-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4522-4526
doi:10.5923/j.ajmms.20251512.70
Received: Nov. 18, 2025; Accepted: Dec. 10, 2025; Published: Dec. 22, 2025

Dynamics of Clinical and Laboratory Indicators in Patients with Non-Alcoholic Fatty Liver Disease Depending on the Stage of Fibrosis and Ongoing Therapy
Abdigaffor Gadayev1, Saodat Agzamkhodjaeva1, Nuriddin Nuritdinov1, Nigora Pirmatova2
1Tashkent State Medical University, Tashkent, Uzbekistan
2National Medical Center, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Non-alcoholic fatty liver disease (NAFLD) is one of the most widespread chronic hepatological disorders globally and is closely linked to metabolic syndrome, cardiovascular pathology, and systemic inflammatory responses. The progression from simple steatosis to fibrosis represents a critical stage associated with increased risks of metabolic decompensation and cardiovascular complications. This study aimed to analyze the dynamics of clinical, biochemical, and instrumental parameters in male patients with NAFLD depending on the stage of fibrosis and the effectiveness of comprehensive therapy. A total of 206 Uzbek men diagnosed with NAFLD were examined using clinical evaluation, laboratory markers, and non-invasive diagnostics in accordance with international EASL–EASD–EASO recommendations. Baseline data showed a consistent increase in blood pressure, liver cytolysis enzymes (ALT, GGT), low-density lipoproteins (LDL), fatty liver index (FLI), and Young’s modulus measurements with increasing fibrosis stage (F0–F2). These findings confirm that fibrogenesis is closely associated with oxidative stress, dyslipidemia, altered vascular reactivity, and deterioration of liver tissue elasticity. Following a two-month course of combination therapy including the hepatoprotective agent Phosphogliv Urso and the ACE inhibitor lisinopril, significant improvements were observed across all fibrosis groups. Notably, systolic and diastolic blood pressure decreased, ALT and GGT levels were reduced, LDL concentrations declined, HDL levels improved, and Young’s modulus values indicated decreased liver stiffness. Additionally, the FLI demonstrated a marked reduction, reflecting decreased hepatic steatosis. The positive therapeutic dynamics support the hypothesis that targeted treatment aimed at controlling metabolic and inflammatory processes in NAFLD exerts not only hepatoprotective but also cardioprotective effects. The results underscore the importance of early detection of fibrosis progression and the inclusion of comprehensive therapy to prevent cardiovascular complications. The study highlights ALT, GGT, LDL, FLI, and Young’s modulus as informative markers for monitoring treatment efficacy and disease progression. These findings contribute to the growing evidence supporting an integrated hepatology–cardiology approach to NAFLD management.
Keywords: Non-alcoholic fatty liver disease (NAFLD), Liver fibrosis, Steatosis, Metabolic syndrome, Cardiovascular risk, Hypertension, ALT, GGT, LDL, HDL, Young’s modulus, Liver stiffness
Cite this paper: Abdigaffor Gadayev, Saodat Agzamkhodjaeva, Nuriddin Nuritdinov, Nigora Pirmatova, Dynamics of Clinical and Laboratory Indicators in Patients with Non-Alcoholic Fatty Liver Disease Depending on the Stage of Fibrosis and Ongoing Therapy, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4522-4526. doi: 10.5923/j.ajmms.20251512.70.
1. Introduction
- Non-alcoholic fatty liver disease (NAFLD) is one of the most common chronic liver conditions and is closely associated with metabolic syndrome and its components (arterial hypertension, obesity, dyslipidemia, and hyperglycemia). In recent years, the incidence of NAFLD has been steadily increasing, making it a significant medical and social concern. Particular attention is being paid to the association between NAFLD and cardiovascular diseases, which are the leading cause of mortality in this patient population [3,4,6].Despite significant progress in understanding the pathogenesis of NAFLD, the mechanisms of liver fibrosis progression and its relationship with lipid metabolism disorders, as well as the expression of key receptor genes involved in lipoprotein metabolism and atherogenesis, remain insufficiently studied. Identifying these associations is of great clinical importance, as it allows for the optimization of diagnostics, disease course prognosis, and the development of personalized strategies for the prevention of cardiovascular complications.
2. Materials and Methods
- To address the objectives outlined in this study, 206 Uzbek male patients with non-alcoholic fatty liver disease (NAFLD) who received inpatient treatment at the Department of Therapy of the Republican Scientific-Practical Medical Center for Therapy and Medical Rehabilitation (RSPMCTR and MR) during 2023–2024 were examined. The average age of the patients was 46.7 ± 0.57 years. The diagnosis of NAFLD was based on patient complaints, medical history, objective findings, and laboratory-instrumental assessments. The disease was diagnosed and evaluated in accordance with the recommendations of the European Association for the Study of the Liver (EASL), the European Association for the Study of Diabetes (EASD), and the European Association for the Study of Obesity (EASO).
3. Results
- As NAFLD progresses, not only do morphofunctional changes occur in the liver, but systemic disorders affecting the cardiovascular system also develop. One important clinical aspect is the dynamics of blood pressure (BP), which reflect the extent of vascular regulation involvement in the pathological process.In patients with NAFLD at the initial stage of fibrosis (F0), BP values remained within the physiological norm: systolic BP averaged 119.6 mmHg, and diastolic BP was 78.7 mmHg. In patients at the F1 stage, sub-hypertensive increases in BP were observed (SBP: 132.1 mmHg; DBP: 85.2 mmHg), indicating the onset of vascular regulation disturbances. At the F2 stage, values corresponding to grade 1 arterial hypertension were recorded (SBP: 142.3 mmHg; DBP: 90.3 mmHg), confirming cardiovascular system involvement in the pathological process.As part of the comprehensive treatment, the hepatoprotective drug Phosphogliv Urso (1 tablet twice a day) was used, along with the angiotensin-converting enzyme (ACE) inhibitor lisinopril (5 mg once a day) for antihypertensive purposes, over a two-month period. Lisinopril was selected because it is excreted unchanged by the kidneys in its active form, without undergoing hepatic metabolism.All patient groups showed a statistically significant reduction in blood pressure:- F0 stage: Systolic BP decreased to 110.1 mmHg, and diastolic BP to 70.5 mmHg.- F1 stage: Systolic BP decreased to 120.5 mmHg, and diastolic BP to 75.4 mmHg.- F2 stage: Systolic BP decreased to 121.7 mmHg, and diastolic BP to 77.6 mmHg.These results confirm the effectiveness of the combined therapy, including the use of Phosphogliv Urso and lisinopril, in reducing blood pressure and potentially mitigating cardiovascular risks in patients with NAFLD at various stages of liver fibrosis.These changes indicate the effectiveness of the ongoing therapy in controlling blood pressure even in patients with advanced liver fibrosis. The likely mechanisms of improvement include the reduction of systemic inflammation, normalization of the metabolic profile, and improvement of vascular tone due to the comprehensive approach to treating NAFLD. Considering the high risk of cardiovascular complications with progressive fibrosis, especially at stages F1–F2, regular blood pressure monitoring and the use of antihypertensive therapy should be regarded as important components of managing patients with NAFLD.Thus, regular blood pressure monitoring and the inclusion of antihypertensive therapy as part of the comprehensive treatment for patients with NAFLD are particularly relevant at fibrosis stages F1–F2, when the risk of cardiovascular complications increases.As is well known, alanine aminotransferase (ALT) is one of the key enzymes reflecting the degree of cytolytic damage to hepatocytes. Elevated activity of this enzyme is characteristic of inflammatory and destructive processes in the liver, making this indicator an important marker of non-alcoholic steatohepatitis activity.
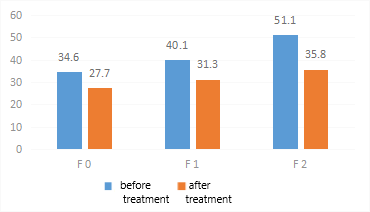 | Figure 1. Changes in alanine aminotransferase (ALT) levels before and after treatment depending on the stage of liver fibrosis |
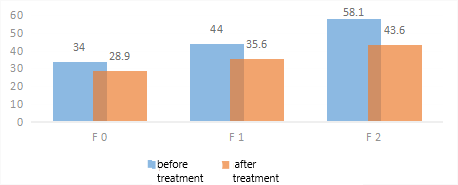 | Figure 2. Dynamics of gamma-glutamyltransferase (GGT) levels in patients with non-alcoholic fatty liver disease during therapy |
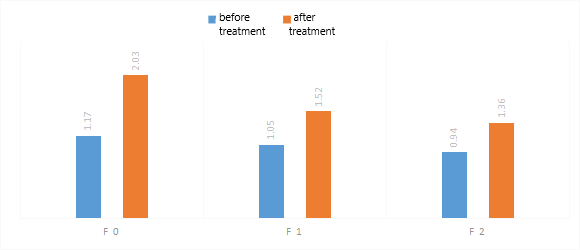 | Figure 3. Dynamics of low-density lipoproteins (LDL) during therapy |
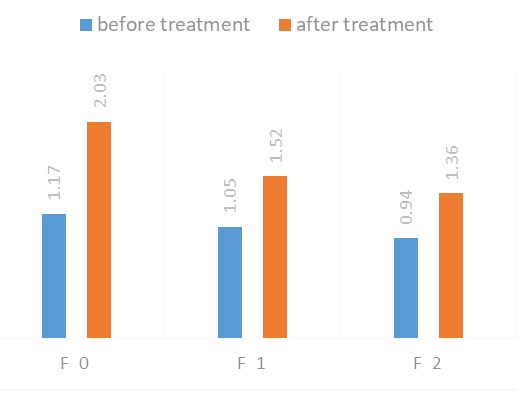 | Figure 4. Dynamics of high-density lipoprotein (HDL) levels in patients with non-alcoholic fatty liver disease during therapy |
4. Discussion
- The findings of this study clearly demonstrate that the progression of liver fibrosis in patients with non-alcoholic fatty liver disease (NAFLD) is closely intertwined with significant alterations in cardiovascular function, metabolic status, and biochemical indicators. As fibrosis advances from stage F0 to F2, patients exhibit a steady increase in systolic and diastolic blood pressure, reflecting early disturbances in vascular tone regulation and endothelial function. These results are consistent with previously published data indicating that fibrosis itself serves as an independent predictor of cardiovascular risk and that even mild fibrotic changes may contribute to subclinical hemodynamic abnormalities. The observed blood pressure reductions following therapy across all fibrosis stages once again highlight the need to incorporate antihypertensive management into the routine care of NAFLD patients, especially those in the early fibrotic stages where cardiovascular risk begins to rise.A similar stage-dependent pattern was observed in liver cytolysis markers. ALT and GGT, which reflect the extent of hepatocellular injury and oxidative stress, progressively increased with fibrosis severity. Elevated GGT is often associated with impaired antioxidant defense, mitochondrial dysfunction, and systemic inflammation, all of which are commonly described in metabolic liver disease. The significant reduction in ALT and GGT after therapy suggests that the combined treatment helped restore hepatocyte integrity, reduce oxidative damage, and modulate inflammatory responses. These enzymatic changes confirm the therapeutic potential of Phosphogliv Urso and lisinopril, whose hepatoprotective and anti-inflammatory properties likely contributed to the observed improvements.Metabolic parameters also showed a clear relationship with fibrosis progression. Before treatment, LDL levels rose substantially from F0 to F2 while HDL levels declined, indicating a worsening atherogenic profile. Such dyslipidemia is well recognized in NAFLD and plays a critical role in the development of cardiovascular complications, including endothelial dysfunction and accelerated atherosclerosis. The post-treatment decline in LDL and increase in HDL levels indicate significant metabolic improvement, likely driven by the combined effects of liver-directed therapy, improved inflammatory status, and better blood pressure control. These changes support the concept that NAFLD treatment should not focus solely on hepatic health but must also address the systemic metabolic impairments that accompany the disease.Non-invasive markers of liver health, including Young’s modulus and the Fatty Liver Index (FLI), showed strong diagnostic and prognostic value. Both indicators increased progressively with fibrosis stage, confirming their utility in assessing NAFLD severity. After treatment, a significant reduction in liver stiffness was noted, particularly in early stages, suggesting partial reversibility of fibrosis and improvement in inflammatory and metabolic conditions. The marked decrease in FLI across all groups likewise highlights the therapy’s effectiveness in reducing hepatic fat accumulation. These improvements underline the importance of using dynamic, non-invasive tools for monitoring disease progression and treatment response.Taken together, the results of the present study reinforce the understanding that NAFLD is a systemic disorder involving multiple pathological pathways, including inflammation, oxidative stress, dyslipidemia, insulin resistance, and endothelial dysfunction. Fibrosis represents a key turning point where hepatic pathology begins to manifest more profoundly in the cardiovascular system. This integrated pathophysiology underscores the importance of early detection and targeted therapy, as timely treatment can slow or partially reverse adverse metabolic and cardiovascular changes. The clear therapeutic benefits observed in this study demonstrate that even short-term combined treatment can yield measurable improvements in biochemical, metabolic, and hemodynamic parameters.Although the study presents strong evidence, certain limitations should be acknowledged. The exclusive inclusion of male patients limits generalizability to the broader population, and the relatively short duration of therapy does not allow for full evaluation of long-term fibrosis regression. Nevertheless, the improvements observed over two months indicate promising potential for sustained benefits with prolonged treatment. Future studies should expand the demographic diversity, include longer follow-up periods, and incorporate additional biomarkers to further elucidate the mechanisms linking NAFLD progression and cardiovascular risk.Overall, the study highlights the close interaction between hepatic and cardiovascular dysfunction in NAFLD and demonstrates that comprehensive treatment targeting metabolic and fibrotic mechanisms can produce substantial clinical benefits. The improvement in liver enzymes, lipid levels, blood pressure, liver stiffness, and FLI collectively indicate that a multifaceted therapeutic approach is essential for effective management of NAFLD and prevention of associated cardiovascular complications.
5. Conclusions
- Thus, the progression of liver fibrosis in NAFLD represents not only a localized hepatological disorder but also a systemic pathology that contributes to the development of cardiovascular complications. The intensification of fibrogenesis is accompanied by disturbances in lipid and carbohydrate metabolism, activation of inflammatory cascades, increased oxidative stress, and insulin resistance. All these factors lead to pathological changes in the vascular wall, impair endothelial function, and accelerate the development of atherosclerosis. Consequently, the degree of fibrosis can be considered an independent marker of cardiovascular risk.In this context, timely and effective treatment of NAFLD aimed at reducing the degree of steatosis and fibrosis exerts not only hepatoprotective but also pronounced cardioprotective effects, promoting improvements in hemodynamic parameters, normalization of the lipid profile, and reduction in the frequency of cardiovascular complications. This underscores the necessity of a comprehensive approach to NAFLD therapy from both hepatological and cardiological perspectives.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML