-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4527-4531
doi:10.5923/j.ajmms.20251512.71
Received: Nov. 28, 2025; Accepted: Dec. 16, 2025; Published: Dec. 22, 2025

To Study the Role of the LYS198ASN Polymorphism of the END1 Regulatory Gene in the Development of Chronic Heart Failure and Renal Dysfunction in Individuals of Uzbek Ethnicity
Zakirova Gulnoza1, Masharipova Dilyafruz1, Boboev Qodirjon2, Tagaeva Dilnoza1
1“Republican Specialized Scientific - Practical Medical Center of Therapy and Medical Rehabilitation”, Tashkent, Uzbekistan
2Republican Center for Scientific and Practical Medicine of Specialized Hematology, Tashkent, Uzbekistan
Correspondence to: Tagaeva Dilnoza, “Republican Specialized Scientific - Practical Medical Center of Therapy and Medical Rehabilitation”, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Chronic heart failure (CHF) and associated renal dysfunction represent significant medical and social challenges, particularly given the increasing prevalence of cardiovascular disease. Endothelial dysfunction is a key factor in the pathogenesis of CHF. Molecular genetic factors play a significant role in the development of this disease. Recently, particular attention has been paid to the Lys198Asn polymorphism of the endothelin-1 (END1) gene, which controls vascular tone and is involved in fibrosis and inflammation. Studying the clinical significance and prevalence of the Lys198Asn polymorphism among Uzbeks with CHF and renal dysfunction will help us better understand the genetic mechanisms underlying the disease and develop personalized methods for early diagnosis and prognosis. Chronic heart failure (CHF) and associated renal dysfunction, defined as a decrease in glomerular filtration rate (GFR) and/or the presence of albuminuria corresponding to chronic kidney disease stages 1–3, represent significant medical and social challenges, particularly given the increasing prevalence of cardiovascular disease. Endothelial dysfunction is a key factor in the pathogenesis of CHF. Molecular genetic factors play a significant role in the development and progression of this disease. Recently, particular attention has been paid to the Lys198Asn polymorphism of the endothelin-1 (END1) gene, which regulates vascular tone and is involved in fibrotic and inflammatory processes. Studying the clinical significance and prevalence of this polymorphism among Uzbeks with CHF and renal dysfunction will help better understand the genetic mechanisms underlying cardiorenal interactions and develop personalized approaches for early diagnosis and prognosis.
Keywords: Renal dysfunction, Clinical and humoral aspects, Genetic aspects, Polymorphism, CHF
Cite this paper: Zakirova Gulnoza, Masharipova Dilyafruz, Boboev Qodirjon, Tagaeva Dilnoza, To Study the Role of the LYS198ASN Polymorphism of the END1 Regulatory Gene in the Development of Chronic Heart Failure and Renal Dysfunction in Individuals of Uzbek Ethnicity, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4527-4531. doi: 10.5923/j.ajmms.20251512.71.
1. Introduction
- Among the many factors contributing to renal dysfunction, endothelial dysfunction plays a significant role and often underlies the development of renal failure. A thorough understanding of the clinical, humoral, and genetic mechanisms of endothelial dysfunction in renal pathologies is crucial for the development of practical diagnostic and therapeutic approaches in nephrology [6].Chronic kidney disease (CKD) is not only a localized kidney lesion but also a systemic disorder affecting multiple organs and physiological systems, including the cardiovascular system [7]. Endothelial dysfunction, oxidative stress, and chronic low-grade inflammation are central mechanisms in CKD progression. The interaction and imbalance of these processes aggravate structural and functional renal damage and promote the development of cardiovascular and metabolic complications. In this context, humoral and genetic factors substantially influence disease onset and progression, highlighting the importance of an integrated diagnostic and therapeutic approach [14]. Endothelial dysfunction represents an imbalance between vasodilatory and vasoconstrictive mechanisms regulated by endothelial cells lining the vascular wall. In kidney disease, this imbalance manifests through altered vascular tone, enhanced inflammatory activation, increased oxidative stress, and a prothrombotic state, all of which contribute to progressive renal injury. Humoral factors play a key role in regulating endothelial function in the renal microenvironment. An imbalance of vasoactive substances, including nitric oxide, endothelin-1, prostaglandins, and reactive oxygen species, disrupts endothelial homeostasis and exacerbates renal tissue damage [10].In our review of the scientific literature, we sought to elucidate the complex relationships among clinical, humoral, and genetic factors that influence endothelial dysfunction in kidney disease. We believe that in-depth studies of the molecular mechanisms, clinical manifestations, and genetic predisposition to endothelial dysfunction can facilitate the development of new therapeutic strategies and the application of precision medicine principles to improve patient care [11].A comprehensive understanding of endothelial dysfunction in renal disease is essential for improving diagnostic approaches, treatment efficacy, and prognostic assessment [2]. The integration of clinical, humoral, and genetic data creates opportunities to develop personalized therapeutic strategies to correct endothelial disorders and optimize patient management. Endothelial dysfunction is a key pathogenetic factor in both cardiovascular and renal diseases. Endothelin-1, regulated in part by the NOS3 gene, is a critical mediator of endothelial activity. An imbalance between endothelin-1, a potent vasoconstrictor, and nitric oxide, a vasodilator, contributes to disease progression in the cardiorenal continuum [18]. Endothelial dysfunction begins early in CKD, worsens with disease progression, and ultimately contributes to end-stage renal disease (ESRD) [15].Various factors, such as oxidative stress, inflammation, hyperglycemia, and genetic predisposition, can disrupt these functions. In the context of kidney disease, endothelial dysfunction is particularly significant, as impaired vascular regulation can exacerbate renal tissue damage and accelerate the progression of chronic kidney disease [3].Hemodialysis is vital for the treatment of end-stage renal failure, but its effectiveness is primarily determined by the condition of the vascular endothelium and endothelin-1 (ET-1) levels. Endothelin is a potent vasoconstrictor synthesized by endothelial cells and plays a key role in regulating vascular tone and hemodynamic balance. In chronic renal failure, endothelin-1 levels are typically elevated, contributing to vasoconstriction, hypertension, and impaired tissue perfusion. In patients undergoing hemodialysis, elevated ET-1 levels are associated with endothelial dysfunction, inflammation, and an increased risk of cardiovascular complications. Genetic variations in the EDN1 gene, which encodes endothelin-1, can affect its expression level and biological activity [4].
2. Objective
- The objective of this study was to evaluate the distribution of allelic and genotypic variants of the Lys198Asn polymorphism of the END1 gene and to assess their relationship with glomerular filtration rate (GFR) levels in patients with chronic heart failure.
3. Methods
- The study included 200 patients with CHF. Of these, 110 had a GFR greater than 60 ml/min/1.73 m², while 90 patients had a GFR less than 60 ml/min/1.73 m². DNA samples from 120 apparently healthy Uzbek donors with no signs of cardiovascular or renal dysfunction served as controls.The Lys198 polymorphism of the END1 gene was determined using test systems from Scientific Production Company Litekh (Russia) according to the manufacturer's protocol. Amplification of a polymorphic region located in the promoter region of the END1 gene was performed using a Rotor Gene Q thermal cycler (Quagen, Germany).The polymerase chain reaction (PCR) was performed in a 25 µl volume. It included the following steps: denaturation at 95°C for 5 minutes, 35 cycles of denaturation at 95°C (30 seconds), primer annealing at 60°C (30 seconds), DNA synthesis at 72°C (1 minute), and a final extension step at 72°C for 10 minutes.The obtained data were analyzed using the OpenEpi v9.2 statistical package.
4. Results
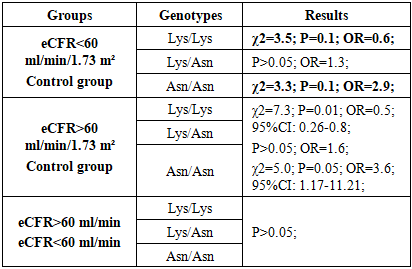
- In the studied patient and control samples, the observed genotype distributions for this locus in both groups correspond to those expected under the Hardy-Weinberg equilibrium. Analysis of the Lys198 Asn polymorphism frequency in the END1 gene in the study group, subgroups with different SCFs, and the control sample demonstrates a tendency toward differences in the frequencies of allelic and genotypic variants.In the study group and the control sample, the Lys and Asn allele frequencies were 74.0% and 26.0%, respectively, compared with 84.6% and 20.8%, respectively (χ2=5.5, p=0.03). The relative risk of detecting a mutant Asn allele among patients was OR=1.7 (95% TI: 1.09–2.27) compared to controls. Consequently, the Lys198 Asn polymorphism in the END1 gene is associated with an increased risk of developing renal dysfunction in patients with CHF.
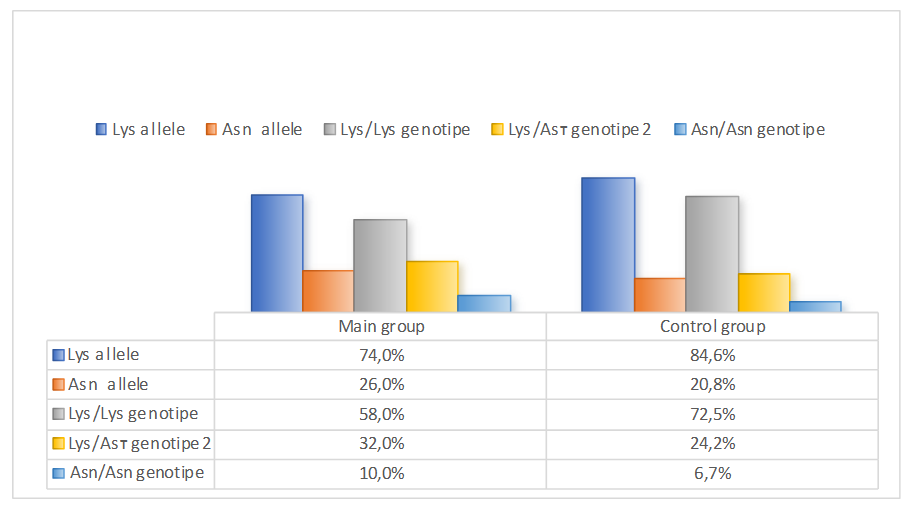 | Diagram 1. Differences in the frequency of allelic and genotypic variants of the END1 (Lys198) gene polymorphism in patients with CHF |
|
5. Discussion
- Investigating the role of the Lys198Asn polymorphism in the END1 (endothelin-1) gene in the development of chronic heart failure (CHF) and renal dysfunction in individuals of Uzbek ethnicity is a relevant area of modern cardionephrology. Endothelin-1 is one of the most potent vasoconstrictors produced by endothelial cells and plays a key role in regulating vascular tone, systemic and renal hemodynamics, and myocardial remodeling. Dysregulation of endothelin-1 signaling leads to persistent vasoconstriction, arterial hypertension, inflammation, and fibrosis, which contribute to the progression of both cardiac and renal failure [16,17].The Lys198Asn (rs5370) polymorphism of the END1 gene is known to affect endothelin-1 expression and biological activity. The Asn allele has been associated with elevated plasma endothelin-1 levels, increased peripheral vascular resistance, and activation of the renin–angiotensin–aldosterone system (RAAS). These mechanisms promote myocardial hypertrophy, interstitial fibrosis, endothelial dysfunction, and deterioration of glomerular filtration in patients with CHF, thereby reinforcing the cardiorenal interaction [8,16].The results obtained in the present study are consistent with previously published data. Nepal G. et al. (2019) demonstrated a significant association of the Asn198 allele with an increased risk of ischemic stroke and arterial hypertension in Asian populations [9]. Similarly, Gupta A. et al. (2023) reported that carriage of the Asn198 allele in the Indian population was associated with elevated endothelin-1 levels and left ventricular hypertrophy, indicating its adverse cardiovascular effects [5]. Comparable findings were reported by Zhang Y. et al. (2011), who showed that the Asn allele was associated with reduced glomerular filtration rate and more pronounced endothelial dysfunction in patients with chronic kidney disease [13]. At the same time, Annapareddy S.N. et al. (2015) did not identify a significant effect of the Lys198Asn polymorphism on the progression of chronic kidney disease in patients with autosomal dominant polycystic kidney disease, underscoring the context-dependent nature of this genetic variant. These discrepancies suggest that the impact of the Lys198Asn polymorphism may be modified by underlying disease mechanisms, gene–gene interactions, and environmental factors [1]. Of particular interest is the ethnic distribution of the END1 gene polymorphism. The reported frequency of the Asn198 allele varies across populations, ranging from approximately 15–20% in Europeans to 25–30% in Asian populations. According to our data, the frequency of the Asn198 allele in the Uzbek population was 26%, comparable to that observed in East Asian cohorts and supporting the hypothesis of population-specific genetic susceptibility. These findings indicate that, in Uzbeks, carriage of the Asn198 allele may contribute to an increased risk of reduced glomerular filtration rate in patients with CHF [1,9,13]. In addition to genetic predisposition, endothelial dysfunction itself plays a critical role in the progression of renal impairment. Elevated endothelin-1 levels promote vascular stiffness, impaired renal microcirculation, and oxidative stress, further exacerbating endothelial injury. Yilmaz M.I. et al. (2006) demonstrated that increased levels of asymmetric dimethylarginine (ADMA) and oxidative stress are key contributors to endothelial dysfunction in chronic kidney disease, leading to reduced nitric oxide bioavailability and accelerated vascular damage [12]. These mechanisms may act synergistically with genetic factors, including the Lys198Asn polymorphism, amplifying endothelial and renal dysfunction in CHF. Overall, the findings of the present study support the hypothesis that the Lys198Asn polymorphism of the END1 gene plays a significant pathogenetic role in the development of renal dysfunction in patients with chronic heart failure. The contribution of this polymorphism appears to be influenced by ethnic background and clinical characteristics, emphasizing its relevance in the Uzbek population. The results highlight the potential value of END1 genotyping for early risk stratification and the development of personalized therapeutic approaches in patients with CHF and concomitant renal dysfunction.
6. Conclusions
- The results of this study indicate that the Lys198Asn polymorphism of the END1 gene is significantly associated with renal dysfunction in patients with chronic heart failure. A higher prevalence of the mutant Asn allele and the Asn/Asn genotype was observed in patients with reduced GFR, suggesting their contribution to impaired renal function and endothelial dysfunction.Functionally, the Lys198Asn polymorphism may influence END1 gene activity and endothelin-1 synthesis, leading to enhanced vasoconstriction, microcirculatory disturbances, and progression of endothelial dysfunction. These mechanisms can aggravate both renal impairment and the clinical course of chronic heart failure, supporting the role of this polymorphism in the cardiorenal continuum.The findings highlight the potential clinical value of genetic testing for the Lys198Asn polymorphism as a prognostic marker for early identification of patients at increased risk of renal dysfunction and for implementing personalized therapeutic strategies. Further prospective studies are warranted to clarify its prognostic significance in chronic heart failure, hypertension, and metabolic disorders, as well as its potential utility in clinical practice.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML