-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4508-4510
doi:10.5923/j.ajmms.20251512.67
Received: Nov. 26, 2025; Accepted: Dec. 20, 2025; Published: Dec. 22, 2025

Features of the Immune Status in Patients with Various Clinical Forms of Chronic Suppurative Hidradenitis
U. Yu. Sabirov1, A. B. Rakhmatov1, Sh. K. Yuldasheva2
1Doctor of Medical Sciences, Professor, Republican Specialized Scientific and Practical Medical Center of Dermatovenereology and Cosmetology, Ministry of Health of the Republic of Uzbekistan
2Researcher, Republican Specialized Scientific and Practical Medical Center of Dermatovenereology and Cosmetology, Ministry of Health of the Republic of Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Chronic suppurative hidradenitis (HS) is a recurrent inflammatory disease of apocrine gland–bearing skin regions. Despite its prevalence, the immunopathogenesis remains poorly defined. Objective: To investigate the features of immune status in patients with different Hurley stages of HS. Methods: Thirty-six patients with HS (Hurley I–III) were studied using flow cytometric immunophenotyping of peripheral blood lymphocytes (CD3, CD4, CD8, CD16, CD20, CD45). Findings were compared with 20 healthy controls. Results: Early-stage HS (Hurley I) showed nearly normal immune parameters. In Hurley II–III, significant decreases in CD3⁺ and CD4⁺ T-lymphocytes, increased CD8⁺ cytotoxic T-cells, reduced CD4/CD8 ratio, and elevated CD20⁺ B-cells and CD16⁺ NK-cells were observed. Conclusion: Chronic suppurative hidradenitis is associated with progressive systemic immune dysregulation, with a clear correlation between immune imbalance and disease severity.
Keywords: Hidradenitis suppurativa, Chronic suppurative hidradenitis, Immune status, T-lymphocytes, CD3, CD4, CD8, B-cells, NK-cells, Immunoregulation
Cite this paper: U. Yu. Sabirov, A. B. Rakhmatov, Sh. K. Yuldasheva, Features of the Immune Status in Patients with Various Clinical Forms of Chronic Suppurative Hidradenitis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4508-4510. doi: 10.5923/j.ajmms.20251512.67.
Article Outline
1. Introduction
- Hidradenitis suppurativa (HS), or chronic suppurative hidradenitis, is a chronic, recurrent, and debilitating inflammatory dermatosis that affects apocrine gland–bearing regions such as the axillae, groin, and perianal areas; it manifests with painful nodules, abscesses, sinus tracts, and scarring, leading to severe impairment of patients’ quality of life [1].The global prevalence of HS is estimated to be 1–4%, with a predominance among young and middle-aged women [2]. Despite significant clinical relevance, the pathogenesis of HS remains incompletely understood. Growing evidence suggests that immune dysregulation involving both innate and adaptive immune responses plays a pivotal role [3,4].While earlier research primarily focused on cytokine dysregulation and inflammatory mediators [5], less is known about the cellular immune mechanisms and their alterations across disease stages. Understanding the immune status of patients with various clinical forms of HS can contribute to elucidating disease mechanisms and developing individualized treatment strategies [6].Objective:To assess the features of immune status in patients with different Hurley stages of chronic suppurative hidradenitis.
2. Materials and Methods
- Study populationThe study included 36 patients diagnosed with HS according to standard clinical criteria. The distribution by Hurley stage was as follows:• Stage I — 6 patients (16.7%)• Stage II — 14 patients (38.9%)• Stage III — 16 patients (44.4%)The control group consisted of 20 healthy volunteers matched by age and sex. All participants provided written informed consent.ImmunophenotypingPeripheral blood lymphocytes were analyzed using flow cytometry with monoclonal antibodies against CD3, CD4, CD8, CD16, CD20, and CD45. The following parameters were evaluated: total T-lymphocytes (CD3⁺), helper T-cells (CD3⁺CD4⁺), cytotoxic T-cells (CD3⁺CD8⁺), B-lymphocytes (CD20⁺), natural killer (NK) cells (CD3⁻CD16⁺), and the immunoregulatory index (CD4/CD8 ratio).Statistical analysisData were analyzed using Student’s t-test with p < 0.05 considered statistically significant. Results are expressed as mean ± SD. Pearson’s correlation coefficient was used to assess relationships between immune parameters and clinical stages.
3. Results
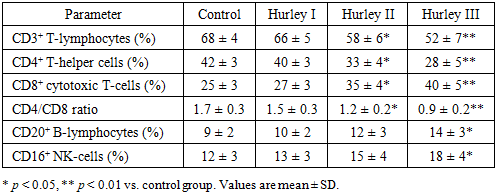
- A total of 36 patients (18 females, 18 males; mean age 34.7 ± 7.5 years) participated in the study. Most cases corresponded to moderate-to-severe disease (Hurley II–III). The control group included 20 age- and sex-matched healthy individuals. Baseline hematologic indicators did not differ significantly between groups.Flow cytometry revealed progressive alterations in lymphocyte subpopulations with advancing disease stage (Table 1).
|
4. Discussion
- The study demonstrates progressive immune dysregulation in chronic suppurative hidradenitis, involving both adaptive and innate immunity. The principal findings include decreased total CD3⁺ T-lymphocytes, pronounced deficiency of CD4⁺ helper T-cells, relative predominance of CD8⁺ cytotoxic lymphocytes, reduced CD4/CD8 ratio, and increased CD20⁺ B-lymphocytes and CD16⁺ NK-cells.These observations resonate with current literature that characterizes HS as a systemic immunoinflammatory disorder rather than a purely local skin disease. Increased cytotoxic activity and CD8⁺ infiltration in lesional tissue have been reported previously [2,4], and peripheral CD8⁺ expansion observed here parallels those cutaneous findings. Dysregulated Th1/Th17 cytokine expression (e.g., IL-17, TNF-α) described by other authors likely underlies the helper T-cell dysfunction we observed [6]. Elevated immunoglobulins and B-cell activation reported elsewhere correspond to the increased CD20⁺ counts in advanced stages [7].Mechanistically, chronic antigenic stimulation in HS (e.g., due to follicular rupture, biofilms, or persistent inflammation) may lead to activation-induced cell death or exhaustion of CD4⁺ T-cells, while sustained antigen exposure and tissue damage drive expansion of CD8⁺ and NK-cell populations. The decline of the CD4/CD8 ratio below 1.0 in severe cases reflects a loss of immune homeostasis comparable to patterns seen in other chronic inflammatory and autoimmune disorders [3,4].From a clinical perspective, these immune alterations have diagnostic and therapeutic implications. Monitoring lymphocyte subpopulations could support disease staging, aid in prognostication, and guide the use of targeted immunomodulatory therapy (e.g., anti-TNF-α or anti-IL-17 agents). Restoration of CD4/CD8 balance during treatment may represent a useful biomarker of therapeutic response.Study limitations include the modest sample size and cross-sectional design, which preclude causal inference and longitudinal assessment of immune dynamics. Future studies incorporating serial sampling, cytokine profiling, tissue immunohistochemistry, and response-to-therapy analyses are warranted to strengthen the translational relevance of these findings.
5. Conclusions
- In conclusion, progression of hidradenitis suppurativa is accompanied by stepwise immune dysregulation—most notably CD4⁺ depletion and CD8⁺/CD20⁺/CD16⁺ expansion—underscoring the systemic immunological nature of HS and supporting integrative immunological approaches in patient management.
Funding
- No external funding was received for this study.
Ethical Approval
- The study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional ethics committee.
Conflict of Interest
- The authors declare no conflicts of interest.
Author Contributions
- • U.Yu. Sabirov — Conceptualization, study design, supervision, manuscript drafting, final approval.• A.B. Rakhmatov — Patient recruitment, clinical examinations, laboratory analysis, data collection.• Sh.K. Yuldasheva — Statistical analysis, data interpretation, literature review, critical revision of the manuscript.All authors read and approved the final manuscript and agree to be accountable for all aspects of the work.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML