-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4532-4535
doi:10.5923/j.ajmms.20251512.72
Received: Nov. 26, 2025; Accepted: Dec. 20, 2025; Published: Dec. 25, 2025

Risk Stratification of Neurological Adverse Events Following COVID-19 Vaccination Using Clinical Scales and Biomarkers
Javlon Salimjonov1, Nilufar Rashidova2, Khanifa Khalimova3
1PhD student, Tashkent State Medical University, Tashkent, Uzbekistan
2DSc, Associate Professor, Department of Neurology and Medical Psychology, Tashkent, Uzbekistan
3DSc, Professor, Department of Neurology and Medical Psychology, Tashkent, Uzbekistan
Correspondence to: Javlon Salimjonov, PhD student, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Mass vaccination against SARS-CoV-2 has significantly reduced COVID-19–related morbidity and mortality worldwide. Although neurological adverse events following immunization are rare, their timely identification and clinical assessment remain challenging due to the absence of validated risk stratification tools. Methods: This observational case–control study included 140 vaccinated individuals: 70 patients with post-vaccination neurological complications and 70 vaccinated controls without neurological manifestations. Clinical assessment was performed using WHODAS 2.0, Hospital Anxiety and Depression Scale (HADS), and COMPASS-31. Serum levels of interleukin-6 (IL-6) and syndecan-1 (SDC-1) were measured using enzyme-linked immunosorbent assay. Statistical analysis included group comparisons and correlation analysis. Results: Patients with post-vaccination neurological complications demonstrated significantly higher scores on WHODAS 2.0, HADS, and COMPASS-31 compared to controls (p < 0.05). Serum IL-6 and SDC-1 levels were also significantly elevated in the affected group and showed positive correlations with clinical severity indices. Based on combined clinical and laboratory findings, a biomarker-assisted risk stratification model was proposed to identify individuals at higher risk of clinically significant neurological adverse events. Conclusion: The integration of standardized clinical scales with neuroimmune and endothelial biomarkers provides a feasible approach for early risk stratification of neurological adverse events following COVID-19 vaccination. This strategy may enhance post-vaccination monitoring and support individualized clinical management without undermining the overall safety of vaccination programs.
Keywords: COVID-19 vaccination, Neurological adverse events, Risk stratification, Interleukin-6, Syndecan-1, Neuroinflammation, Endothelial dysfunction
Cite this paper: Javlon Salimjonov, Nilufar Rashidova, Khanifa Khalimova, Risk Stratification of Neurological Adverse Events Following COVID-19 Vaccination Using Clinical Scales and Biomarkers, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4532-4535. doi: 10.5923/j.ajmms.20251512.72.
1. Introduction
- The coronavirus disease 2019 (COVID-19) pandemic has resulted in a substantial global burden of morbidity and mortality and has posed unprecedented challenges to healthcare systems worldwide. Mass vaccination against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has been widely recognized as the most effective public health intervention to control the pandemic, significantly reducing severe disease, hospitalization, and mortality across diverse populations [1–3]. As a result, billions of vaccine doses have been administered globally, necessitating robust post-marketing surveillance systems to monitor vaccine safety in real-world conditions [4].Neurological complications associated with COVID-19 vaccination have been reported rarely and occur at a markedly lower frequency than neurological manifestations observed after SARS-CoV-2 infection [5–7]. Nevertheless, reported post-vaccination neurological adverse events include a broad spectrum of central and peripheral nervous system disorders, such as Guillain–Barré syndrome, transverse myelitis, encephalitis, stroke-like syndromes, and cranial neuropathies [8–10]. Although causality is often difficult to establish, these conditions may lead to transient or persistent functional impairment, underscoring the importance of timely recognition and appropriate clinical management.Current pharmacovigilance frameworks primarily focus on the detection, classification, and reporting of adverse events following immunization (AEFI), as recommended by the World Health Organization and regulatory agencies [4,11]. However, these systems provide limited guidance for individual-level risk stratification or early prognostic assessment in clinical practice. Neurologists are frequently faced with heterogeneous clinical presentations, making it challenging to distinguish benign, self-limited symptoms from conditions requiring closer monitoring or targeted intervention.Emerging evidence suggests that immune-mediated inflammation and endothelial dysfunction may represent key biological mechanisms underlying neurological adverse events following immune stimulation, including vaccination [12–14]. Interleukin-6 (IL-6) is a central pro-inflammatory cytokine involved in acute-phase responses, cytokine signaling cascades, blood–brain barrier permeability, and neuroimmune interactions. Elevated IL-6 levels have been associated with neuroinflammatory processes and adverse neurological outcomes in various infectious and immune-mediated conditions [15,16].Syndecan-1 (SDC-1), a transmembrane proteoglycan and a major component of the endothelial glycocalyx, has emerged as a sensitive marker of endothelial injury and vascular dysfunction. Increased circulating SDC-1 levels reflect glycocalyx degradation and have been linked to systemic inflammation, microvascular damage, and impaired vascular integrity, all of which may contribute to neurovascular and neuroinflammatory pathology [17–19]. The combined assessment of IL-6 and SDC-1 may therefore provide complementary insights into inflammatory and endothelial pathways relevant to neurological complications.In parallel, standardized clinical instruments such as the World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0), the Hospital Anxiety and Depression Scale (HADS), and the Composite Autonomic Symptom Score (COMPASS-31) allow for objective quantification of functional disability, psycho-emotional disturbances, and autonomic dysfunction [20–22]. Despite their widespread validation and use in neurological research, these scales are rarely integrated with biological markers in post-vaccination safety studies.Given the absence of clinically applicable tools that combine functional assessment with pathophysiologically relevant biomarkers, there is a clear need for integrated risk stratification approaches. The present study aimed to evaluate a combined clinical and biomarker-based model for early risk stratification of neurological adverse events following COVID-19 vaccination. By integrating standardized clinical scales with serum levels of IL-6 and SDC-1, this study seeks to provide a practical framework for improving post-vaccination neurological monitoring while reinforcing the overall safety and public health benefits of COVID-19 vaccination programs.
2. Materials and Methods
- This observational case–control study included 80 vaccinated individuals. The case group consisted of 40 patients who developed neurological symptoms following COVID-19 vaccination, while 40 vaccinated individuals without neurological manifestations served as controls. Participants were recruited from neurological inpatient and outpatient settings. Inclusion criteria for the case group were documented COVID-19 vaccination and new-onset neurological symptoms temporally associated with vaccination. Individuals with active infection, autoimmune or neurodegenerative diseases, malignancy, or acute systemic inflammatory conditions were excluded. The study was conducted in accordance with the Declaration of Helsinki, and written informed consent was obtained from all participants. All participants underwent standardized neurological examination. Functional disability was assessed using WHODAS 2.0 (12-item version). Psycho-emotional status was evaluated with the Hospital Anxiety and Depression Scale (HADS), and autonomic dysfunction was assessed using the COMPASS-31 questionnaire. Serum levels of interleukin-6 (IL-6) and syndecan-1 (SDC-1) were measured using enzyme-linked immunosorbent assay. Neuroimaging studies (MRI or CT) were performed in the case group when clinically indicated to exclude alternative causes of neurological symptoms. Continuous variables were expressed as mean ± standard deviation or median with interquartile range. Group comparisons and correlation analyses were performed using appropriate statistical tests. A p-value < 0.05 was considered statistically significant.
3. Results and Discussion
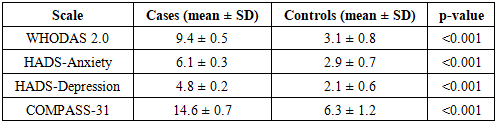
- A total of 80 vaccinated individuals were included in the analysis: 40 patients with post-vaccination neurological complications (case group) and 40 vaccinated individuals without neurological symptoms (control group).The mean age in the case group was 44.8 ± 11.6 years, compared to 43.9 ± 10.9 years in the control group (p = 0.64). Sex distribution was comparable between groups (female: 58.6% vs 55.7%, p = 0.72). Neurological symptoms in the case group developed within a median of 12 days (IQR: 7–18) after vaccination. Central nervous system involvement was observed in 41.4% of cases, while 58.6% presented with peripheral or mixed neurological manifestations. Neuroimaging performed when clinically indicated revealed no alternative acute structural pathology. Functional disability assessed by WHODAS 2.0 was significantly higher in patients with neurological complications (9.4 ± 0.5) compared with controls (3.1 ± 0.8, p < 0.001). Psycho-emotional assessment demonstrated higher anxiety and depressive symptom burden in the case group. Mean HADS-Anxiety score was 6.1 ± 0.3 in patients versus 2.9 ± 0.7 in controls (p < 0.001), while HADS-Depression scores were 4.8 ± 0.2 and 2.1 ± 0.6, respectively (p < 0.001). Autonomic dysfunction assessed using COMPASS-31 was more pronounced in the case group, with a mean score of 14.6 ± 0.7, compared to 6.3 ± 1.2 in controls (p < 0.001), indicating a significantly higher autonomic symptom burden among affected individuals.
|
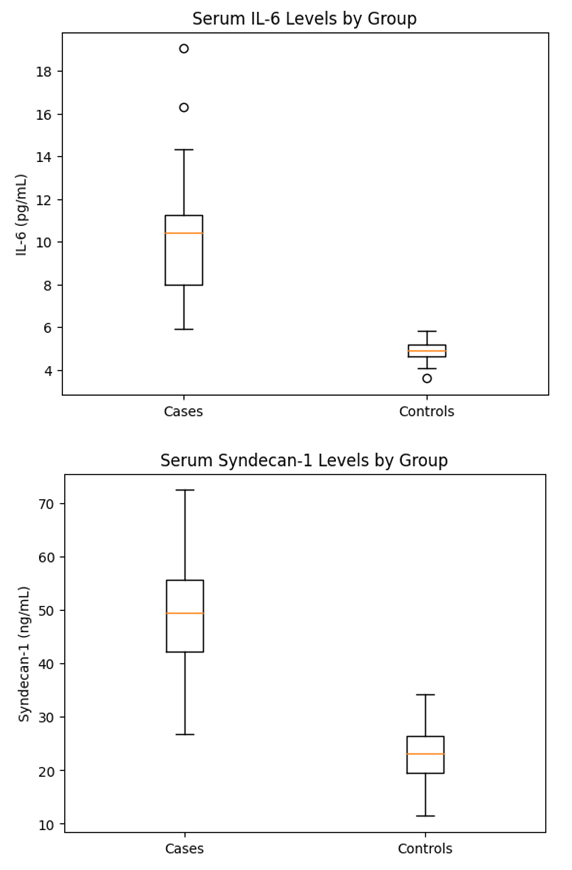 | Figure 1. Serum IL-6 and SDC-1 levels in cases and controls |
4. Conclusions
- In this observational case–control study, neurological adverse events following COVID-19 vaccination were associated with increased functional impairment, psycho-emotional disturbances, and autonomic dysfunction, as reflected by standardized clinical assessment tools. Patients with post-vaccination neurological manifestations demonstrated higher WHODAS 2.0, HADS, and COMPASS-31 scores compared with vaccinated controls without neurological symptoms. These clinical findings were accompanied by elevated serum levels of interleukin-6 and syndecan-1, indicating the involvement of inflammatory and endothelial mechanisms. The observed associations between biomarker levels and clinical severity support the role of neuroimmune activation and endothelial dysfunction in the development of post-vaccination neurological symptoms.Importantly, the identified neurological adverse events were rare and occurred at a substantially lower frequency and severity than neurological complications associated with SARS-CoV-2 infection itself. The results of this study do not challenge the established safety profile of COVID-19 vaccines but rather emphasize the importance of systematic post-vaccination monitoring in a small subset of individuals. COVID-19 vaccination remains a safe, effective, and essential public health intervention for reducing severe disease, hospitalization, and mortality. The proposed clinical and biomarker-based risk stratification approach may enhance vaccine safety surveillance, support individualized patient care, and further strengthen public confidence in vaccination programs.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML