-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4511-4514
doi:10.5923/j.ajmms.20251512.68
Received: Sep. 4, 2025; Accepted: Oct. 2, 2025; Published: Dec. 22, 2025

Preventive Measures for Gastropathy in Rheumatoid Arthritis
Djabbarova Miyassar Bobokulovna
Senior Teacher of the Department of Traditional Medicine and Occupational Diseases of the Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
Correspondence to: Djabbarova Miyassar Bobokulovna, Senior Teacher of the Department of Traditional Medicine and Occupational Diseases of the Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The article presents data on the frequency of gastropathy development in rheumatoid arthritis (RA) patients receiving treatment with non-selective and selective non-steroidal anti-inflammatory drugs (NSAIDs), which are the most popular means of suppressing the symptoms of this disease. NSAIDs can cause serious complications from the gastrointestinal tract (GIC) in the form of erosive-ulcerative lesions of the gastro-duodenal zone and the development of esophagitis. Taking into account risk factors, prescribing safer selective NSAIDs and gastroprotectors allows for a reduction in the frequency of these complications.
Keywords: Rheumatoid arthritis, Nonsteroidal anti-inflammatory drugs (NSAIDs), Gastropathy
Cite this paper: Djabbarova Miyassar Bobokulovna, Preventive Measures for Gastropathy in Rheumatoid Arthritis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4511-4514. doi: 10.5923/j.ajmms.20251512.68.
1. Relevance
- Rheumatoid arthritis (RA) is an autoimmune rheumatic disease of unknown etiology, characterized by chronic erosive arthritis and systemic damage to internal organs, leading to early disability and shortening of patients' life expectancy. RA is a common and one of the most severe human diseases, which determines the great medical and socio-economic significance of this pathology [8,11,16]. In the absence of adequate therapy, RA causes persistent disability in half of the patients in the first 3-5 years of the disease and leads to a significant reduction in their life expectancy, primarily due to a high risk of developing comorbid diseases, including cardiovascular diseases, osteoporosis, severe infections, tumors [8,11,16]. According to the literature, gastrointestinal tract lesions in rheumatoid arthritis account for 13 to 62% of cases and occupy a significant place among extra-articular manifestations of this disease. At the same time, the available literature lacks sufficient material on studying the state of the gastrointestinal tract in rheumatoid arthritis [2,5,9].In the mid-1990s. The impressive statistics of the 20th century dictated the need to talk about the “epidemic” of this pathology. It has been proven that the use of non-selective NSAIDs increases the risk of gastrointestinal bleeding by more than 4 times. The frequency of NSAID gastropathy development reached 4% in those who regularly received NSAIDs, and the total number of deaths caused by this pathology reached 5-15 per 100,000 population per year. The widespread introduction into clinical practice of safer selective cyclooxygenase-2 inhibitors (COG-2) and effective “gastroprotective” drugs (GPPs) [8,11,14,16].Existing international regulatory documents recommend stratifying patients receiving NSAIDs into risk groups according to their existing risk factors, which allows for the determination of treatment and prevention tactics for NSAID gastropathy [8,14,16].Nonsteroidal anti-inflammatory drugs (NSAIDs) are widely used in modern medicine. Patients often take them without consulting a doctor. Annually, approximately 300 million people worldwide regularly or sporadically receive NSAIDs, with no more than one-third being prescribed by a doctor. [1]. The risk of gastrointestinal bleeding in patients receiving NSAIDs increases by 3-5 times, ulceration by 6 times, and the risk of death from complications related to gastrointestinal tract involvement by 8 times. Up to 40-50% of all cases of acute gastrointestinal bleeding are caused by NSAIDs [11,15].The aim of the study is. To demonstrate the relationship between the clinical manifestations of NSAID gastropathy and the endoscopic features of the upper gastrointestinal tract in patients with rheumatoid arthritis.
2. Materials and Methods
- 104 patients with significant RA, predominantly seropositive, with moderate activity of the pathological process, were examined. The average age of patients was 52.6 ± 11.4 years. The gastrointestinal tract pathology was determined by a thorough clinical examination. Endoscopic examination was performed using the “Pentax-EC-3890 hn” device.
3. Results and Discussion
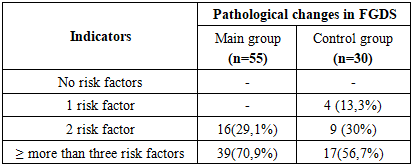
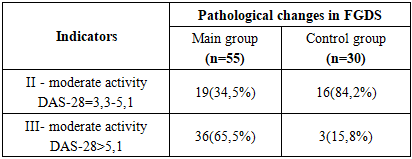
- The study included 138 patients aged 19-90 years who were hospitalized with rheumatoid arthritis for 6 months (January-June) in the rheumatology department of the Bukhara Regional Multidisciplinary Medical Center. The diagnosis of pneumatic aphtitis was made based on the clinical criteria ACR (1987) and ACR/EULAR (2010). When studying the history of the disease, the following data were taken into account: the patient’s age, the nature of the disease, its duration, and the presence of the disease.The disease activity level was assessed using the DAS-28 (Disease Activity Score) scale. When determining the frequency of gastropathy risk factors in patients with pneumatic appetite, their genesis, gender, age, stressful states, alcohol consumption, smoking, Helicobacter pylori detection, and the frequency of gastropathy symptoms were also assessed. Analysis of 104 patients in the scientific study revealed that 74 (71,1%) patients with rheumatoid arthritis exhibited signs of gastropathy, which allowed us to identify the main 74 and 30 control groups.EFGDS and gastric radiography data were used to determine the risk of gastropathy development. The progression of gastropathy in these patients was determined by gastric mucosal radiography using a modified F.L. Lanza scale and barium radiography.Analysis of the study showed that out of 74 patients, 58 (78.4%) were women with a mean age of 51.9±11.6 years, 16 (21.6%) were men with a mean age of 55.2±10.8 years, while in the control group, out of 30 patients, 17 (56.7%) were women with a mean age of 42.3±9.2 years, and 13 (43.3%) were men with a mean age of 44.2±12.8 years.Analysis of the age classification adopted by the World Health Organization (WHO) in 2021 revealed that 15 (20.3%) patients were aged 18-44, 39 (52.7%) were aged 45-59, 17 (22.9%) were aged 60-74, and 3 (4.1%) were aged 75-90.According to the DAS-28 index, 1 patient (1.4%) showed remission, 2 patients (2.7%) showed low activity, 42 (56.8%) showed moderate activity II, and 29 (39.1%) showed high activity III (Figure 1).
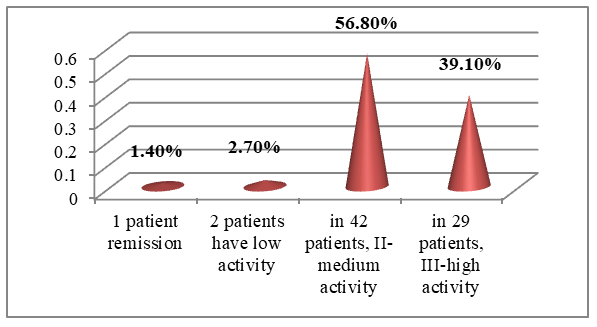 | Figure 1. Analysis of disease activity in patients |
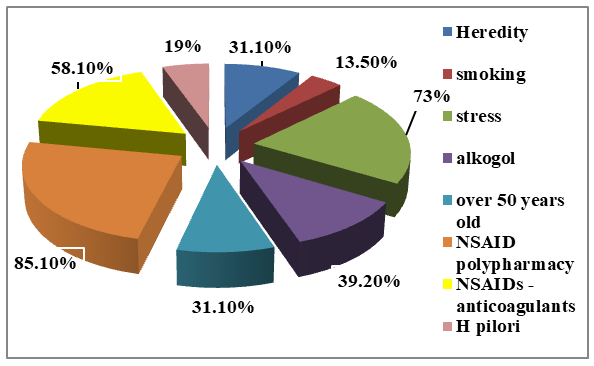 | Figure 2. The frequency of gastropathy risk factors |
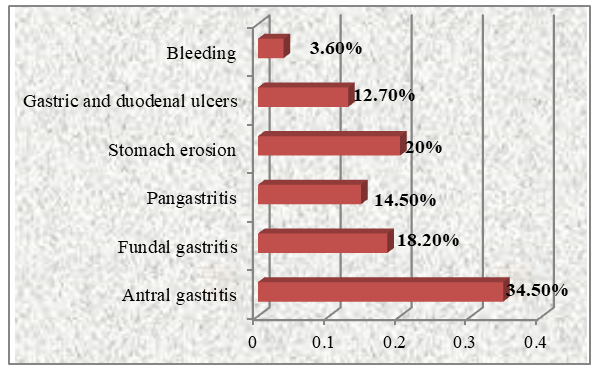 | Figure 3. FGDS results in 55 patients with gastropathy |
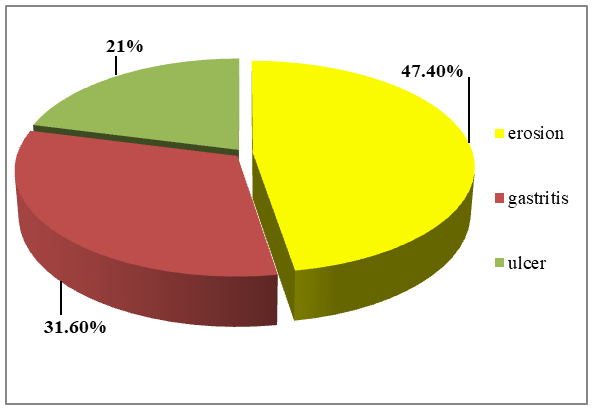 | Figure 4. The results of the radiological examination of 19 patients |
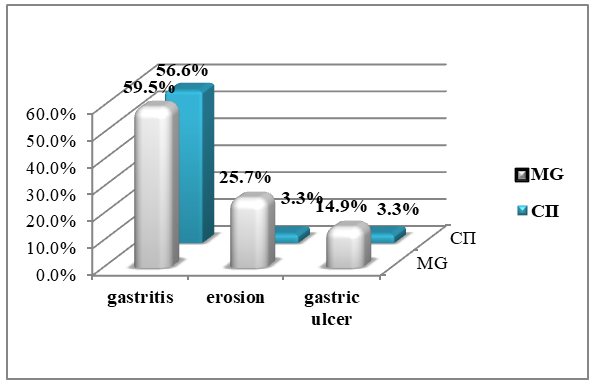 | Figure 5. Analysis of indicators in the main and control groups |
|
|
4. Conclusions
- 1. NSAID gastropathy was most often detected in women with autoaggression factors (stress, heredity, gender, drug polypharmacy, H. pylori, antiplatelet agents), as well as NSAIDs administered for more than five years.2. It is characterized by imbalance between the clinical picture of NSAID gastropathy and endoscopic manifestations of inflammation in the stomach. The mild symptomatic clinical course of NSAID gastropathy corresponds to clear endoscopic and morphological signs of anthral gastritis activity.3. Endoscopic examination of RA patients allowed for a detailed description of the clinical and functional state arising from the aggressive action of NSAIDs and glucocorticosteroids on the gastric mucosa, scientifically substantiated the inclusion of gastric mucosa protective drugs in the complex therapy of patients to prevent inflammation of the gastric mucosa and subsequent severe complications.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML