electrolytes and QT/QTc parameters in clinical practice to improve the safety of antitumor treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML