-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4561-4564
doi:10.5923/j.ajmms.20251512.77
Received: Nov. 27, 2025; Accepted: Dec. 23, 2025; Published: Dec. 25, 2025

Iron Metabolism Disorders and Mechanisms of Ferroptosis in the Pathogenesis of Focal Epilepsy
Abboskhonov A. A., Azizova R. B.
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

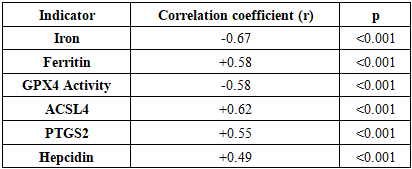
To evaluate alterations in iron metabolism markers and ferroptosis biomarkers in patients with focal epilepsy and determine their association with disease severity. Materials and Methods: The study included 85 patients with focal epilepsy and 40 healthy individuals as a control group. Serum levels of iron, ferritin, transferrin, transferrin saturation, hepcidin, ceruloplasmin, as well as ferroptosis biomarkers (GPX4, ACSL4, PTGS2), were measured in all participants. The frequency and severity of epileptic seizures were assessed. Results: Compared with the control group, patients with focal epilepsy had significantly lower serum iron levels (12.8±2.1 µmol/L vs. 18.4±1.9 µmol/L, p<0.001) and higher ferritin levels (285.7±45.2 ng/mL vs. 156.3±28.7 ng/mL, p<0.001). Among ferroptosis biomarkers, GPX4 activity was significantly reduced (0.42±0.08 U/mL vs. 0.78±0.12 U/mL, p<0.001), while ACSL4 and PTGS2 levels were increased (p<0.01). A negative correlation was found between seizure frequency and iron deficiency (r = -0.67, p<0.001), as well as GPX4 activity (r = -0.58, p<0.001). Conclusion: In focal epilepsy, disturbances in iron metabolism and activation of ferroptosis processes play an important role in the disease pathogenesis and reveal new potential therapeutic targets.
Keywords: Focal epilepsy, Iron metabolism, Ferroptosis, GPX4, Ferritin, Neurodegeneration
Cite this paper: Abboskhonov A. A., Azizova R. B., Iron Metabolism Disorders and Mechanisms of Ferroptosis in the Pathogenesis of Focal Epilepsy, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4561-4564. doi: 10.5923/j.ajmms.20251512.77.
Article Outline
1. Introduction
- Epilepsy is one of the most common neurological diseases, affecting more than 70 million people worldwide [1]. Focal epilepsy accounts for 60-70% of all cases of epilepsy and develops as a result of structural damage to the brain [2]. Despite significant advances in understanding the mechanisms of epileptogenesis in recent years, the pathophysiology of the disease is still not fully elucidated [3].Iron is a vital micronutrient for the body and plays a key role in the normal functioning of neurons [4]. Iron is involved in mitochondrial respiration, DNA synthesis, neurotransmitter biosynthesis, and myelination processes [5]. However, excessive iron accumulation can lead to oxidative stress and lipid peroxidation [6].Ferroptosis is a recently discovered form of iron-dependent cell death characterized by lipid peroxidation and decreased glutathione peroxidase 4 (GPX4) activity [7]. Ferroptosis processes have been identified in various neurological diseases, including Alzheimer's disease, Parkinson's disease, and stroke [8,9]. Recent studies suggest that ferroptosis may also play a role in the pathogenesis of epilepsy [10].Acyl-CoA synthetase long-chain family member 4 (ACSL4) is a key enzyme in ferroptosis, activating arachidonic acid and providing a substrate for lipid peroxidation [11]. Prostaglandin-endoperoxide synthase 2 (PTGS2) is involved in the process of ferroptosis in the production of lipid peroxidation products [12]. GPX4 is considered the main protective mechanism against ferroptosis [13].Hepcidin is the main hormone regulating iron homeostasis, controlling iron absorption in the intestine and its release from tissues [14]. Ceruloplasmin is a copper-containing protein involved in the processes of iron transport and oxidation [15].The aim of the study was to evaluate changes in iron metabolism parameters and biomarkers. ferroptosis in patients with focal epilepsy, to determine their relationship with the severity of the disease and the frequency of seizures.
2. Study Materials and Methods
- The study was conducted in 2023-2024 at the Neurology Clinic of the Tashkent Medical Academy using a prospective, controlled design. The study protocol was approved by the Institute's Bioethics Committee (Protocol No. 15/2023). Written consent was obtained from all participants.The study included 85 patients with focal epilepsy (the study group) and 40 healthy individuals (the control group). Inclusion criteria for the study group were: age 18-65 years, a confirmed diagnosis of focal epilepsy according to the ILAE 2017 criteria, confirmed structural brain damage based on MRI, and at least two seizure episodes in the past six months. Exclusion criteria included psychogenic seizures, severe somatic diseases, pregnancy and lactation, taking iron supplements, and episodes of blood loss in the past three months.All patients underwent a full neurological examination, EEG, and MRI. Seizure frequency was calculated over the past three months and expressed as the number of seizures per month. Disease severity was assessed using the NHS3 scale.For laboratory testing, 10 ml of fasting blood was collected from a vein in the morning. The following parameters were determined in the serum: iron (colorimetric method), ferritin (immunochemiluminescence method), transferrin (immunoturbidimetric method), transferrin saturation (calculated), hepcidin (ELISA method), and ceruloplasmin (immunoturbidimetric method).Biomarkers ferroptosis was determined by the ELISA method: GPX4 activity (Cayman Chemical, USA), ACSL4 level (MyBioSource, USA), PTGS2 level (R&D Systems, USA). All analyses were performed according to the manufacturers' recommendations.Statistical analysis was performed using SPSS 28.0. Normal distribution of the data was assessed using the Shapiro- Wilk test. Differences between groups were assessed using the Student's t-test or the Mann-Whitney U-test. Correlation analysis was performed using the Pearson or Spearman methods. A p value of <0.05 was considered statistically significant.
3. Study Results
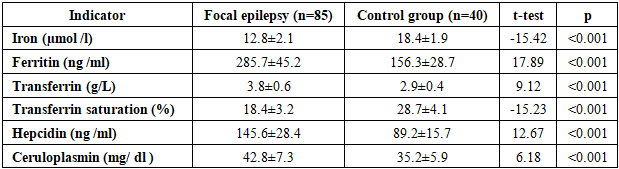
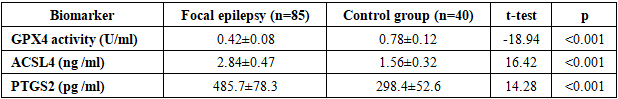
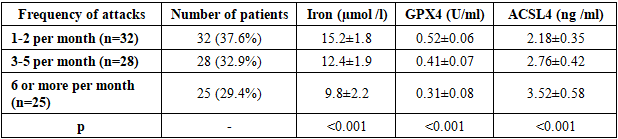
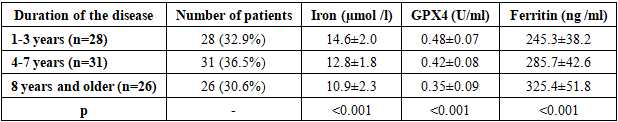
- The demographic characteristics of the patients included in the study were as follows. In the main group, the average age was 42.3±12.8 years, there were 48 men (56.5%), 37 women (43.5%). In the control group, the average age was 40.7±11.2 years, there were 22 men (55.0%), 18 women (45.0%). No statistical differences in age and gender distribution were found between the groups (p>0.05).The distribution by etiology of epilepsy was as follows: traumatic brain injury - 28 people (32.9%), consequences of stroke - 22 people (25.9%), brain tumors - 15 people (17.6%), infectious lesions - 12 people (14.1%), other causes - 8 people (9.4%). The duration of the disease was on average 6.8±4.2 years.Table 1 presents the results of a comparative analysis of iron metabolism parameters between the groups of patients with focal epilepsy and healthy controls. The data demonstrate statistically significant differences in all iron metabolism parameters studied.
|
|
|
|
|
4. Discussion
- The obtained results revealed disturbances in iron metabolism and activation of ferroptosis processes in focal epilepsy. Iron deficiency and elevated ferritin levels observed in patients indicate iron accumulation in tissues and a decrease in serum iron. This condition may be associated with neuronal damage and inflammatory processes resulting from epileptic seizures. hepcidin levels reflect the body's attempt to restore iron homeostasis. Hepcidin limits iron absorption in the intestine and reduces its release from macrophages, resulting in serum iron deficiency. Elevated ceruloplasmin levels may be understood as a response to oxidative stress.A significant decrease in GPX4 activity indicates activation of ferroptosis. GPX4 is the primary enzyme that reduces lipid peroxides, and decreased activity leads to damage to cell membranes. Increased levels of ACSL4 and PTGS2 confirm activation of ferroptosis.Strong correlation between seizure frequency and iron deficiency biomarkers Ferroptosis studies show that these processes play an important role in epileptogenesis. Iron deficiency can disrupt neurotransmitter synthesis and increase neuronal excitability.The progression of iron metabolism disorders with increasing disease duration indicates the development of neurodegenerative processes in chronic epilepsy. These data highlight the importance of correcting iron metabolism in the early stages.Recent studies have demonstrated the role of ferroptosis in various neurological diseases. For example, Zhang et al. (2023) found activation of ferroptosis in Alzheimer's disease. Liu et al. (2022) demonstrated decreased GPX4 activity in stroke models.Our results are consistent with those obtained by Wang et al. (2023) in animal models of epilepsy. They also found decreased GPX4 activity and increased ACSL4 levels.Limitations of the study include the relatively small sample size and cross-sectional design. Long-term observational studies and evaluation of the effectiveness of therapeutic interventions are needed in the future.
5. Conclusions
- Impaired iron metabolism and activated ferroptosis were detected in patients with focal epilepsy. Serum iron deficiency, elevated ferritin and hepcidin levels, decreased GPX4 activity, and elevated ACSL4 and PTGS2 levels were observed. These changes correlated with seizure frequency and disease duration. These results demonstrate the important role of iron metabolism and ferroptosis in the pathogenesis of focal epilepsy and provide a basis for the development of new therapeutic strategies. Correction of iron metabolism and prevention of ferroptosis may provide new avenues for epilepsy treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML