Nizamov I. U., Mamatkulov I. B.
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
To improve the quality of anesthetic management for endourological surgeries in children. The study is based on an analysis of the effectiveness of anesthesia in 137 children aged 1 to 14 years who underwent endourological surgeries at the clinic of the Tashkent Pediatric Medical Institute. All patients were divided into two groups depending on the anesthesia method used: Group 1 (119 children): Combined general anesthesia (CGA), Group 2 (18 children): Combined inhalation anesthesia (CIA). Research methods included pulse oximetry, monitoring of end-tidal carbon dioxide concentration, transthoracic echocardiography, electroencephalographic studies of brain activity, and analysis of stress hormone activity in the blood. EEG studies demonstrated the absence of significant suppression of the bioelectrical activity of the cerebral cortex, which explains the rapid and complete recovery of neuropsychological status after surgery. The course of surgical interventions under various combined anesthesia options was characterized by the absence of significant shifts in vital functions and the metabolism of the pediatric body. After surgery and anesthesia, the stress impact on the body ceased, and vital functions began to adapt to the new conditions created by the surgical correction. Combined anesthesia using ketamine in combination with propofol at an induction dose of 3.2±1.2 mg/kg and a maintenance dose of 4±0.5 mg/kg/hour provides effective protection of the child's body from surgical trauma during endourological procedures.
Keywords:
Combined general anesthesia (CGA), Electroencephalography (EEG), Hemodynamics, Propofol, Sevoflurane
Cite this paper: Nizamov I. U., Mamatkulov I. B., Comparative Evaluation of General Anesthesia Options in Endourological Surgeries in Children, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4540-4544. doi: 10.5923/j.ajmms.20251512.74.
1. Introduction
Intravenous anesthesia, since its introduction into practice, has always attracted the attention of clinicians and is perceived as a promising direction. At the current stage of anesthesiology development, existing general anesthesia methods possess many positive and effective qualities. However, the progress of pharmacological science and industry continues to provide new forms of anesthetics and hypnotics with expanded advantages over previously used drugs [1,2].Among modern drugs, propofol has specific positive properties that make it particularly suitable for surgical interventions in children. The availability of this drug brings us closer to ensuring the maximum safety of anesthesia, which undoubtedly enhances the quality of pediatric surgeries [3,4].One notable property of propofol is its comprehensive distribution in the body without adverse effects on metabolism, leading to effective action, excellent control of anesthesia depth, and smooth and rapid emergence from anesthesia. Administering a standard induction dose of propofol allows rapid penetration into the brain and tissues due to the high lipophilicity of the anesthetic. As a result, loss of consciousness occurs within 30 seconds [5,6].Propofol undergoes rapid metabolism in the body into desired components that provide sufficient and necessary effects. Total clearance exceeds renal blood flow (1.5 liters per minute) during both bolus and infusion administration. This suggests that extrahepatic metabolism of propofol may play a significant positive role. In the bloodstream, 98% of propofol is bound to plasma proteins [7,8].Propofol has been successfully used for short-term surgical interventions in recent years to maintain general anesthesia. It offers specific advantages over isoflurane and sevoflurane. Propofol's antiemetic effect is another critical aspect of its widespread use.Objective:To improve the quality of anesthetic management for endourological surgeries in children.
2. Materials and Methods
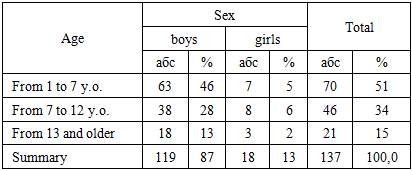
The study is based on an analysis of anesthesia effectiveness in 137 children aged 1 to 14 years who underwent endourological surgeries at the Tashkent Pediatric Medical Institute (Table 1).Table 1. Distribution of patients accordingly to age and sex
 |
| |
|
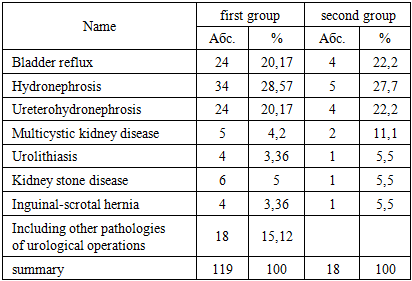
The majority of the examined patients belonged to the age group of 1 to 7 years. The maximum age of the patients was 14 years, while the minimum age was 1 year.The somatic status of the patients according to the ASA classification corresponded to classes I–II. Out of 137 children, 97 (70%) were in satisfactory condition before surgery, with their somatic status corresponding to ASA I, characterized by the absence of systemic disorders. Children with an ASA II somatic status accounted for 30%, which represents mild systemic disturbances without functional impairments (e.g., weight and height deficiency, slight hemoglobin reduction compared to age norms).All patients were divided into two groups depending on the anesthesia method used.Group 1 (119 children): Combined general anesthesia (CGA).Group 2 (18 children): Combined inhalation anesthesia (CIA).Table 2. Distribution of patients by the nature of surgical pathology
 |
| |
|
The studies were conducted at the following stages: baseline, premedication, induction, maintenance of anesthesia, and awakening period.Anesthesia Technique:The method involved combined general anesthesia using propofol in combination with ketamine. In Group 1, combined general anesthesia (CGA) was administered. Premedication was performed via intramuscular injection of atropine sulfate 0.1% (0.01 mg/kg), diphenhydramine 1% (0.1 mg/kg), sibazone 0.5% (0.3 mg/kg), and ketamine 5% (3 mg/kg). After 15–20 minutes, a peripheral vein was cannulated with a venous cannula. Induction was carried out by intravenous bolus injection of 1% propofol solution at a dose of 2 mg/kg. Five minutes later, a 5% ketamine solution was administered at a dose of 3 mg/kg. Oxygenation was provided through a facial mask with an oxygen flow rate of 1–3 L/min. Monitoring included blood pressure (BP), heart rate (HR), respiratory rate (RR) (PROTOCOL 102E, USA), SaO2, and ETCO2 (NORMACAP-200 DATEX/FINLAND).Maintenance of Anesthesia:An infusion of 1% propofol was administered via linear infusion pump (FRESENUS) at a dose of 150 mcg/kg/min, along with bolus administration of 5% ketamine at a dose of 1.5 mg/kg. Propofol infusion was discontinued after skin closure. All patients were transferred to the postoperative ward, where monitoring of vital functions continued. In the postoperative ward, the awakening level was assessed using the Kortilla and Aldrete scales.
3. Results
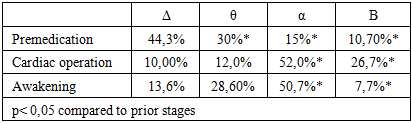
EEG studies during combined inhalation anesthesia did not reveal significant suppression of the bioelectrical activity of the cerebral cortex. It should be noted that the EEG dynamics correlated with the clinical signs of anesthesia depth. It was observed that changes in the frequency spectrum of the EEG occurred with the administration of minimal doses of the anesthetic (Table 3).Table 3
 |
| |
|
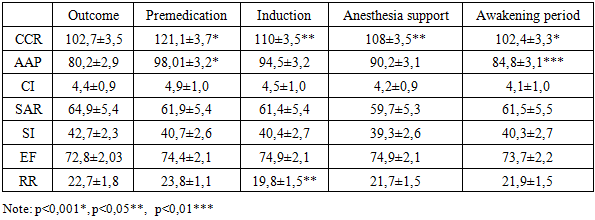
Thus, the EEG results indicate the absence of deep suppression of the bioelectrical activity of the cerebral cortex, which explains the rapid and complete recovery of neuropsychological status after the surgery.The considered method of combined general anesthesia with propofol and ketamine ensures effective protection of the child's body from surgical trauma. This is evidenced by stable EEG parameters and clinical observations during the surgical intervention.To evaluate the effectiveness of anesthetic management, the state of hemodynamic parameters was studied at different stages of the surgical procedure. The normative values for central and peripheral hemodynamics were based on data proposed by T.S. Agzamkhodzhaev et al. (2018), developed at the Tashkent Pediatric Medical Institute. For patients in Group 1, who underwent combined intravenous anesthesia (CIVA), changes in hemodynamic parameters during anesthesia for endourological surgeries are presented in Table 4. An increase in systolic arterial pressure (SAP) during the premedication period by 12% compared to baseline values was noted. However, during the induction stage, there was a statistically significant decrease in SAP (from 98.01±3.2 to 94.58±1.05), amounting to 5%. By the awakening stage, SAP decreased by 15% compared to the second stage and approached baseline values. The arterial hypertension observed during the premedication period was associated with the sympathomimetic effect of ketamine. Heart rate (HR) also showed significant fluctuations during the study stages. This trend was most pronounced during the premedication stage, with an HR increase of 11.8% (from 102.7±3.5 to 121.05±1.09). During the induction period, a statistically significant decrease in HR was observed (p<0.05). By the awakening stage, HR values became identical to baseline. The increase in HR was attributed to the effect of the anticholinergic drug, 0.1% atropine sulfate. troke volume index (SVI) increased by 7% after premedication. At other stages of anesthesia, SVI values were similar to baseline.Table 4. Hemodynamic parameters in patients with combined general anesthesia of propofol in combination with ketamine (М ± m)
 |
| |
|
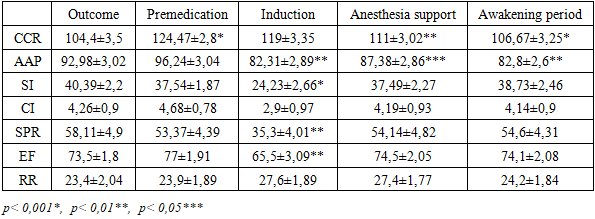
An increase in heart rate (HR) and stroke volume index (SVI) led to a rise in cardiac index (CI) in response to the increased oxygen demand of tissues during the premedication period. In the subsequent stages of the study, there was a tendency toward a decrease in CI. Total peripheral resistance (TPR) decreased by 9% in the second stage (from 64.8±2.4 to 61.9±1.2), but no statistically significant changes were observed throughout the anesthesia stages. A dynamic analysis of TPR during the anesthesia stages suggests that there were no values indicating peripheral vasodilation or vasoconstriction. The primary component of afterload, TPR, and its indicators remained practically unchanged during anesthesia stages.The contractility index (CI) remained high at all anesthesia stages, with no statistically significant changes in its values. During the induction stage, a statistically significant decrease in respiratory rate (RR) by 17% was noted, associated with the respiratory-depressive effect of propofol. No significant fluctuations were observed during other stages of anesthesia management. By the end of anesthesia, the RR reached levels comparable to baseline values.The hemodynamic parameters of patients in the second group, who underwent combined inhalation anesthesia with sevoflurane, are presented in Table 5. After premedication, a statistically significant increase in HR by 19% (from 104.4±3.5 to 124.47±2.81) was observed. During the induction, maintenance, and awakening periods, there was a tendency for HR to decrease and return to baseline levels. Systolic arterial pressure (SAP) increased by 4% (from 92.98±3.02 to 96.24±3.04) after premedication. In the induction period, SAP significantly decreased by 15% (p<0.01), and by the awakening period, SAP values remained unchanged.Table 5. Hemodynamic parameters in patients with combined inhalation anesthesia with sevoflurane (М ± m)
 |
| |
|
During the anesthesia stages, a tendency for SVI to decrease was observed. SVI decreased by 36% during the induction period compared to the premedication stage. CI decreased by 37% compared to the second anesthesia stage. A statistically significant reduction in CI (p<0.05) was noted during the third anesthesia stage compared to baseline values. The hemodynamic response to SVI reduction was manifested by a chronotropic compensatory increase in HR to maintain adequate CI.TPR also showed a tendency to decrease during the induction period by 35% compared to the second anesthesia stage, indicating a sharp drop in vascular resistance. The contractility index increased by 10% during the second stage of anesthesia compared to the first stage, while it decreased by 16% during the induction period. RR increased by 15% during the induction period compared to the premedication stage.The levels of cortisol, capnography, and the features of the awakening period during combined general anesthesia with propofol are being examined (Table 6). In response to surgical intervention, the body develops a protective and adaptive reaction aimed at ensuring the adaptation of vital functions under conditions of surgical stress. While reflex mobilizing actions lead to an increased release of essential regulatory hormones and energy-supplying metabolites into the bloodstream for working tissues, neurohumoral mechanisms contribute to tuning organs—effectors—to various levels of metabolic activity. The primary task of anesthetic management under these conditions is to prevent dangerous pathological reactions to surgical stress and to stabilize the functioning of vital systems and the body's metabolism.Table 6. Dynamics of cortisol content in the blood during surgical intervention in the conditions of CGA and CIA
 |
| |
|
The conducted studies showed that with the use of various combinations of anesthesia, the course of surgical interventions was characterized by the absence of significant shifts in the main vital functions and metabolism of the child’s body. The period of awakening from anesthesia is one of the most critical stages of general anesthesia, as important as the induction and maintenance phases. This is because, after the operation and anesthesia are finished, their stress impact on the body ceases, and vital functions begin to adapt to the new working conditions created by the surgical correction. We assert that the use of this new calculation system to determine the patient’s readiness for transportation from the operating room will reduce the need for nursing interventions to administer parenteral medications in the recovery room. In the first group, after the operation, the level of consciousness at the 10th minute of awakening with minimal stimulation was 1.0 ± 0.2 points; response to a simple question at the 20th minute was 1.9 ± 1.2 points; at the 40th minute, it was 2 points, which corresponded to clear consciousness and good orientation in space and time. Breathing at the 10th minute was 1.7 ± 0.4 points; patients at the 20th minute (1.7 ± 0.4 points) were able to take a deep breath. Recovery of psychomotor functions occurred quite quickly: at the 20th minute, it was 1.4 ± 0.4 points, and at the 40th minute, 1.9 ± 0.1 points after the operation. One patient experienced peripheral denervation in the postoperative period, lasting about 1 minute, with no negative impact on vital parameters (Table 7).Table 7. Changes in values during the awakening period after CGA
 |
| |
|
In the second group, the recovery time for the studied functions was slightly longer: the response to the first question after 40 minutes was 1.8 ± 0.4 points, and to the second question, more than 60 minutes after the operation. Hemodynamic indicators, such as blood pressure, were lower by 15% compared to baseline values. The mean arterial pressure at the 10th minute was 1.6 ± 0.4, and at the 40th minute, the total score reached 1.9 ± 0.2. Postoperative nausea and vomiting were observed in the CIA group at the 20th minute with a score of 0.9 ± 0.6; by the 40th minute, the score decreased to 0.7 ± 0.6 (Table 8).Table 8. Changes in values during the awakening period after CIA
 |
| |
|
4. Discussion
The use of combined general anesthesia with ketamine and propofol in children has been developed and scientifically substantiated for use in endourological surgeries. The possibility of adequate management of sedation depth during anesthesia and rapid, complication-free awakening during endourological operations in children has been established. The effectiveness of CGA with ketamine and propofol has been proven.
5. Conclusions
Combined anesthesia with ketamine and propofol, with an induction dose of 3.2 ± 1.2 mg/kg and a maintenance dose of 4 ± 0.5 mg/kg/hour, provides effective protection of the child’s body from surgical trauma during endourological operations. Combined anesthesia with ketamine and propofol has minimal impact on hemodynamics and bioelectrical activity (BEA) of the brain, which is characterized by a tendency to reduce heart rate and blood pressure by 3-5%. Anesthesia with the combination of ketamine and propofol allows for the preservation of spontaneous breathing during the surgical intervention. Propofol possesses qualities that make it close to the “ideal” intravenous anesthetic: rapid onset of drug-induced sleep, good anesthesia control, smooth awakening, and minimal postoperative complications.
References
| [1] | Babaev B.D., Povovarov S.A., Shishkov M.V. Diprivan as a Component of Anesthesia in Emergency Surgical Interventions in Children. Anesthesiology and Resuscitation. 1998; 1: 15–16. ISSN 0201-7563. |
| [2] | Babak S.I., Krutoy S.V., Zlochevsky A.N., Pantes A.V. Experience of Using the Drug Propofol-Novo in Our Clinic. MNS. 2014; 6(61). Available at: https://cyberleninka.ru/article/n/opyt-primeneniya-preparata-propofol-novo-v-nashey-klinike. |
| [3] | Vabishchevich A.V., Meshcheryakov A.V. Clinical Experience of Using the Intravenous Hypnotic Pofol (Propofol) During Anesthesia of Various Durations. Anesthesiology and Resuscitation. 2001; 3: 13–15. ISSN 0201-7563. |
| [4] | Dmitriev D.V. Use of Propofol in Children, or Some New Information About a Well-Known Drug. MNS. 2014; 6(61). Available at: https://cyberleninka.ru/article/n/ispolzovanie-propofola-u-detey-ili-nemnogo-novoy- informatsii-ob-izvestnom-preparate. |
| [5] | Kuklin V.N. Anesthesia in the One-Day Surgery Department: A Literature Review. Bulletin of Intensive Care Named After A.I. Saltanov. 2020; 4: 88–98. doi:10.21320/1818-474X-2020-4-88-98. |
| [6] | Potievskaya V.I., Zabolotskikh I.B., Gridchik I.E., Gritsan A.I., Eremenko A.A., Kozlov I.A., Lebedinsky K.M., Levit A.L., Mazurok V.A., Molchanov I.V., Nikolaenko E.M. Sedation of Patients in Intensive Care Units. Anesthesiology and Resuscitation. 2020; 5: 7–22. https://doi.org/10.17116/anaesthesiology20200517. |
| [7] | Satvaldieva E.A., Faiziev O.Ya., Yusupov A.S., Agzamova Sh.A., Faiziev Ya.N. Epidural Anesthesia as a Component of Postoperative Analgesia in Children. Russian Pediatric Journal. 2023; 26(3): 194–198. https://doi.org/10.46563/1560-9561-2023-26-3-194-198. EDN: eepvoj. |
| [8] | Sorokina E.Yu. Propofol in Modern Multicomponent General Anesthesia. MNS. 2014; 3(58). Available at: https://cyberleninka.ru/article/n/propofol-v-sovremennoy. |
| [9] | Sahib A., Yaqoob Q., Sasaa M., Atabi T., Radeef A. Evaluation of Pain When Using the Fentanyl–Propofol Combination: Two Dosage Levels. Bulletin of Anesthesiology and Resuscitation. 2023; 20(4): 40–45. https://doi.org/10.24884/2078-5658-2022-20-4-40-45. |
| [10] | Basaranoglu G., Erden V., Delatioglu H. Reduction of Pain on Injection of Propofol: A Comparison of Fentanyl with Remifentanil. Anesthesia and Analgesia. 2002; 94: 1040–1041. https://doi.org/10.1097/00000539-200204000-00053. |
| [11] | Cheong Mi A. Ephedrine Reduces the Pain from Propofol Injection. Anesthesia and Analgesia. 2002; 95(5): 1293–1296. https://doi.org/10.1097/00000539-200211000-00035. |
| [12] | Eriksson M., Englesson S., Niklasson F., et al. Effect of Lignocaine and pH on Propofol-Induced Pain. British Journal of Anaesthesia. 1997; 78: 502–506. https://doi.org/10.1093/bja/78.5.502. |
| [13] | Helmers J.H., Kraaijenhagen R.J., van Leeuwen L., et al. Reduction of Pain on Injection Caused by Propofol. Canadian Journal of Anaesthesia. 1990; 37: 267–268. https://doi.org/10.1007/BF03005485. |
| [14] | King S.Y., Davis F.M., Wells J.E., et al. Lidocaine for the Prevention of Pain Due to Propofol Injection. Anesthesia and Analgesia. 1992; 74: 246–249. https://doi.org/10.1213/00000539-199202000-00013. |
| [15] | Kizilcik N., Menda F., Bilgen S., et al. Effect of a Fentanyl–Propofol Mixture on Propofol Injection Pain: A Randomized Clinical Trial. Korean Journal of Anesthesiology. 2015; 68(6): 556–560. https://doi.org/10.4097/kjae.2015.68.6.556. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML






