-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4501-4507
doi:10.5923/j.ajmms.20251512.66
Received: Nov. 26, 2025; Accepted: Dec. 16, 2025; Published: Dec. 22, 2025

Comprehensive Diagnostic Algorithm for Craniocervical Junction Anomalies: Clinical–Neurophysiological Rationale, Quantitative Indices, and Application Results
R. O. Ismailova, Sh. T. Arzikulov, A. I. Ibragimov, R. M. Yuldashev
Republican Specialized Scientific and Practical Medical Center of Neurosurgery, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Craniocervical junction (CCJ) anomalies vary widely in presentation and often combine ventral compression, posterior fossa crowding, and instability. Diagnostic pathways remain inconsistent, and isolated radiological indices do not fully capture functional impairment. Objective: To develop and evaluate a comprehensive diagnostic algorithm for CCJ anomalies integrating craniometric measurements, 3D volumetric assessment, and neurophysiological testing. Methods: Twenty-six symptomatic patients with MRI/MDCT-confirmed CCJ anomalies were studied: basilar invagination (n=6), platybasia (n=3), Chiari malformation (n=8), and Chiari with syringomyelia (n=9). Craniometric assessment included the Grabb–Oakes index, Chamberlain/McGregor/Harris lines, Redlund-Johnell & Hansson, Schmidt-Fischer angle, craniocervical angle (CVA), Klaus index, atlanto-dens interval (ADI), Cruveilhier joint, and Wackenheim line. Posterior fossa volume was analyzed using 3D CT, and dynamic MDCT was performed to detect atlantoaxial instability. Neurophysiology included BAEP interpeak latencies (RI–RV, RIII–RV) and stimulation EMG of median and tibial nerves. Results: Basilar invagination showed the greatest number of pathological craniometric indices, while platybasia primarily demonstrated reduced posterior fossa volume. Chiari malformation presented combined ventral and dorsal imbalance, and Chiari with syringomyelia exhibited the most severe neurophysiological abnormalities. BAEP prolongation and reduced peripheral conduction velocity correlated with structural severity (p<0.05). Conclusion: Integrating craniometry, volumetric imaging, and neurophysiology improves diagnostic accuracy and functional interpretation of CCJ anomalies. The proposed algorithm enhances risk stratification and supports individualized management, including the choice between conservative treatment, decompression, and stabilization.
Keywords: Craniocervical junction, Basilar invagination, Platybasia, Chiari malformation, Syringomyelia, Craniometry, BAEP, EMG
Cite this paper: R. O. Ismailova, Sh. T. Arzikulov, A. I. Ibragimov, R. M. Yuldashev, Comprehensive Diagnostic Algorithm for Craniocervical Junction Anomalies: Clinical–Neurophysiological Rationale, Quantitative Indices, and Application Results, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4501-4507. doi: 10.5923/j.ajmms.20251512.66.
Article Outline
1. Introduction
- The craniocervical junction is a biomechanically loaded region where the dynamics of the skull base and upper cervical spine are combined. [1]. Small angular and distance changes can lead to significant clinical consequences (bulbar disorders, long‑tract signs, instability). [2]. Despite accumulating data on the Grabb–Oakes index, Chamberlain and McGregor lines, Harris measurement, and other metrics, real‑world diagnostic pathways remain heterogeneous. The lack of a unified algorithm hampers early identification of patients who should undergo decompression and/or stabilization. This work establishes a step‑by‑step diagnostic protocol and demonstrates its clinical–functional informativeness. Technical aspects of decompression and the role of duraplasty are widely discussed in the literature. [3], [7], [10], [9].
2. Materials and Methods
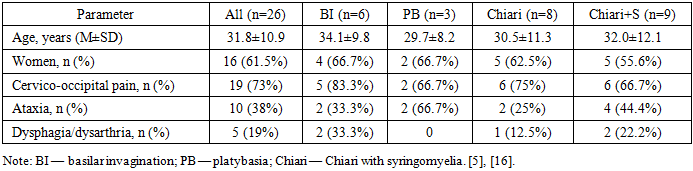
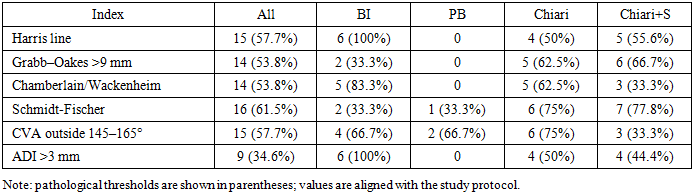
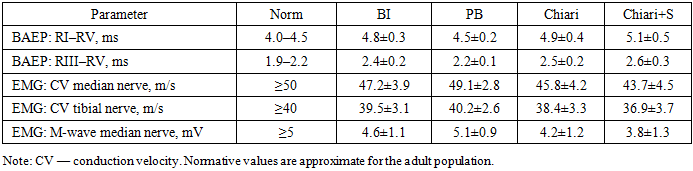
- Study design: single‑center case series (n=26). Inclusion criteria: confirmed CCJ anomalies on MRI/MDCT and the presence of clinical symptoms or neurophysiological deviations. Groups: basilar invagination (n=6), platybasia (n=3), Chiari malformation (n=8), and Chiari with syringomyelia (n=9). [4], [9], [17].Neuroimaging: brain and cervical MRI (T1/T2; assessment of tonsillar ectopia, syringomyelia, medullary compression); functional MDCT of the CCJ (flexion/extension); 3D CT for posterior fossa volume calculation. Craniometry: Grabb–Oakes (normal ≤9 mm), Chamberlain (>5 mm) and McGregor (>10 mm) lines, Harris (≤12 mm), Redlund‑Johnell & Hansson (≤8 mm), Schmidt‑Fischer (124–127°), CVA (145–165°), Klaus index (30–35 mm), ADI (≤3 mm), Cruveilhier joint (≤4 mm), Wackenheim line. Neurophysiology: BAEPs (latencies RI–RV; interpeak intervals RI–RV and RIII–RV; amplitudes); stimulation EMG (conduction velocity and M‑wave amplitude) in the median and tibial nerves. Statistics: descriptive measures (mean±SD), intergroup comparisons using two‑tailed t‑tests, significance set at p<0.05. [11], [12], [13].
3. Results
- Demographic and Clinical Characteristics Mean age was 31.8±10.9 years; 61.5% were women. Predominant complaints: cervico‑occipital pain (73%), vertigo (42%), ataxia (38%), dysarthria/dysphagia (19%). Pyramidal signs and dissociated sensory disturbances were more frequent in Chiari with syringomyelia, while static ataxia predominated in platybasia.
|
 | Diagram 1. Objective clinical symptoms in craniovertebral junction anomaly |
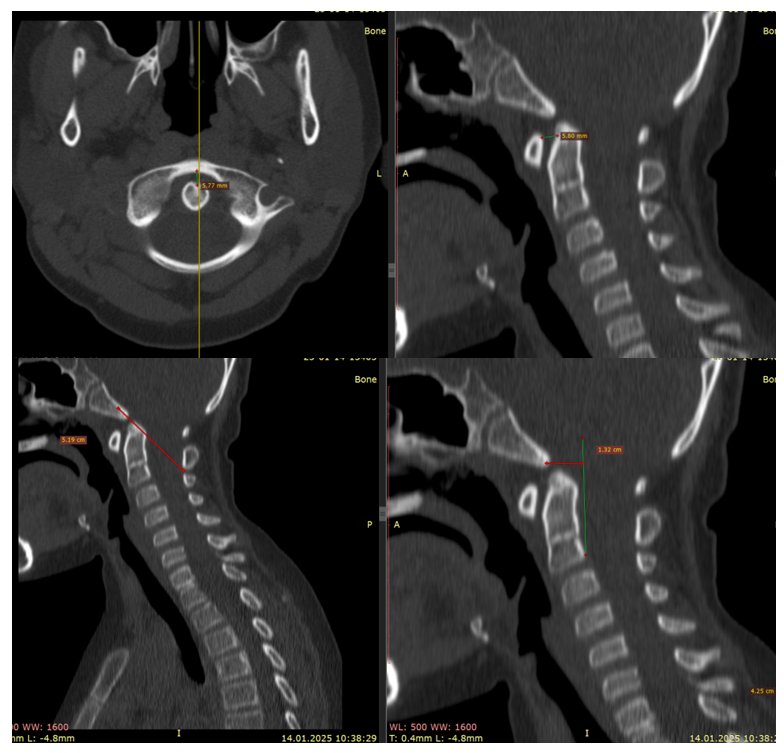 | Figure 1. CT craniometric indices (Harris line, atlanto‑dens interval, Wackenheim line) indicating ventral compression and craniocervical instability |
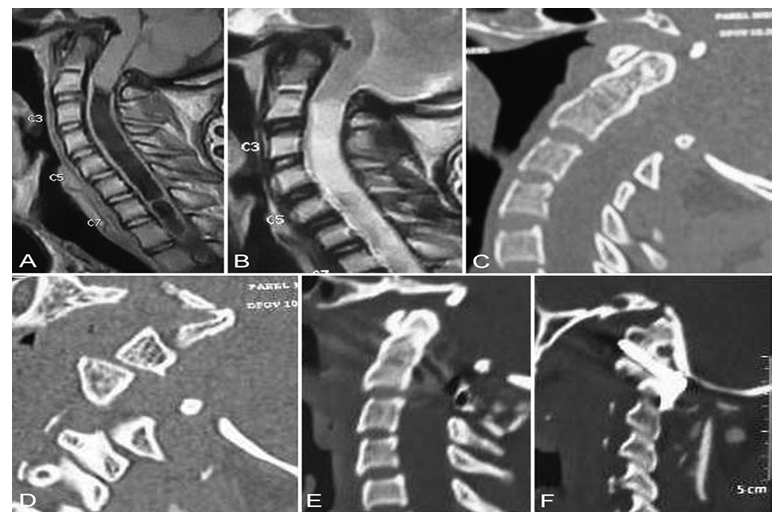 | Figure 2. Types of atlantoaxial dislocation on MDCT |
|
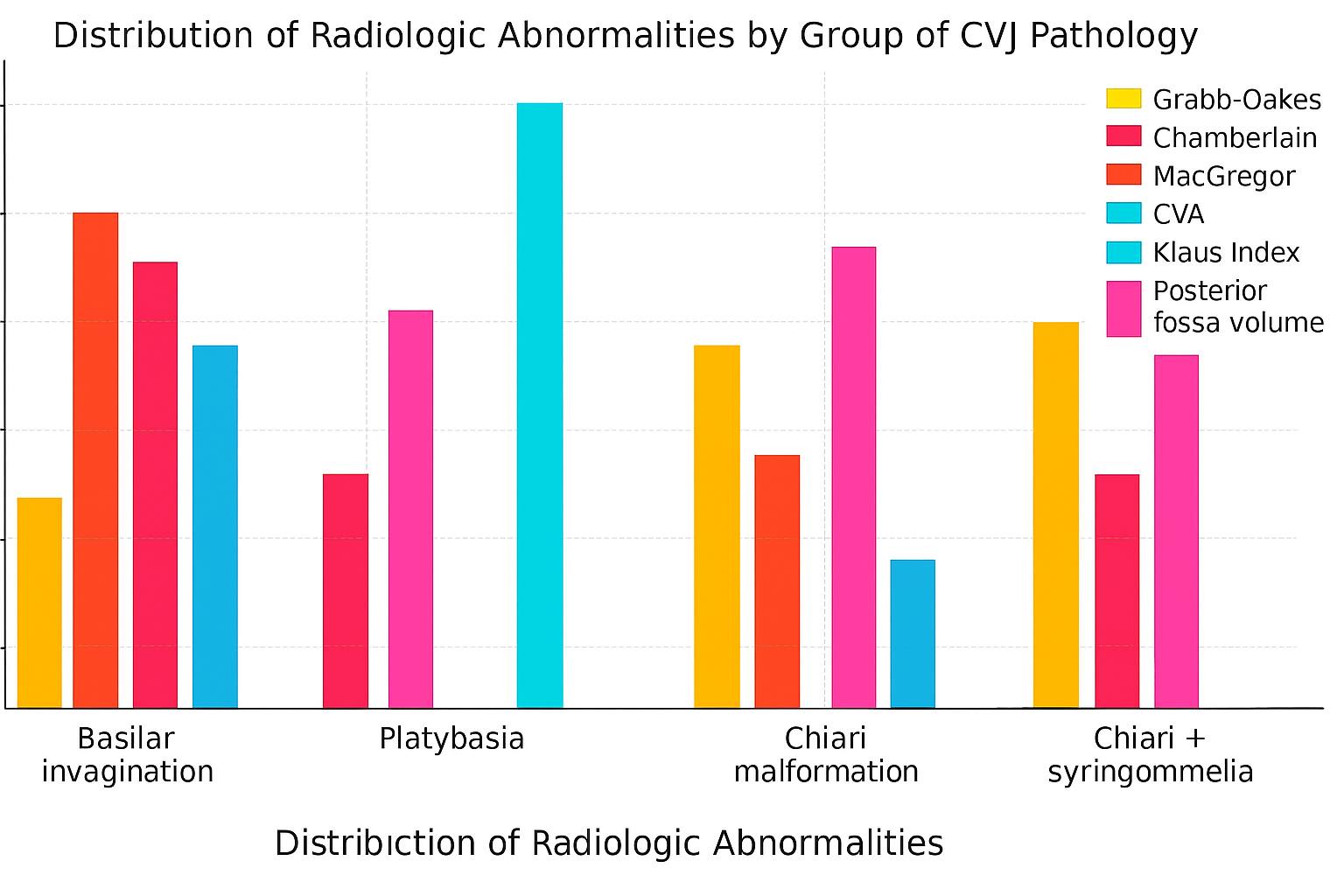 | Diagram 2 |
|
4. Discussion
- Pathophysiologic contextThe spectrum of craniocervical junction (CCJ) anomalies in our cohort—basilar invagination, platybasia, and Chiari I with/without syringomyelia—reflects distinct but overlapping mechanisms of brainstem compression and CSF flow disturbance. Ventral compromise stems from odontoid retroflexion, upward migration of the dens, and occult atlantoaxial instability; dorsal crowding arises from posterior fossa underdevelopment and tonsillar ectopia. These configurations differentially load corticospinal and cerebellobulbar tracts, explaining the predominance of pyramidal signs and bulbar symptoms in ventral compression and ataxia/pain in dorsal crowding.Structure–function correlationsWe found a graded relationship between morphometry and neurophysiology. Metrics of ventral encroachment (Grabb–Oakes > 9 mm, pathological Harris, ADI > 3 mm, abnormal Wackenheim) coincided with BAEP interpeak prolongation (RI–RV, RIII–RV) and reduced motor nerve conduction velocities—changes most pronounced in the Chiari + syringomyelia subgroup. While BAEP indexes brainstem conduction, peripheral conduction slowing likely reflects segmental stress from chronic traction and microvascular compromise in the cervicomedullary junction. In platybasia, conspicuous geometric flattening did not translate into comparable BAEP change, reinforcing that location and direction of compression, not magnitude alone, determine functional impact.Comparative perspective vs. literatureThe literature underscores the limitations of single‑index decision‑making and the clinical heterogeneity of CCJ pathology [5–7,9]. Goel and others highlight that unrecognized atlantoaxial instability may underlie a subset of Chiari and syringomyelia, with fixation yielding clinical and radiographic improvement even without extensive decompression [16,18]. Traditional decompression‑first strategies remain effective where CSF obstruction predominates without instability [7,9,10]. Our convergence of pathological Harris/ADI values with BAEP delay supports routine instability screening and argues against a one‑size‑fits‑all approach.Algorithm performance and triageThe proposed pathway operates as a triage funnel: (1) clinical screen, (2) MRI for tonsillar ectopia/CSF pathways, (3) 3D CT + dynamic MDCT for quantitative indices (Grabb–Oakes, Harris, Schmidt–Fischer, CVA, ADI, Wackenheim), (4) neurophysiology for functional corroboration. Concordant high‑risk profiles (e.g., Grabb–Oakes > 9 mm + pathological Harris + ADI > 3 mm with BAEP delay) prioritize stabilization combined with decompression; isolated dorsal crowding with normal instability metrics favors decompression alone. Discordant results (borderline morphometry with normal BAEP) justify observation with standardized re‑imaging and repeat BAEP at defined intervals.Clinical phenotypes and thresholdsThree practical phenotypes emerged. (A) Ventral compression–instability: ADI elevation and pathological Harris with retroflexed dens and Wackenheim deviation; patients exhibit long‑tract signs and greatest BAEP delay. (B) Dorsal crowding: reduced CVA and posterior fossa volume with tonsillar descent; pain and ataxia predominate, BAEP changes are mild. (C) Mixed with syringomyelia: multiple craniometric outliers and poorest neurophysiology. These map to widely used thresholds (Grabb–Oakes > 9 mm; ADI > 3 mm) and emphasize confirming borderline cases with dynamic CT before committing to fixation.Implications for individualized managementCoupling structure with function reframes the decompression‑vs‑fixation choice as a testable hypothesis. In instability‑dominant cases, early fixation may mitigate postoperative deterioration reported after decompression‑only strategies [5]. Where dorsal crowding dominates without measurable instability, tailored decompression with duraplasty can restore CSF pulsatility and reduce syrinx volume [7,9,10]. For mixed phenotypes, sequencing fixation and decompression in a single session—or staging when anesthetic risk is high—can be guided by a composite of Grabb–Oakes, Harris/ADI, and BAEP severity. This precision approach also refines counseling about expected pain relief versus neurological recovery, which tracks with preoperative BAEP normalization potential and syrinx chronicity.Measurement standardization and reproducibilityGiven the sensitivity of angle and distance metrics to imaging planes, we advocate strict protocolization: midline sagittal MRI for Grabb–Oakes, neutral/flexion/extension MDCT for Harris/ADI/Wackenheim, and predefined threshold tables. Embedding screenshots of landmarks and storing measurement templates reduce inter‑observer variance and enable longitudinal audit. Semi‑automated tools for angle/line detection may further enhance reliability, particularly for Schmidt–Fischer angle and CVA.Translational outlookProspective validation should link algorithmic strata to hard endpoints (gait/bulbar function, syrinx regression, re‑operation rates, quality of life). The incremental value of CSF flow studies and upright MRI in borderline cases warrants study. Predictive models combining morphometrics and neurophysiology could yield individualized probability scores for instability and response to decompression versus fixation, aiding multidisciplinary decision‑making and patient counseling.Clinical Implications• The algorithm ensures early identification of high‑risk patients and reduces diagnostic delay.• Formalized indices facilitate communication at multidisciplinary boards.• Operation planning (decompression ± stabilization) becomes more evidence‑based, potentially lowering re‑operation rates.
5. Conclusions
- The proposed diagnostic algorithm, based on the integration of craniometric and neurophysiological data, significantly improves the accuracy of patient stratification with craniovertebral junction anomalies. The combined use of the Grabb-Oakes, Harris, Schmidt-Fischer, and ADI indices ensures the early detection of instability and conduction abnormalities. Furthermore, the use of standardized criteria facilitates the unification of clinical decisions and reduces the risk of over- or underdiagnosis. The obtained results confirm the importance of a multidisciplinary approach, as well as the need for further multicenter studies to clarify the prognostic value of the developed algorithm. Thus, the proposed model may become an effective tool for optimizing diagnosis, planning surgical tactics, and improving treatment outcomes for patients with craniovertebral junction anomalies.
Study Limitations
- Single‑center design and a limited sample reduce generalizability; no prospective validation. Long‑term functional outcomes and quality of life were not assessed; multicenter protocols with unified endpoints are needed.
Ethics
- The study was conducted in accordance with the Declaration of Helsinki. Local ethics approval: [RSCN 0025/27 ?03.09.2025].
Funding
- Source of funding: [specify]. The sponsor had no influence on study design, data collection/analysis, manuscript preparation, or the decision to publish.
Conflict of Interest
- The authors declare no conflicts of interest.
Author Contributions (CRediT)
- Conceptualization and design: Sh.T. Arzikulov; Data curation: Sh.T. Arzikulov; Analysis: R.O. Ismailova; Writing: R.O. Ismailova; Sh.T. Arzikulov. Review and approval of the final version: all authors.
Data Availability
- Summary data are available from the corresponding author upon reasonable request.
Abbreviations
- CCJ — craniocervical junction; PF — posterior fossa; BAEP — brainstem auditory evoked potentials; EMG — electromyography; ADI — atlanto‑dens interval; CVA — craniocervical angle.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML