Uzakov D. K.1, Mirubaydev N. Sh.1, Mardonova D. U.1, Ibragimov N. K.2
1Department of Anesthesiology and Intensive Care, Navoi Regional Branch of the Republican Specialized Scientific-Practical Medical Center of Endocrinology named after Academician Yo. Kh. Turakulov, Navoi, Uzbekistan
2Dsc., Professor, Department of Anesthesiology and Intensive Care, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Uzakov D. K., Department of Anesthesiology and Intensive Care, Navoi Regional Branch of the Republican Specialized Scientific-Practical Medical Center of Endocrinology named after Academician Yo. Kh. Turakulov, Navoi, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In patients with diabetes mellitus complicated by purulent septic soft-tissue lesions, cardiovascular insufficiency develops under the combined influence of systemic, central, and microcirculatory disturbances. The severity of hemodynamic dysfunction is closely associated with the degree of endogenous intoxication and follows a cascade transition from compensated circulatory patterns to overt circulatory decompensation. Metabolic derangements including lactic acidosis, alterations in pH, and base excess further exacerbate the progression of cardiovascular failure and substantially increase the risk of septic shock.
Keywords:
Iabetes mellitus, Necrotizing soft tissue infection, Sepsis, Septic shock, Hemodynamic phenotypes, Endogenous intoxication, Microcirculatory dysfunction, Vasoplegia, Hyperlactatemia, Echocardiography, VExUS score
Cite this paper: Uzakov D. K., Mirubaydev N. Sh., Mardonova D. U., Ibragimov N. K., Features of Hemodynamic Disorders in Purulent–Septic Complications in Patients with Diabetes Mellitus, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4545-4553. doi: 10.5923/j.ajmms.20251512.75.
1. Introduction
Hemodynamic instability in patients with diabetes mellitus complicated by purulent septic soft-tissue infections exhibits a distinctly polymorphic pattern [1,3,8,9,14]. The simultaneous influence of vasoplegia, myocardial dysfunction, and microcirculatory impairment leads to rapid exhaustion of circulatory compensatory mechanisms [17,20]. The presence of diabetic cardiomyopathy, atherosclerotic vascular disease, and diabetic nephropathy creates an initial vulnerability of the cardiovascular system to septic stress [4,5,11,12,19]. Massive release of cytokines and nitric oxide in sepsis causes profound vascular tone reduction, disruption of blood-flow autoregulation, and contributes to the progression of circulatory failure [10,13,16,18].The combination of metabolic, vascular, and infectious aggression makes the clinical course highly unpredictable and necessitates an integrated endocrinological, surgical, and critical-care approach. [2,6,22]. Patients in this category frequently develop multiple organ dysfunction, long-term dependence on vasoactive support, and sustain a persistently high risk of mortality, underscoring the need for differentiated algorithms for hemodynamic correction [7,15,21].Study Aim To improve treatment outcomes in patients with diabetes mellitus complicated by purulent–septic endotoxicosis through early identification of central hemodynamic disturbances and determination of hemodynamic phenotypes that enable targeted and optimized correction during intensive care.Research ObjectivesTo achieve this aim, the following objectives were defined:1. To determine the key pathophysiological mechanisms of central hemodynamic disturbances in patients with diabetes mellitus complicated by purulent septic soft tissue lesions.2. To characterize the sequence of hemodynamic disorder development and identify their phenotypic structure.3. To identify common pathogenetic determinants that define the clinical and diagnostic relevance of the identified phenotypes.4. To develop an algorithm for diagnostic identification of hemodynamic phenotypes based on instrumental and clinical indicators.5. To determine early predictors of hemodynamic decompensation in the setting of sepsis.
2. Materials and Methods
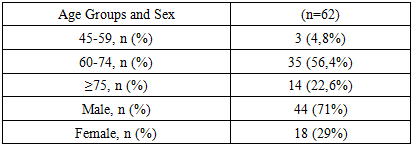
General Characteristics of the PatientsThe study included 62 patients with diabetes mellitus complicated by purulent-septic soft tissue lesions, who were treated in the Intensive Care Unit of the Navoi Regional Branch of the Republican Specialized Scientific Practical Medical Center of Endocrinology named after Academician Yo. Kh. Turakulov during the period 2021–2023.The cohort consisted of patients presenting with foot and lower-leg phlegmon, necrotizing fasciitis, abscesses, and purulent-necrotic gangrene that developed on the background of type 1 or type 2 diabetes mellitus.All 62 patients (100%) received treatment during 2021-2023.During this period, traditional infusion and vasoactive therapy protocols were applied, without advanced hemodynamic monitoring, and the assessment of clinical status was based on standard central hemodynamic parameters.Analysis of demographic characteristics showed that the majority of patients were aged 60–74 years (56.4%), which reflects the typical age-related predisposition to diabetes-associated purulent-septic complications. Patients aged ≥75 years accounted for 22.6%, whereas individuals younger than 60 years were considerably less common (4.8%).The gender distribution was characterized by a pronounced predominance of men (71%) over women (29%), which corresponds to the higher incidence of severe infectious-necrotic processes and peripheral angiopathy among males with long-standing diabetes mellitus (Table 1).Table 1. Age and Sex Distribution of the Patients
 |
| |
|
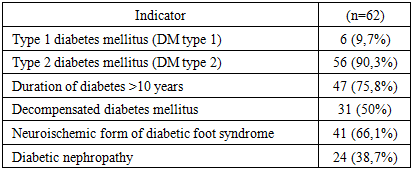
Brief Characteristics of the Patients’ Diabetological StatusIn this cohort, the majority of patients had type 2 diabetes mellitus (90.3%), whereas type 1 diabetes was considerably less common (9.7%). Most patients had a long disease duration of more than 10 years (75.8%), indicating advanced diabetic angiopathy and neuropathy.Half of the patients (50%) were admitted in a state of decompensated carbohydrate metabolism. The neuroischemic form of diabetic foot was identified in 66.1% of cases, confirming the key role of combined angiopathy and neuropathy in the development of purulent-necrotic lesions. Diabetic nephropathy was present in 38.7% of patients, further increasing the risk of endotoxicosis and hemodynamic instability (Table 2).Table 2. Characteristics of Diabetes Mellitus in Patients with Purulent-Septic Soft Tissue Lesions
 |
| |
|
The analysis of clinical forms of purulent-necrotic complications demonstrated that phlegmon of the foot and lower leg was the most prevalent condition, accounting for 43.5% of all cases. This reflects the typical spread of infection along inter-fascial planes in diabetic foot syndrome.Necrotizing fasciitis was identified in 17.7% of patients, while necrotizing myonecrosis was found in 9.7%, confirming the high aggressiveness of the infectious process in individuals with diabetes.Forms associated with ischemia were also common. Moist gangrene of the toes and foot occurred in 12.9% of patients, whereas mixed gangrene was present in 8.1%, indicating a significant ischemic component in disease progression.Additionally, combined lesions were observed in 8.1% of cases and were associated with the most severe clinical course, carrying a markedly increased risk of rapid transition to sepsis (Table 3).Table 3. Distribution of patients by clinical forms of purulent-septic necrotic soft-tissue lesions in the setting of diabetes mellitus
 |
| |
|
At admission, most patients demonstrated signs of systemic inflammatory response and varying degrees of organ dysfunction. Nearly half of the cohort (45.2%) presented with a qSOFA score ≥ 2, indicating a high risk of adverse outcomes.An infectious process without systemic inflammatory response was observed in 22.6% of patients.Sepsis (SOFA ≥ 2 without septic shock) was diagnosed in 53.2%, whereas septic shock was identified in 24.2%, reflecting the high prevalence of severe septic states in this population.Multiple organ dysfunction syndrome (MODS) was documented in 17.7% of patients, indicating substantial systemic impairment at the time of admission (Table 4).Table 4. Distribution of patients with diabetes mellitus complicated by purulent-septic soft tissue lesions according to Sepsis-3 and qSOFA criteria
 |
| |
|
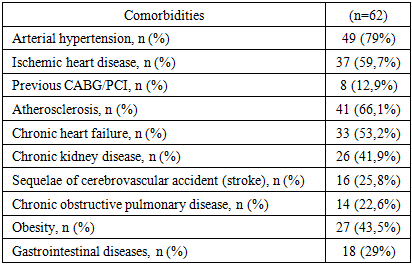
The cardiovascular profile dominated the structure of comorbidities and determined the baseline vulnerability to hemodynamic decompensation (Table 5).Table 5. Comorbidities in patients with diabetes mellitus complicated by purulent-septic soft tissue lesions
 |
| |
|
Thus, the combination of demographic, metabolic, and comorbid characteristics determined the severe course of necrotizing and septic soft-tissue infections in patients with diabetes mellitus and underscored the need for advanced systemic hemodynamic monitoring and pathogenetically oriented therapeutic correction strategies.MethodsClinical observation was performed from the moment of admission until completion of treatment in the Intensive Care Unit. Demographic data, characteristics of diabetes mellitus, features of the purulent-septic process, and physiological parameters (level of consciousness, temperature, respiratory rate, heart rate, urine output, and pain intensity) were recorded. Disease severity was assessed at admission and subsequently at 24, 48, and 72 hours.Laboratory monitoring included complete blood count, biochemical profile, coagulation tests, arterial blood gas analysis, and acid–base status, performed at the same time points. Hourly urine output was used as a perfusion marker: a rate below 0.5 mL/kg/h for ≥2 hours was considered an early indicator of organ dysfunction according to Sepsis-3 criteria.The degree of endogenous intoxication was assessed using three integral indices: the Leukocytic Intoxication Index (LII), the Hematologic Poisoning Index (HPI), and the Middle Molecular Peptides (MMP).The LII was calculated according to the Kalf-Kalif formula.The HPI was determined as the product of the LII and correction coefficients reflecting the severity of leukocytosis and the acceleration of ESR.The MMP level was measured spectrophotometrically at 238/254/280 nm with subsequent calculation of diagnostic ratios indicating the accumulation of toxic peptide fractions.Grading of endogenous intoxication was performed according to a three-component system:• Mild: LII < 2; HPI < 1.5; MMP ≤ 0.270• Moderate: LII 2–4; HPI 1.5–2.5; MMP 0.271–0.32• Severe: LII > 4; HPI > 2.5; MMP > 0.320Instrumental MethodsInstrumental assessment included measurement of mean arterial pressure (MAP), heart rate (HR), central venous pressure (CVP), urine output, and echocardiography (ECHO) parameters.To determine the degree of venous congestion, the VExUS score (0–3) was applied based on Doppler ultrasonography of the hepatic, portal, and renal veins.A standard electrocardiogram (ECG) was also performed.The clinical, laboratory, and instrumental findings were subsequently integrated to stratify patients according to systemic hemodynamic phenotypes.Study DesignThe study was conducted in a retrospective format and included patients treated in the Intensive Care Unit of the Navoiy Regional Branch of the Republican Specialized Scientific-Practical Medical Center of Endocrinology named after Academician Yo. Kh. Turakulov during the period 2021–2023.The study design complied with the principles of the Declaration of Helsinki (2013) and was approved by the Local Ethics Committee of Tashkent Medical University.Inclusion Criteria• age older than 18 years;• confirmed type 1 or type 2 diabetes mellitus;• presence of necrotizing purulent-septic soft tissue infections requiring intensive therapy;• feasibility of dynamic central hemodynamic monitoring;• absence of contraindications to standard intensive care;• availability of informed consent.Exclusion Criteria• age <18 years;• terminal stages of chronic heart, liver, or renal failure;• active oncological diseases;• HIV infection;• specific chronic infections (including tuberculosis);• severe neuropsychiatric disorders;• pregnancy and postpartum period <6 weeks;• absence of informed consent.The study design represented a sequential analytical cycle aimed at identifying, stratifying, and subsequently validating hemodynamic disturbance phenotypes in patients with diabetes mellitus complicated by necrotizing purulent-septic soft tissue infections.Study StagesStage 1. Retrospective AnalysisA systematic processing of clinical, laboratory, and instrumental data was performed for patients treated between 2021 and 2023. This stage enabled the formation of a representative control cohort and allowed assessment of the natural course of the disease as well as the structure of hemodynamic instability in necrotizing purulent-septic soft tissue infections (NPSTI) occurring on the background of diabetes mellitus.Stage 2. Analytical Phase Focused on Phenotype IdentificationThe analytical component of the study was centered on:• comparing the degree of endogenous intoxication with macro- and microhemodynamic parameters;• assessing systemic and regional perfusion;• identifying stable patterns of hemodynamic disturbances;• forming hemodynamic phenotypes according to the severity of central hemodynamic impairment.Thus, the study design was deliberately oriented toward determining hemodynamic disturbance phenotypes and elucidating their pathophysiological mechanisms in patients with diabetes mellitus complicated by necrotizing purulent-septic soft tissue infections.The complex of therapeutic and diagnostic interventions in this patient cohort was carried out in accordance with current clinical guidelines for the management of necrotizing soft-tissue infections and diabetes-related complications, as well as international protocols (SSC 2021, IDSA 2023).
3. Results
The analysis of central hemodynamics was based on the preliminary stratification of patients according to the degree of endogenous intoxication (EI).Since EI reflects the level of systemic toxic impact and determines the nature of hemodynamic responses, its gradation was used as the starting point for phenotyping. Subsequently, for each EI category, the following parameters were analyzed:• the structure of the hemodynamic profile;• the characteristics of compensatory cardiovascular responses;• the severity of systemic and regional perfusion impairments;• the formation of hemodynamic phenotypes.The results demonstrated that the majority of patients belonged to the group with moderate endogenous intoxication 26 cases (41.9%). Mild EI was recorded in 19 patients (30.6%), whereas severe EI was observed in 17 patients (27.5%).A comparative analysis of qSOFA scores across subgroups stratified by the degree of endogenous intoxication revealed a consistent relationship between the severity of toxemia and the clinical severity of the condition (Table 6).Table 6. Distribution of patients according to qSOFA values depending on the degree of endogenous intoxication
 |
| |
|
In patients with mild endogenous intoxication (EI), qSOFA = 1 was observed significantly more often than in those with more severe intoxication 2.2 times more frequently compared with the moderate EI group and 7.1 times more frequently than in the severe EI group. This pattern reflects a predominantly compensated systemic response and a relatively preserved hemodynamic stability.In patients with moderate EI, a shift toward higher qSOFA scores was observed: the frequency of qSOFA ≥ 3 was 2.2 times higher than in the mild EI group, accompanied by a statistically significant increase in the mean qSOFA value (p < 0.05). This trend indicates a transition from compensated forms of toxemia to more pronounced systemic disturbances and emerging signs of hypoperfusion.In patients with severe EI, the proportion of cases with qSOFA ≥ 3 was twice as high as in the moderate EI group and 4.5 times higher than in the mild EI group. The mean qSOFA values increased consistently from 1.74 ± 0.78 (mild EI) to 2.15 ± 0.84 (moderate EI) and 2.88 ± 0.62 points (severe EI), p < 0.01. These findings confirm the progressive intensification of systemic disturbances and the development of a clinical picture consistent with sepsis-associated hypoperfusion.Overall, the obtained data demonstrate a clear relationship between the severity of EI and the extent of central hemodynamic impairment as assessed by the qSOFA scale.The analysis of central hemodynamic parameters revealed statistically significant differences between the subgroups stratified according to the degree of EI (Table 7).Table 7. Central Hemodynamic Parameters in Control Group Patients According to the Degree of Endogenous Intoxication (EI)
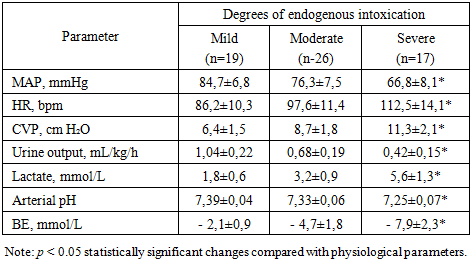 |
| |
|
In cases of mild EI, systemic hemodynamic parameters remained close to physiological values: MAP was within the normal range, heart rate was moderately elevated, and CVP was at the lower limit of reference values. Urine output exceeded the minimum threshold of 1 mL/kg/h by approximately 1.5 times, while lactate and acid base balance parameters remained within normal limits. These findings indicate a preserved compensatory reserve and adequate tissue perfusion under conditions of moderate activation of the sympathoadrenal system.In moderate EI, a transition toward a hyperdynamic circulation was observed: MAP decreased (by approximately 1.1-fold), heart rate increased, and CVP rose 1.4-fold. Urine output declined 1.3-fold, blood lactate increased 1.7-fold, and a mild metabolic acidosis developed (moderate reduction of pH and a negative BE). These changes indicate the onset of systemic perfusion disorganization.In severe EI, pronounced decompensation was observed: MAP decreased an additional 1.15-fold compared with the moderate EI group, HR exceeded the values of the mild EI group by 1.3-fold, and CVP increased to 11.3 ± 2.1 cm H₂O. Urine output fell below the physiological threshold, while lactate levels rose 3.1-fold compared with mild EI. Acid base analysis revealed moderate metabolic acidosis with a markedly negative BE. The combination of these findings is indicative of severe hypoperfusion and progressive tissue ischemia.The overall analysis demonstrated a pronounced relationship between the severity of endogenous intoxication and the pattern of hemodynamic disturbances. Statistically significant differences were identified for MAP, urine output, lactate levels, and BE (p < 0.01), confirming a direct pathophysiological link between the depth of toxemia, reduced perfusion, and progressive tissue hypoxia. Thus, EI serves not only as an indicator of infection severity but also as a key determinant in the development of macro- and microhemodynamic disorders, making it an essential parameter in selecting an optimal intensive care strategy.A frequency analysis showed that the prevalence of critical hemodynamic disturbances correlated strongly with the severity of endogenous intoxication (Table 8). A reduction in MAP below 65 mmHg was observed in 10.5% of patients with mild EI, increased 2.9-fold in moderate EI (30.8%), and reached 76.5% in severe EI (χ² = 22.9; p < 0.001). A similar pattern was found for elevated CVP: 15.8% in mild EI, 34.6% in moderate EI, and 70.6% in severe EI (χ² = 14.8; p < 0.01). These changes indicate a transition from compensatory redistribution of circulation toward marked venous congestion and loss of vascular reactivity as toxemia intensifies.Table 8. Frequency of major hemodynamic and perfusion disorders in patients depending on the degree of endogenous intoxication
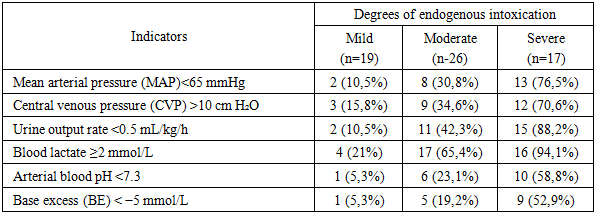 |
| |
|
The dynamics of renal function demonstrated a clear dependence on the severity of endogenous intoxication. The frequency of a decrease in urine output below 0.5 mL/kg/h increased sequentially from 10.5% in patients with mild endogenous intoxication to 42.3% in those with moderate intoxication and reached 88.2% in cases of severe intoxication (χ² = 29.5; p < 0.001). Thus, the risk of oliguria in severe toxemia was 8.4 times higher than in mild intoxication. These changes reflect a progressive deterioration of microcirculation and activation of circulatory centralization mechanisms, making reduced urine output one of the most sensitive indicators of worsening hypoperfusion.Lactate concentration demonstrated a pronounced dependence on the severity of endogenous intoxication. Levels ≥2.0 mmol/L were observed in 21% of patients with mild intoxication, 65.4% with moderate intoxication, and 94.1% with severe intoxication, corresponding to a 4.5-fold increase in the prevalence of hyperlactatemia (χ² = 27.3; p < 0.001). This trend reflects a stepwise escalation of tissue hypoxia and progressive metabolic derangements as toxemia intensifies.In patients with severe endogenous intoxication, hyperlactatemia was accompanied by more frequent signs of metabolic acidosis: arterial pH <7.3 was recorded in 58.8% of cases (χ² = 15.6; p < 0.01), and a negative base excess (BE < 5 mmol/L) in 52.9%, which was 11- and 10-fold higher, respectively, than in patients with mild intoxication (χ² = 14.2; p < 0.01). These findings confirm profound disturbances of cellular metabolism and a critical reduction in the efficiency of tissue perfusion.The obtained data demonstrate that, as endogenous intoxication intensifies, hemodynamic disturbances not only become more frequent but also acquire a combined character. Severe endogenous intoxication is characterized by the simultaneous presence of decreased mean arterial pressure, pronounced oliguria, metabolic acidosis (reduced pH and base excess), and elevated blood lactate levels in the setting of increased central venous pressure, reflecting the development of persistent systemic hypoperfusion.The incidence of septic shock demonstrated a clear dependence on the severity of endogenous intoxication. In patients with mild endogenous intoxication, signs of septic shock were observed in 26.3% of cases. As toxemia intensified, the proportion of such patients increased markedly: by 2.5-fold in moderate endogenous intoxication and reaching 88.2% in severe endogenous intoxication (χ² = 19.7; p < 0.001) (Table 9).Table 9. Comparative distribution of patients according to the Sepsis-3 classification depending on the degree of endogenous intoxication (EI)
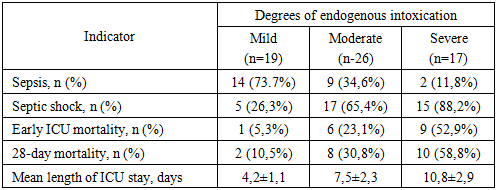 |
| |
|
This pattern indicates the leading role of endogenous intoxication in the development of hemodynamic instability. The progression of toxemia is accompanied by worsening vasoplegia, redistribution of blood flow, and a reduction in perfusion pressure, which predictably increases the incidence of conditions meeting the criteria for septic shock according to the Sepsis-3 classification (Table 9).Mortality in the ICU demonstrated a clear dependence on the severity of endogenous intoxication. At a moderate degree of EI, mortality was 4.3 times higher than at a mild degree, while at a severe degree it increased by 9.9 times (χ² = 14.9; p < 0.01). A similar trend persisted over the 28-day period: mortality increased 2.9-fold from mild to moderate EI and exceeded a fivefold increase at severe EI (χ² = 12.7; p < 0.01) (Table 9).This pattern reflects a stepwise deepening of microcirculatory and tissue perfusion disorders as toxemia progresses. A characteristic feature was the high rate of early mortality: the majority of deaths among patients with moderate and severe endogenous intoxication occurred within the first 1–3 days of ICU stay. This indicates a rapidly progressive course of septic shock and a limited compensatory reserve in the setting of severe intoxication. Thus, an increase in the incidence of septic shock directly correlates with a rise in early mortality cases (Table 9).The length of ICU stay showed a clear dependence on the severity of endogenous intoxication. In patients with moderate EI, the duration of treatment increased 1.8-fold compared with those with mild EI, while in severe EI it increased 2.6-fold (p < 0.001). Thus, increasing EI severity was associated with delayed stabilization of central hemodynamics and a greater need for complex correction of volume status and vascular parameters (Table 9).A comprehensive analysis of central hemodynamics in patients with diabetes mellitus and purulent-septic necrotic complications revealed a clear dependence of the severity of systemic disturbances on the degree of endogenous intoxication (EI).In mild EI, the changes were minimal: macrocirculation remained stable, compensatory sympathoadrenal mechanisms ensured adequate perfusion, and lactate levels and urine output were within normal ranges.In moderate EI, a hyperdynamic circulatory pattern developed, characterized by a decrease in mean arterial pressure, an increase in central venous pressure, moderate tachycardia, reduced urine output, and elevated lactate levels. These features reflected progressive hypoperfusion and insufficiency of compensatory responses.In severe EI, a decompensated hemodynamic profile was observed, including persistent hypotension, elevated central venous pressure, marked reduction in urine output, and refractory hyperlactatemia unresponsive to correction. This indicated profound microcirculatory failure and refractoriness to standard therapy.An increase in the degree of endogenous intoxication (EI) was associated with higher early and 28-day mortality rates (p < 0.01), underscoring the pivotal role of toxemia in the formation of hemodynamic phenotypes and outcome prediction.Taken together, the findings demonstrate that progression of endogenous intoxication is accompanied by a stepwise deterioration of systemic hemodynamics. In mild EI, hemodynamic deviations were minimal and adequately compensated. In moderate EI, a hyperdynamic profile emerged with signs of initial hypoperfusion, including decreased mean arterial pressure, increased central venous pressure, moderate tachycardia, reduced urine output, and rising lactate levels. In severe EI, a decompensated circulatory pattern developed, characterized by persistent hypotension, marked venous congestion, a sharp decline in urine output, and sustained hyperlactatemia.These patterns reflect a transition from compensated disturbances to circulatory decompensation and confirm the role of endogenous intoxication as a key factor in the destabilization of central hemodynamics. The identified findings formed the basis for the subsequent analytical phase of the study.
4. Discussion
The obtained results indicate that in purulent-septic necrotic lesions (PSNL), the development of septic shock is driven by the sequential involvement of four fundamental pathophysiological mechanisms: vasoplegia, myocardial dysfunction, venous congestion, and microcirculatory disturbances (Fig. 1). 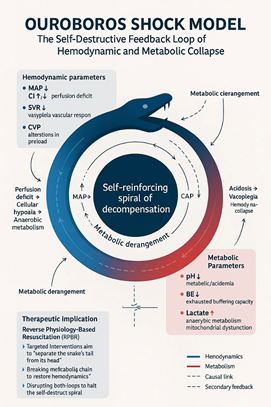 | Figure 1. The Ouroboros shock model illustrating the self-reinforcing feedback loop between hemodynamic instability and metabolic derangement leading to progressive circulatory collapse |
These mechanisms have a mutually potentiating nature and are incorporated in an evolutionary cascade, resulting in a progressive impairment of systemic circulation. At the early pathogenetic stages, the manifestation of these mechanisms is not universal; rather, they occur in different combinations depending on the severity of the underlying pathophysiological process. This determines the need to assess the central hemodynamic response not through isolated parameters, but by means of a comprehensive, system-based approach.From this perspective, the typification of early hemodynamic responses made it possible to gain a deeper understanding of the disease pathogenesis. The conducted differential analysis allowed the identification of five clinico-pathophysiological phenotypes, each characterized by a unique combination of:• systemic parameters (arterial tone, systemic vascular resistance, perfusion pressure),• central parameters (cardiac output, central venous pressure, preload–afterload relationships),• tissue-level indicators (lactate concentration, oxygenation status, microcirculation).These characteristics determine the direction of subsequent decompensation. Such phenotypic stratification reveals the complexity of septic shock development, as hemodynamic disturbances involve different regulatory levels gradually, engaging pathogenic mechanisms sequentially rather than simultaneously, in an evolutionary manner. As a result, each phenotype is characterized by its own dominant pathogenetic vector, which creates a rationale for the development of individualized, phenotype-oriented therapeutic strategies.Such phenotyping makes it possible to clearly delineate the transition from compensated vasoplegia to congestive–metabolic forms of cardiovascular failure (Table 10).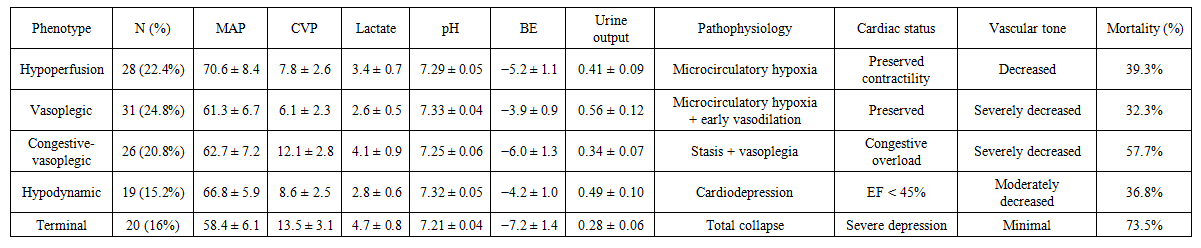 | Table 10. Hemodynamic phenotypes and associated pathophysiological characteristics |
The first phenotypic variant was the hypoperfusion phenotype, which included 28 patients (22.4%) of the studied cohort. This phenotype was characterized by a moderate decrease in mean arterial pressure (MAP: 70.6 ± 8.4 mmHg) in combination with a pronounced increase in blood lactate levels (3.4 ± 0.7 mmol/L) and metabolic acidosis (pH 7.29 ± 0.05; base excess 5.2 ± 1.1 mmol/L). Central venous pressure remained within normal limits (7.8 ± 2.6 cm H₂O), indicating preserved myocardial contractile function and reduced systemic vascular resistance (Table 10).In this group, a predominantly microcirculatory form of hypoxia without marked venous congestion was identified, where impaired tissue oxygen utilization represented the principal determinant of severity. Urine output in patients with this phenotype averaged 0.41 ± 0.09 mL/kg/h, and ICU mortality reached 39.3% (Table 10).The vasoplegic phenotype, identified in 31 patients (24.8%), reflects the classic presentation of early septic shock, characterized by a decrease in mean arterial pressure (MAP to 61.3 ± 6.7 mmHg), low central venous pressure (CVP: 6.1 ± 2.3 cm H₂O), and a moderate increase in blood lactate concentration (2.6 ± 0.5 mmol/L) (Table 10).This variant was typical of the early phase of systemic vasodilation, in which hypotension is primarily driven by a reduction in systemic vascular resistance while cardiac pump function remains preserved. Clinically, this phenotype demonstrated the highest responsiveness to vasopressor therapy, and ICU mortality in this group remained relatively moderate (32.3%).In patients with this hemodynamic phenotype, mean urine output was 0.56 ± 0.12 mL/kg/h, arterial blood pH was 7.33 ± 0.04, and base excess was 3.9 ± 0.9 mmol/L, indicating mild metabolic disturbances consistent with early-stage circulatory dysfunction.The third hemodynamic phenotype (congestive vasoplegic) was identified in 26 patients (20.8%) and combined features of venous congestion and systemic vasoplegia. This phenotype was characterized by an increase in central venous pressure (CVP to 12.1 ± 2.8 cm H₂O) in the presence of low mean arterial pressure (MAP: 62.7 ± 7.2 mmHg) and a marked reduction in urine output (0.34 ± 0.07 mL/kg/h). These changes were accompanied by the most pronounced lactic acidosis (lactate 4.1 ± 0.9 mmol/L; pH 7.25 ± 0.06; BE 6.0 ± 1.3 mmol/L).Such a hemodynamic configuration indicates a combination of central venous congestion and microcirculatory impairment, which is typical of the late stage of septic shock, associated with signs of myocardial depression and redistribution of circulating volume toward the venous compartment. Mortality in this group reached 57.7%, reflecting the severity and advanced nature of circulatory failure in this phenotype.The hypodynamic phenotype was identified in 19 patients (15.2%) and was characterized by normal or moderately elevated central venous pressure (8.6 ± 2.5 cm H₂O), reduced mean arterial pressure (66.8 ± 5.9 mmHg), and moderate hyperlactatemia (2.8 ± 0.6 mmol/L).The key distinguishing feature of this phenotype was a reduced left ventricular ejection fraction (<45%) on echocardiography, indicating the presence of primary myocardial contractile dysfunction. This group was typically characterized by a combination of diabetes-associated cardiomyopathy and sepsis-induced myocardial depression, rendering these patients particularly sensitive to inotropic support and careful preload optimization.In this cohort, urine output decreased to 0.49 ± 0.10 mL/kg/h, arterial pH was 7.32 ± 0.05, and base excess was 4.2 ± 1.0 mmol/L. The ICU mortality rate in this phenotype reached 36.8%, reflecting a significant contribution of myocardial dysfunction to hemodynamic instability and clinical outcomes.Finally, the combined (terminal) phenotype, identified in 20 patients (16%), represents the most severe form of systemic decompensation. It is characterized by critical hypotension (mean arterial pressure 58.4 ± 6.1 mmHg), pronounced venous hypertension (central venous pressure 13.5 ± 3.1 cm H₂O), anuria (urine output 0.28 ± 0.06 mL/kg/h), and severe lactic acidosis (blood lactate 4.7 ± 0.8 mmol/L, pH 7.21 ± 0.04, base excess −7.2 ± 1.4 mmol/L).Patients within this cohort exhibited a combination of marked venous congestion, global hypoperfusion, and profound energy failure, reflecting a terminal breakdown of compensatory mechanisms. This phenotype was associated with the highest ICU mortality rate, reaching 73.5%, underscoring its prognostic severity and refractoriness to standard intensive therapy.
5. Conclusions
In patients with diabetes mellitus complicated by purulent-septic soft tissue infections, central hemodynamic disturbances develop as a result of the sequential involvement of four principal mechanisms vasoplegia, myocardial dysfunction, venous congestion, and microcirculatory failure.• The combination of these mechanisms allows the identification of five clinico-pathophysiological phenotypes, each of which determines the pattern and direction of subsequent circulatory decompensation.• The common final pathway for all phenotypes is microcirculatory hypoperfusion and anaerobic metabolism, which directly lead to the development of septic shock.• Reliable differentiation of hemodynamic phenotypes requires targeted instrumental monitoring, including echocardiography, VTI, VExUS, ScvO₂, the Pv–aCO₂ gradient, lactate clearance, and capillary refill time (CRT).• The analysis demonstrated that the key early determinants of decompensation are elevated lactate levels, decreased pH and base excess (BE), reduced urine output, and arterial hypotension, which define the transition from perfusion deficit to myocardial dysfunction.
List of Abbreviations
• AH — arterial hypertension• BE — base excess• CABG — coronary artery bypass grafting• CKD — chronic kidney disease• CRT — capillary refill time• CVP — central venous pressure• DAS — diabetes-associated sepsis• DM — diabetes mellituso DM type 1 — type 1 diabetes mellituso DM type 2 — type 2 diabetes mellitus• DN — diabetic nephropathy• ECHO — echocardiography• ED — endothelial dysfunction• EI — endogenous intoxication• ECG — electrocardiography• EF — ejection fraction• EF-LV — left ventricular ejection fraction• GIT — gastrointestinal tract• GPI — hematological intoxication index• PSNSTI — purulent-septic necrotizing soft-tissue infections• HF — heart failure• HIV — human immunodeficiency virus• ICU — intensive care unit• IDSA — Infectious Diseases Society of America• ISS — intoxication severity score• LII — leukocyte intoxication index• LV — left ventricle• MAP — mean arterial pressure• MSM — middle-molecular-weight molecules• NI-DFS — neuro-ischemic form of diabetic foot syndrome• OPSS / SVR — systemic vascular resistance• OR — odds ratio• qSOFA — quick Sequential Organ Failure Assessment• ScvO₂ — central venous oxygen saturation• Sepsis-3 — Third International Consensus Definitions for Sepsis and Septic Shock• SBP — systolic blood pressure• SIRS — systemic inflammatory response syndrome• SOFA — Sequential Organ Failure Assessment• SSC 2021 — Surviving Sepsis Campaign, 2021 guidelines• SV — stroke volume• SVR — systemic vascular resistance• VExUS — Venous Excess Ultrasound Score• VTI — velocity–time integral• Pv–aCO₂ — venous-to-arterial carbon dioxide gradient
References
| [1] | Glukhov AA, Sergeev VA, Semenova GA. Results of treatment of patients with purulent-necrotic complications of diabetic foot syndrome using programmable technologies. Vestnik Khirurgii im. I.I. Grekova. 2018; 177(6): 63–68. |
| [2] | Ivanusa SYa, Zubarev PN, Risman BV, et al. Possibilities of local treatment of purulent-necrotic complications of diabetic foot syndrome. In: Sepsis et cetera: Proceedings of the Conference of the Association of General Surgeons, Yaroslavl, May 18–19, 2020. Yaroslavl: Digital Printing House; 2020. p. 255–258. |
| [3] | Kaziev UK, Musaev US, Imankulova AS, Miklukhin DS. Experience with ultrasound cavitation in wound treatment in patients with purulent-necrotic complications of diabetic foot syndrome. Wounds and Wound Infections. Prof. B.M. Kostyuchenok Journal. 2024; 11(2): 28–34. |
| [4] | Kurlaev PP, Gritsenko VA, Belozertseva YuP. Antibacterial therapy of purulent-necrotic complications of diabetic foot syndrome. Kursk Scientific and Practical Bulletin “Man and His Health”. 2018; (4): 80–87. |
| [5] | Petrova VV, Spesivtsev YuA, Larionova VI, et al. Pathogenetic and clinical features of purulent-necrotic complications of diabetic foot syndrome. Vestnik Khirurgii im. I.I. Grekova. 2010; 169(2): 121–124. |
| [6] | Rundo AI, Volov IV. Clinical portrait of patients with diabetic foot syndrome complications. In: Surgery of Belarus at the Present Stage: Proceedings of the XVI Congress of Surgeons of the Republic of Belarus, Grodno, Nov 1–2, 2018. Part 2. Grodno: Grodno State Medical University; 2018. p. 145–150. |
| [7] | Cai W, Chong K, Huang Y, et al. Empagliflozin improves mitochondrial dysfunction in diabetic cardiomyopathy by modulating ketone body metabolism and oxidative stress. Redox Biology. 2024; 69: 103010. |
| [8] | Carnicelli P, Otsuki DA, Monteiro Filho A, et al. Effects of dexmedetomidine on hemodynamics, oxygenation, microcirculation, and inflammatory markers in a porcine model of sepsis. Acta Cirurgica Brasileira. 2022; 37(7): e370703. |
| [9] | Chiu C, Legrand M. Epidemiology of sepsis and septic shock. Current Opinion in Anaesthesiology. 2021; 34(2): 71–76. |
| [10] | Contou D, Painvin B, Daubin D, et al. Hemodynamic and neurological presentations of invasive meningococcal disease in adults: a nationwide study across more than 100 French ICUs. Intensive Care Medicine. 2025; 51(9): 1587–1602. |
| [11] | Feng Y, Peng JY, Peng Z. Blood purification in sepsis and systemic inflammation. Current Opinion in Critical Care. 2021; 27(6): 582–586. |
| [12] | Jeschke MG. The importance of sepsis in surgical patients. Surgery. 2023; 174(2): 398–399. |
| [13] | Leone M, Russell L, Cecconi M, et al. Hemodynamic failure during sepsis: what clinicians and researchers must know. Anaesthesia Critical Care & Pain Medicine. 2025; 101602. |
| [14] | Liu P, Zhang Z, Chen H, Chen Q. Pyroptosis: mechanisms and links with diabetic cardiomyopathy. Ageing Research Reviews. 2024; 94: 102182. |
| [15] | Liu Y, Liu L, Zhang J. Protective role of matrine in sepsis-associated cardiac dysfunction through regulation of the lncRNA PTENP1/miR-106b-5p axis. Biomedicine & Pharmacotherapy. 2021; 134: 111112. |
| [16] | Lorenzo-Almorós A, Cepeda-Rodrigo JM, Lorenzo Ó. Diabetic cardiomyopathy. Revista Clínica Española. 2022; 222(2): 100–111. |
| [17] | Martin L, Derwall M, Thiemermann C, Schürholz T. The heart in sepsis: molecular mechanisms, diagnosis and therapy of septic cardiomyopathy. Der Anaesthesist. 2017; 66(7): 479–490. |
| [18] | Mi Y, Yu H, Wang P, et al. Tyrosine hydroxylase-positive neurons in the rostral ventrolateral medulla mediate sympathetic activation in sepsis. Life Sciences. 2024; 358: 123118. |
| [19] | Nakamura K, Miyoshi T, Yoshida M, et al. Pathophysiology and treatment of diabetic cardiomyopathy and heart failure in patients with diabetes mellitus. International Journal of Molecular Sciences. 2022; 23(7): 3587. |
| [20] | Nordin NZ, Akhtar W, Ostermann M. Fluid management in sepsis-associated acute kidney injury. Journal of Critical Care. 2025; 90: 155188. |
| [21] | Wan H, Zhao S, Zeng Q, et al. Circular RNAs in diabetic cardiomyopathy. Clinica Chimica Acta. 2021; 517: 127–132. |
| [22] | Zhong X, Li H, Chen Q, et al. Association between different mean arterial pressure levels and 30-day mortality in sepsis patients: a propensity-score-matched retrospective cohort study. BMC Anesthesiology. 2023; 23(1): 116. |




 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML







