-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4489-4491
doi:10.5923/j.ajmms.20251512.63
Received: Nov. 5, 2025; Accepted: Nov. 27, 2025; Published: Dec. 22, 2025

Disorder of Liver Pigment Metabolism in Experimental Toxic Hepatitis
Akhmedov Kamoliddin Hakimovich1, Ergashov Mansur Abdukarim ugli2
1DSc, Professor, Termez Branch of the Tashkent State Medical Academy, Uzbekistan
2PhD, Assistant, Termez Branch of the Tashkent State Medical Academy, Uzbekistan
Correspondence to: Akhmedov Kamoliddin Hakimovich, DSc, Professor, Termez Branch of the Tashkent State Medical Academy, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Objective of the study: To determine the dependence of pigment metabolism indices on the liver conjugation function in case of carbon tetrachloride toxic hepatitis in hot climate conditions. Materials and methods of the study: The experiment was conducted for 15 days on 32 white Wistar rats weighing 180-220 grams. Results: a sharp decrease in conjugated bilirubin is observed. Conclusions: in hot climate conditions with carbon tetrachloride toxic hepatitis, profound disturbances of the liver pigment metabolism function are observed.
Keywords: Toxic hepatitis, Carbon tetrachloride, Experiment, Rat, Hyperbilirubinemia, Conjugated bilirubin, Unconjugated bilirubin
Cite this paper: Akhmedov Kamoliddin Hakimovich, Ergashov Mansur Abdukarim ugli, Disorder of Liver Pigment Metabolism in Experimental Toxic Hepatitis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4489-4491. doi: 10.5923/j.ajmms.20251512.63.
1. Introduction
- Liver diseases are a pressing issue in modern medicine worldwide, including in our country. Depending on their origin, they include viral and alcoholic hepatitis, with toxic hepatitis being the most common [2]. This pathology is of great medical and social significance due to the high financial costs associated with treatment and rehabilitation of patients, disability, and high mortality. However, there is an increasing incidence of severe forms of hepatitis, which carries the risk of developing chronic liver damage [4,5].Hepatorenal syndrome (HRS) and hepatic encephalopathy (HE) are two typical complications of acute liver injury. Acute liver injury (ALI) is an excellent experimental setting for testing therapeutic strategies and understanding pathophysiological mechanisms [3].Due to its location and the presence of blood vessels and bile ducts directly connecting the liver to the intestines and heart, the liver is strategically positioned to detect toxic compounds, sense metabolic changes, and respond to them through the production of paracrine and endocrine signals [6]. Furthermore, the liver and brain are connected by the parasympathetic and sympathetic nervous systems, enabling two-way communication and further control of liver and body homeostasis by the central nervous system (CNS). While afferent fibers sense and transmit changes in blood composition in the portal vein to the central nervous system, sympathetic fibers of the splanchnic and vagus nerves penetrate the liver parenchyma and liver lobules [7] and the presence of intercellular gap junctions allows for the efficient transmission of autonomic and hormonal feedback signals to control liver function [8]. The liver, in turn, continuously receives nutrients from the gastrointestinal tract and bacterial compounds through the portal system and accepts hormones. As a result, the gastrointestinal tract and liver are involved in various immunological interactions. A striking example here is primary sclerosing cholangitis and the concomitant ulcerative disease colitis. This heterogeneous role of the liver-brain axis is realized asymmetrically via the vagus nerve [5].We studied the toxic effects of carbon tetrachloride on rat liver. Carbon tetrachloride is emitted by chemical plants and is an environmentally harmful chemical toxicant. Its atmospheric concentration is increasing daily. The potential presence of carbon tetrachloride in the liver is manifested by inflammatory reactions caused by free radicals.In liver damage, the conjugation of bilirubin with glucuronic acid in hepatocytes is disrupted, leading to bile backflow into the sinusoids. This necrosis of liver cells is associated with the intrahepatic bile ducts being filled with thick bile and their permeability being impaired. This condition causes an increase in conjugated bilirubin levels in the blood. As a result, intracellular biological oxidation processes are slowed, mitochondrial tissue respiration and associated oxidation are disrupted, and the inhibitory effect of bilirubin, based on inhibited phosphorylation, increases in parallel with the increase in its levels [1]. These issues remain relevant today, and the mechanisms of liver adaptation to xenobiotic exposure require further study, particularly in the context of acute carbon tetrachloride intoxication.Objective of the study: to determine the dependence of pigment metabolism indices on the conjugation function of the liver in carbon tetrachloride toxic hepatitis in hot climates.
2. Materials and Methods
- The experiment was conducted over 15 days on 32 Wistar albino rats weighing 180-220 grams. Intoxication was induced by administering carbon tetrachloride (CTC) to the rats at a dose of 0.2 ml/100 g in 40% olive oil twice daily. Samples were collected for analysis from the control group and the experimental groups on days 3, 7, and 15. Total, conjugated, and free bilirubin in the serum were determined using a Biossays 240 Plus automated biochemical analyzer.
3. Results and Discussion
- It is known from the results obtained in the authors' studies presented in the literature that the indicators of biochemical markers in the blood plasma of experimental animals differ from the results obtained from donor human plasma. Therefore, biochemical parameters from the blood plasma of animals of the intact group were evaluated as a reference unit. As a result, the amount of total bilirubin, one of the main components of bile, was on average - 5.3 μmol/l (bound bilirubin - 1.54 μmol/l; unbound bilirubin 3.76 μmol/l) (Table 1).
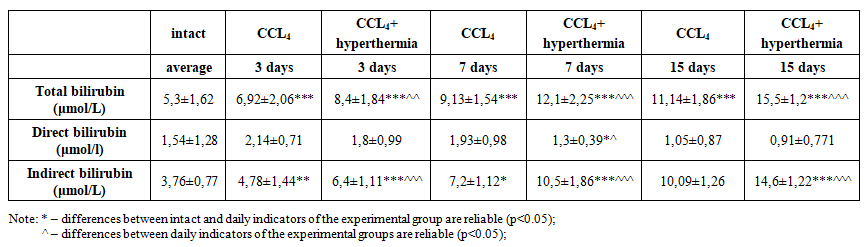 | Table 1. Biochemical parameters in acute toxic hepatitis in hot and normal climates |
4. Conclusions
- Thus, in hot climates, severe disturbances in liver pigment metabolism are observed in cases of carbon tetrachloride toxic hepatitis:1. Hyperbilirubinemia in experimental animals was evident on the third day of the study.It reached its maximum value on the seventh and fifteenth days of the study.2. In carbon tetrachloride toxic hepatitis occurring in hot climates, hyperbilirubinemia is primarily due to unconjugated bilirubin. This indicates that liver pigment metabolism is seriously impaired.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML